Nonfluoroscopic Ablation of Atrial Fibrillation Using Cryoballoon
Mansour Razminia, M.D., F.A.C.C.
, Hany Demo, M.D. , Carlos Arrieta-Garcia, M.D., Oliver J. D’Silva, M.D., Theodore Wang, M.D., F.A.C.C, Richard F. Kehoe, M.D., F.A.C.C.
Advocate Illinois Masonic Medical Center, 836 West Wellington Avenue, Chicago, IL, USA.
The conventional method of cryoballoon ablation of atrial fibrillation involves the use of fluoroscopy for visual guidance. The use of fluoroscopy is accompanied by significant radiation risks to the patient and the medical staff. Herein, we report our experience in performing successful nonfluoroscopic pulmonary vein isolation using cryoballoon ablation in 5 consecutive patients with paroxysmal atrial fibrillation
Five consecutive patients with paroxysmal atrial fibrillation underwent cryoballoon ablation for pulmonary vein isolation using a nonfluoroscopic approach. Pre-procedural cardiac computed tomography or cardiac magnetic resonance imaging was not performed in any patient. A total of twenty pulmonary veins were identified and successfully isolated (100%) with the guidance of intracardiac echocardiography and 3-dimensional electroanatomic mapping. No fluoroscopy was used for the procedures. There were no major procedural adverse events.
In an unselected group of patients undergoing cryoballoon ablation, a nonfluoroscopic approach is feasible and can be performed safely and effectively while eliminating the risks associated with radiation to both the patient and the medical staff.
Correspondence to: Mansour Razminia, M.D., F.A.C.C. 3000 North Halsted Street, Suite 803, Chicago, IL 60657
The conventional method of cryoballoon ablation (CBA) of atrial fibrillation (AF) involves the use of fluoroscopy for visual guidance. The use of fluoroscopy is accompanied by significant radiation risks to the patient and the medical staff.1 Radiation exposure has been linked to an increased lifetime risk of malignancies, skin injuries, genetic defects, and cataracts.2 Given the long duration of AF ablation procedures, the common use of pre-procedural cardiac computed tomography (CCT) scans for definition of the cardiac and extracardiac anatomy, and the not infrequent need for repeat ablation procedures, the cumulative lifetime radiation exposure is potentially significant.3 In addition, the use of fluoroscopy during ablation procedures exposes the staff to the hazards of radiation and increases the potential for orthopedic injuries from the cumulative adverse effects from bearing the weight of leaded apparel.4
The commonplace use of 3-dimensional (3D) electroanatomic mapping (EAM) systems along with intracardiac echocardiography (ICE) allows the operator to decrease fluoroscopy times.5 Nonfluoroscopic radiofrequency ablation (RFA) of AF has been described before as well as the ablation of other atrial and ventricular arrhythmias.6,7 The notion that such complex ablation procedures could be performed without using any fluoroscopy is promising from a safety standpoint for both the patients and electrophysiology (EP) staff.
CBA has offered a single-delivery approach to pulmonary vein isolation (PVI) that is an alternative to the traditional point-to-point RFA methodology.8-10 In all previous reports of CBA, positioning of the cryoballoon has been accomplished using fluoroscopy with fluoroscopy times averaging 20 to 60 minutes.8,11,12 Herein, we report our experience in performing successful nonfluoroscopic PVI using CBA in 5 consecutive patients with paroxysmal AF (PAF). To our knowledge this represents the first report of successful CBA without the use of fluoroscopy.
In this series, we report the results of five consecutive patients with PAF and documented failure of at least one antiarrhythmic drug who underwent CBA at our institution. A nonfluoroscopic approach was used in all 5 patients. All were informed that while efforts to minimize or eliminate fluoroscopy would be made, fluoroscopy would be immediately available if necessary to enhance procedural success or safety.
Pre-procedural CCT or cardiac magnetic resonance imaging (CMR) was not performed in any patients in the series. A pre-procedural transesophageal echocardiogram (TEE) was obtained in 1 patient prior to the ablation. Pre-procedural anticoagulation included warfarin, rivaroxaban, and aspirin, (Table 1). Post-procedural anticoagulation included continuation of warfarin (2 patients), and the use of rivaroxaban the evening of the procedure (3 patients).
