Typical Flutter Ablation as an Adjunct to
Catheter Ablation of Atrial Fibrillation
Electrophysiology Unit,Cardiology Service,Hopital Cantonal Universitaire de Geneve, Switzerland.
During the past decades there has been a consistent evolution of both surgical and catheter-based techniques for the treatment of stand-alone atrial fibrillation, as alternatives or in combination with anti-arrhythmic drugs. Transcatheter ablation has significantly improved outcomes, despite often requiring multiple procedures and with limited success rates especially in presence of persistent atrial fibrillation. Surgical procedures have dramatically evolved from the original cut-and-sew Maze operation, allowing nowadays for closed-chest epicardial ablations on the beating heart.
Recently, the concept of a close collaboration between the cardiac surgeon and the electrophysiologist has emerged as an intriguing option in order to overcome the drawbacks and suboptimal results of both techniques; therefore, the hybrid approach has been proposed as a potentially more successful strategy, allowing for a patient-tailored therapeutical approach.
We reviewed the recent advancements either from the transcatheter and surgical standpoint, with a peculiar focus on the current option to merge both techniques along with an up-to-date review of the preliminary clinical experiences with the hybrid, surgical-transcatheter treatment of stand-alone atrial fibrillation.
Correspondence to: Dipen Shah, Electrophysiology Unit,Cardiology Service,Hopital Cantonal Universitaire de Geneve, Switzerland.
Typical atrial flutter and atrial fibrillation are frequently observed to coexist.1 In the current context of interventional electrophysiology, curative or at least definitive ablation is available for both arrhythmias. Despite their coexistence, it is not clear whether typical flutter ablation is necessary in all patients undergoing catheter ablation of atrial fibrillation. The following review explores the pathophysiology of both arrhythmias, their interrelationships and the available data pertaining to this theme.
Pathophysiology of Typical Atrial Flutter
Typical atrial flutter is a well known macroreentrant right atrial tachycardia. Although the underlying mechanism of this arrhythmia was intensively debated in the 20th century, a combination of entrainment and activation mapping showed that the re-entrant circuit of typical atrial flutter consisted of counter-clockwise or clockwise activation around the tricuspid annulus.2 The circuit is anteriorly constrained by the tricuspid valve annulus while internal short circuiting (and therefore termination of reentry) is prevented by a vertical zone of block extending from the superior vena cava to the inferior vena cava.3 Clinical studies of the artificial induction of typical atrial flutter concluded that aggressive programmed stimulation or burst stimulation was necessary but did not give consistent results and frequently induced atrial fibrillation.4 The initiation of typical atrial flutter requires the development of unidirectional block in the cavo-tricuspid isthmus and the transformation of a frequently incomplete line of intercaval block (in sinus rhythm or during pacing at slower rates) into a complete zone of block.
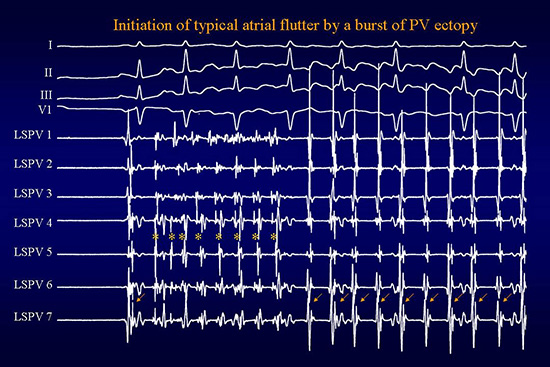
The spontaneous initiation of typical atrial flutter (like those of other arrhythmias) is clearly difficult to study. Based on the limited available data, Waldo et al suggested that typical atrial flutter is necessarily preceded by atrial fibrillation.5 Evidence from Tai et al6 and from our work in Bordeaux7 suggest that in patients with clinical evidence of both atrial fibrillation as well as flutter, a burst of rapid ectopic activity from the pulmonary veins initiates typical flutter(fig 1). Endocardial electrograms demonstrate a short period of rapid and frequently irregular activation, the most rapid rates being observed at the ostia of the pulmonary veins. The surface ECG as well as intracardiac activation are difficult to distinguish from atrial fibrillation, although careful examination can discern organised, similar looking but very rapid atrial activity which is usually masked by overlying ventricular QRSTs. These features suggest a very rapid and irregular tachycardia rather than atrial fibrillation. Whatever be the underlying mechanism, whether tachycardia or fibrillation, close coupling intervals and/or rapid rates may be necessary to convert an often incomplete line of intercaval block into one that is complete from the superior to the inferior vena cava. Once the intercaval line of block is rendered complete by rapid activation, the quiescence or slowing of competing rapid activation is necessary in order to allow the flutter to manifest. In the EP lab, endocardial recordings in patients with both atrial flutter and fibrillation often show a sudden transition to regular, slower and passive activation of the pulmonary veins (fig 1).