Table 1. Baseline Patient Characteristics
| Total Number of Patients |
5 |
| Age (yrs) |
59 +/-13 |
| Male |
5/5 |
| Hypertension |
0 |
| Diabetes |
1/5 |
| CAD |
0 |
| Left Atria AP diameter (mm) |
38 +/- 4 |
| LVEF % |
51 +/- 17 |
| NYHA Class |
|
| None / I |
4/5 |
| II |
0 |
| III or higher |
1/5 |
| Previous Cardioversion |
1/5 |
| History of Flutter |
0 |
| CHADS2 Score |
0.4 +/- 0.9 |
| CHA2DVASC Score |
0.8 +/- 1.3 |
| Prior RF Ablation |
1/5 |
| Number of Prior Drugs Failed |
|
| 0 |
0 |
| 1 |
5/5 |
| 2 |
0 |
| 3 |
0 |
| Failure more than 1 AF drug |
0 |
| Efficacy Failed AF drugs |
|
| Flecainide |
2/5 |
| Propafenone |
1/5 |
| Sotalol |
1/5 |
| Dofetalide |
1/5 |
| Anticaogulation |
|
| ASA |
2/5 |
| Coumadin |
2/5 |
| Rivaroxaban |
1/5 |
| Baseline Medication Class |
|
| ACE-I |
1/5 |
| ARB |
0 |
| Beta Blocker |
4/5 |
| Calcium Channel Blocker |
0 |
| Cardiac Glycoside |
0 |
| Cholesterol Lowering |
3/5 |
| Diuretic |
1/5 |
Values are mean +/- SD or % CAD: coronary artery disease; AP: anterior-posterior; LVEF: left ventricle ejection fraction; ACE-I: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker; AF: atrial fibrillation;
Dexmedetomidine was used for procedural sedation under the supervision of an anesthesiologist.
Initial Catheter Positioning And Creation Of Right Atrial Geometry
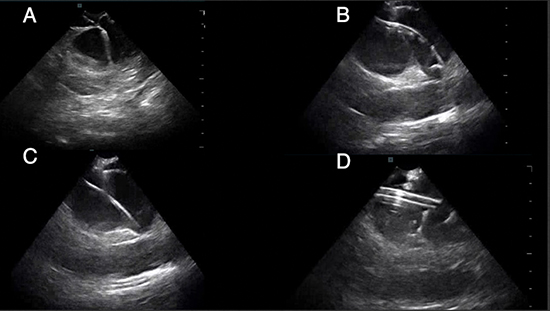
Percutaneous femoral venous access was obtained by the modified Seldinger technique guided by vascular ultrasound. Two 8-Fr sheaths (Input PS, Medtronic, Minneapolis, MN, USA) were placed in the right femoral vein; one 10-Fr long sheath (Check-Flo Performer introducer, Cook, Bloomington, IN, USA) and another 7-Fr sheath (Input PS, Medtronic) were placed in the left femoral vein. A 9-Fr ICE catheter (ViewFlex, St. Jude Medical, St. Paul, MN, USA) was inserted through the 10-Fr long sheath in the left femoral vein and advanced to the inferior vena cava (IVC) while maintaining an echo free-space at the tip of the transducer (Figure 1), The ICE catheter was then advanced to the right atrium (RA).
Figure 1. ICE images showing: (A) the transseptal needle as it punctures the interatrial septum, (B) the J-wire with tip in the LSPV ostium through the transseptal sheath, (C) the J-wire advanced into the LSPV and the transseptal sheath removed, and (D) the 15-Fr FlexCath Advance sheath with the tip proximal to the ostium of the LSPV
Next, a 6-Fr decapolar catheter (InquiryTM, Ten TenTM Diagnostic Catheter, St. Jude Medical) was inserted into the left femoral vein and advanced to the IVC under the guidance of the EAM system (Ensite VelocityTM, St. Jude Medical). Geometry of the IVC was collected as the decapolar catheter was advanced to the IVC. The appearance of electrograms on the distal bipoles of the decapolar catheter indicated that the catheter tip had reached the border of the inferior RA and IVC. The decapolar catheter was then advanced into the RA and into the SVC. The RA-SVC junction was marked by a loss of electrograms signals on the proximal poles of the decapolar catheter as the catheter entered the SVC. Geometry of the SVC was collected on the EAM system. The decapolar catheter was then withdrawn into the RA. Under ICE and EAM guidance, 3D geometries of the RA, coronary sinus (CS), and right atrial appendage were collected. The decapolar catheter was then withdrawn from the body, and used to guide the placement of the transseptal sheath (see below). Upon completion of the transseptal puncture, the decapolar catheter was repositioned in the CS, where it remained for the rest of the procedure.