Figure 1. Surface ECG leads I, II, III ,V1 and 7 bipolar endocardial recordings from the left superior pulmonary vein showing the initiation of typical atrial flutter. The sinus beat is followed by a repetitive rapid discharge (*) originating from the left superior PV associated with the absence of clear P waves on the surface ECG. Typical atrial flutter is observed on the surface ECG coincident with sudden slowing and regularisation in the left superior PV, suggesting quiescence of LSPV discharge. Note that each PV discharge (*) is preceded by a sharp spike whereas during sinus rhythm and typical flutter this spike forms the terminal part of the electrogram (arrows) indicating passive activation (from left atrium to PV myocardium)
Pathophysiology of Atrial Fibrillation
Atrial fibrillation is characterised by the diffuse biatrial occurrence of rapid and irregular activity, frequently at cycle lengths less than 200 ms. The defining characteristic is rapid activity with temporal and spatial variation producing the characteristic surface ECG appearance of the absence of organised P waves.
Two main hypotheses have been advanced to explain this atrial activation pattern. Lewis and Scherf, invoked a locus of abnormally rapid impulse formation, that attempts to drive both atria at very rapid rates but because of heterogeneous electrophysiologic properties results in irregular activation with temporal and spatial variation – so called fibrillatory conduction.8 In a contemporary context, this tachyarrhythmic locus may be single or multiple, and may be active constantly or intermittently and in concert with others or non-synchronously. The underlying mechanism could be abnormal automaticity or triggered activity or even a small reentry circuit. A localised treatment strategy e.g. limited ablation may be expected to eliminate the arrhythmia.
A variant of this theme is the mother wave hypothesis wherein a macro-reentrant circuit or rotor, drives an atrial myocardial substrate and because of fibrillatory conduction results in temporally and spatially variable activation.8 Ablation limited to transection of a critical segment of the mother wave or ablation of the rotor should terminate and eliminate atrial fibrillation. Because of their association, it has often been suggested that typical atrial flutter could be responsible for some episodes of atrial fibrillation.
On the other hand, as suggested by Moe, simultaneously coexisting multiple wandering wavelets may be responsible for rapid and irregular activation. Because of the absence of strict impulse recirculation over the same pathway and the absence of an excitable gap, this form of activation cannot be termed re-entrant in the traditional sense.9 Diffuse participation of wide regions of the atria is integral to this concept and in keeping with this, in experimental studies, it becomes impossible to induce atrial fibrillation in progressively smaller pieces of atrial tissue, thus suggesting the necessity for a critical quantum of anatomical space or tissue mass. However, in the presence of sufficiently slow conduction and/or shortened refractory periods, tissue mass may no longer be a limiting factor. It follows therefore that ablation treatment of a limited amount of atrial tissue is unlikely to suppress the inducibility of this form of atrial fibrillation.
These two mechanisms may not be mutually exclusive. In the absence of diffuse abnormalities, a driving locus would have to be constantly active in order to maintain fibrillation and therefore periods of driver quiescence result in termination and reversion to sinus rhythm. On the other hand, in the presence of diffuse substrate abnormalities such as fibrosis, a driving locus could induce a self sustaining form of multiple wavelet activation. Alternatively, additional drivers may play a role in sustaining rapid irregular activation independent of the influence of the original initiating driver.
Direct support for the above hypotheses remains difficult to obtain in human subjects – given that current technology has significant limitations when applied to activation mapping of irregularly irregular rhythms over the complex terrain of the right and left atrium. Sequential point by point mapping allows coverage of most parts of both atria but cannot be used with any degree of accuracy in atrial fibrillation because of temporal variations in activation patterns. Multi-electrode arrays while providing simultaneous recordings over a larger segment of the endocardium, cannot assure contact with key areas in the atria. Nevertheless, the clinical relevance of pulmonary vein drivers is clear beyond doubt. Intra-cardiac recordings have documented the initiating role of abnormal discharges from pulmonary venous myocardium.10 When PV isolation is performed in patients with paroxysmal atrial fibrillation during atrial fibrillation, sinus rhythm is restored with successful isolation of 1, 2, 3 or all pulmonary veins in most if not all patients. On the other hand, in patients with long-standing persistent or permanent atrial fibrillation, the efficacy of PV isolation is much lower.11
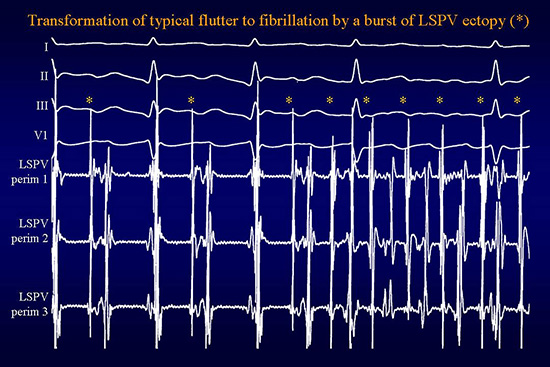
In patients with frequent spontaneous onset atrial flutter and fibrillation, we can clearly demonstrate the role of pulmonary vein discharges during electrophysiologic procedures in the EP lab. Bursts of PV discharges initiate a very rapid tachycardia, frequently conducted only I ntermittently to the atria. The subsequent appearance of atrial flutter on the surface ECG is clearly preceded by with a slowing and organisation of activity in the pulmonary vein which initiated the arrhythmia (fig 1). Conversely, sustained rapid and often fractionated activity in a pulmonary vein, during sinus rhythm or even during ongoing flutter (fig 2), heralds the transformation of typical atrial flutter into fibrillation. In some cases, termination of atrial flutter may also be associated with a burst of activity from the pulmonary veins.
Figure 2. Surface ECG leads I, II, III ,V1 and 3 bipolar endocardial recordings from the left superior pulmonary vein showing the transformation of typical atrial flutter to atrial fibrillation. During typical flutter, active left superior PV discharge (sharp spike preceding farfield atrial activation, *) alternates with passive activation till the middle of the tracing, where a sustained LSPV discharge begins and transforms flutter into fibrillation, clearly evident on the surface ECG
In experimental models of pericardial inflammation, the transition from atrial flutter to fibrillation has been studied with multielectrode plaque mapping. In this model, the gradual coalescence of short lines of block in the right atrial free wall into a longer continuous one is responsible for the transformation from fibrillation to a flutter involving a macroreentrant circuit on the right atrial free wall.2 The epicardial location of the inflammation, correlates well with the right atrial free wall circuit but may not be representative of human atrial fibrillation which typically occurs in the absence of epicardial inflammation.
On the basis of the above considerations, in the patient undergoing catheter ablation for atrial fibrillation, one may opt to 1) ignore the presence of typical atrial flutter and elect to target only the atrial fibrillation or 2) perform cavotricuspid isthmus ablation as an adjunct in all patients undergoing AF ablation or 3) perform adjunctive cavotricuspid isthmus ablation in selected patients undergoing AF ablation. Adjunctive cavotricuspid isthmus ablation may have no clinical salutary effect or may be helpful by preventing the de novo appearance of atrial flutter or by reducing the recurrence of atrial fibrillation itself.
Wazni et al12 randomised consecutive patients with documented atrial fibrillation and typical atrial flutter to undergo PV-LA junction disconnection combined with cavotricuspid isthmus (CTI) ablation vs PV-LA junction disconnection alone. They observed that those patients who did not undergo CTI ablation frequently developed typical atrial flutter, but only during the first 8 weeks. Thereafter, either with electrical cardioversion alone or with supplementary anti-arrhythmic drug therapy (for 8 weeks), they were able to show that no more recurrences of typical flutter occurred. Three patients however did require CTI ablation in this group within this initial period. The mean follow up for this study was around13 months, and it is possible that a longer follow-up could have shown more recurrence of typical atrial flutter. Equally, asymptomatic atrial flutter may have been missed as would have asymptomatic atrial fibrillation. The core of the hypothesis tested in this study was clearly that PV-LA junction isolation could eliminate all initiating triggers of atrial flutter in this group of patients with co-existing atrial fibrillation. But typical flutter is frequently observed in isolation (without atrial fibrillation) and even the patient with clear cut elimination of atrial fibrillation by catheter ablation has residual supraventricular ectopy, a potential if not actual trigger. Moreover, there are electrophysiologically proven non-PV triggers of atrial fibrillation and these may well induce atrial flutter after elimination of a PV substrate of atrial fibrillation.13 Interestingly, while Wazni et al seem to have shown the disappearance of typical atrial flutter after antiarrhythmic drug use, other studies have shown that the use of IC antiarrhythmic drugs actually favours the appearance or stabilisation of typical atrial flutter in patients with atrial fibrillation.14 Certainly, PV isolation plays an important therapeutic role by reducing if not eliminating the most common and the most rapid triggers. The first few weeks to months after PV isolation are often characterised by residual ectopy, and our mapping studies have shown that while conduction recovery within isolated PVs plays a role, many ectopics originate from the atrial edges of the PV isolation lesion. These may be very sensitive to antiarrhythmic drug therapy, perhaps because of their origin from partially ablated tissue. Based on the findings of Wazni et al, it may be argued that performing CTI ablation in these patients with both atrial fibrillation and atrial flutter may reduce the re-hospitalisation and drug administration and cardio-version that was necessary in their study during the initial post-ablation period. In our experience, typical atrial flutter can manifest well after the initial 8 weeks, and as detailed below, we therefore advocate an individualised strategy.
Scharf et al15 described their experience with 133 consecutive patients undergoing catheter ablation for atrial fibrillation, specifically for evaluating the clinical significance of inducible atrial flutter during the atrial fibrillation ablation procedure. In their study, rapid burst atrial pacing was performed from the coronary sinus at cycle lengths of 200 to 180 ms for 10 to 20 seconds at least 5 times. Despite the stated aim of evaluating the significance of inducible atrial flutter, 21% of their patients with either a history of or inducible typical atrial flutter underwent cavotricuspid isthmus ablation during the procedure so that it is difficult to derive systematic insights from the subsequent clinical development of typical atrial flutter. They did observe however that the occurrence of typical atrial flutter during the procedure is predictive of symptomatic atrial flutter during follow up, and that the elimination of (symptomatic) AF by catheter ablation is not associated with freedom from atrial flutter. Unlike the experience of Wazni et al, symptomatic typical atrial flutter occurred during follow-up in 21% of patients free of recurrent AF. Moreover 29% of those with recurrent AF also developed typical atrial flutter (p= ns). Conversely, there was a trend towards a lower recurrence rate of AF in the group of patients who had undergone cavotricuspid isthmus ablation (p=0.07) leading the authors to suggest that atrial flutter may have been responsible for some episodes of AF. It could be speculated that the additional cavotricuspid ablation allowed more time to monitor the pulmonary venous isolation for conduction recovery. The authors nevertheless concluded that catheter ablation of the cavo-tricuspid isthmus should be considered in conjunction with PV isolation to avoid the need for a second catheter ablation procedure.
We chose to prospectively evaluate an individualised strategy of performing adjunctive cavotricuspid isthmus ablation only for those AF ablation patients with a documented history of typical atrial flutter or those in whom typical atrial flutter was observed during the procedure i.e. spontaneous onset or spontaneous conversion of AF to atrial flutter or triggered during catheter manipulation.16 We chose not to perform rapid burst stimulation or programmed stimulation because typical flutter is known to be difficult to induce even in patients with a previous history of this arrhythmia (6.2% of attempts in one study, (4), is equally likely to induce AF and eliminating the induction protocol helped shorten and simplify the procedure. Additionally, the occurrence of typical flutter without an induction protocol probably demonstrates a greater susceptibility to this arrhythmia. 75 of 188 patients underwent CTI ablation and AF ablation; the remaining AF ablation alone. Of the 75, 47 had previously documented typical flutter, 13 developed typical flutter for the first time during the procedure and 15 had had a previous typical flutter ablation. Over a mean follow –up of 30 months, AF and typical flutter recurrences were similar in both groups: AF recurrence 25% in the group with CTI ablation vs. 28% in the group without CTI ablation; typical flutter 1.3 vs 2.6% respectively, p=ns for both comparisons. Therefore withholding CTI ablation in patients undergoing AF ablation without prior or intraprocedural documentation of typical flutter does not result in higher rates of typical flutter occurrence during follow-up. Further, recurrence of AF is also not any lower in the patients undergoing adjunctive right atrial ablation.
The reason for the frequent coexistence of typical atrial flutter and atrial fibrillation is not known, however, the two arrhythmias have substrates in contralateral atria. The interplay between the two arrhythmias might be explained by their sharing of common triggers – ectopy of pulmonary vein origin - as well as the requirement for rapid close coupled activation to potentiate and complete the intercaval zone of block in order to create the typical flutter circuit.
The lack of significant effect of typical flutter ablation on the recurrence of atrial fibrillation after catheter ablation emphasises the independence of the two arrhythmias. Despite the fact that typical flutter ablation is one of the safest and and among the simplest of ablations, there is no justification for routinely performing it as an adjunct to atrial fibrillation ablation. Typical flutter ablation can be difficult in a minority of patients, and the additional procedure times and indwelling catheter times may increase procedural risks, particularly thromboembolic risks.
On the other hand, patients with previously documented typical atrial flutter should undergo this adjunctive ablation because of the high likelihood of recurrence, whether early or late. For those patients without any previous documentation of flutter, the occurrence of typical flutter during the procedure should also prompt consideration of adjunctive ablation. The duration of flutter, whether sustained or non-sustained, and the mechanism of its induction are both indicators of an individual’s propensity to develop typical atrial flutter. On the basis of the available evidence, the adjunctive ablation of sustained typical flutter observed during the procedure without programmed or burst stimulation is advisable as it results in similar flutter occurrence rates in follow-up compared to those without any previous flutter.