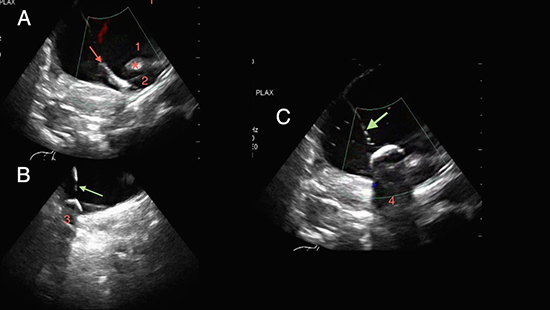
A weight based heparin bolus was administered prior to the transseptal puncture. The activated clotting time (ACT) was monitored throughout the procedure and intravenous heparin boluses were administered to target an ACT of 350 to 450 seconds. A 180 cm 0.032-Fr J-wire (Guide Wire J Tip Fixed Core, Medtronic) was inserted into the right femoral vein and advanced until it was confirmed to be in the RA under ICE. The short introducer sheath was then withdrawn and exchanged for a transseptal sheath-dilator assembly (SL-0, St. Jude Medical). This assembly was advanced over the J-wire approximately 10 to 20 centimeters, depending on the patient size, with the goal of placing the tip of the assembly at the mid IVC level. The wire and dilator were removed. The decapolar catheter, which had been previously used to create geometry, was then advanced beyond the tip of the sheath into the IVC. Once the catheter exited the sheath, it would become visible on the EAM system. Next, the catheter was subsequently advanced to the SVC under EAM guidance. The transseptal sheath was then advanced over the decapolar catheter until a distortion of the catheter shape on the EAM system was noted, thus confirming the sheath’s location in the SVC. The decapolar catheter was removed. The J-wire and dilator were then placed through the sheath with the J-wire tip approximately 10 to 15 centimeters distal to the dilator tip to avoid trauma by the dilator. With the dilator inserted, the wire was removed and the transseptal needle (NRG ® RF Transseptal needle, Baylis Medical, Montreal, QC, CA) was placed in the sheath-dilator assembly. The transseptal assembly was withdrawn into the RA until tenting of the interatrial septum was seen on ICE (Figure 2 ). RF was then delivered through the transseptal needle and the septum was punctured under ICE guidance. The appearance of bubbles in the left atrium (LA) on ICE after saline injection confirmed a successful transseptal puncture. Hemodynamic pressure monitoring was also used to confirm entry into the LA.
Figure 2. Red arrow: cryoballoon; green arrow: FlexCath sheath; 1: LA appendage, 2: left superior PV, 3: right inferior PV, 4: left inferior PV; *: ridge between the left superior PV and LA appendage. intracardiac echocardiographic images showing (A) cryoballoon positioned in the left superior PV prior to inflation, (B) the alignment between the FlexCath sheath and the cryoballoon while occluding the right inferior PV, and (C) the alignment between the FlexCath and the cryoballoon while occluding the left inferior PV
Creation Of Left Atrium And Pulmonary Vein Geometry
A 10-pole circular mapping catheter (Reflexion™, Steerable Catheter, St. Jude Medical) was advanced through the transseptal sheath into the LA under ICE and EAM guidance. The circular mapping catheter was then used to create a 3D geometry of the pulmonary veins (PVs) and LA on the EAM. The ostia of the PVs, as visualized by ICE, were delineated on the EAM.
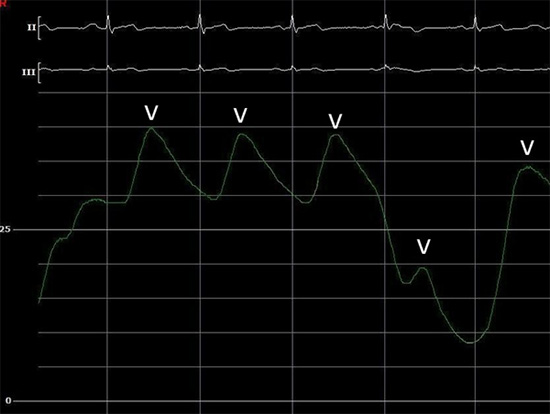
In preparation for CBA, the circular mapping catheter was withdrawn from the body. The J wire was advanced through the transseptal sheath into the left superior PV (LSPV), the location of which was confirmed under ICE (Figure 2). While maintaining the J wire in the LSPV, the SL-0 sheath was withdrawn from the body and exchanged for a 15-Fr steerable sheath (FlexCath® Advance, Medtronic). The 10.5-Fr 28 mm cryoballoon (Arctic Front Advance™, Medtronic) and an 8-pole circular mapping catheter (Achieve™, Medtronic) were placed through the FlexCath into the LA. The 10-pole circular mapping catheter was inserted through the right femoral vein and advanced under EAM guidance to the SVC in order to stimulate the phrenic nerve during CBA of right-sided PVs. The 8-pole circular mapping catheter was advanced into the LSPV. The balloon was inflated and moved to the antrum of the vein. ICE was utilized to confirm the alignment of the transseptal sheath and the balloon prior to cryo application (Figure 3). Achievement of a pulmonary capillary wedge pressure waveform on hemodynamic pressure monitoring signified complete occlusion of the vein as previously described13. A lack of color-flow Doppler within the targeted PV served as an additional indicator of adequate PV occlusion. Cryotherapy was then applied. The goal temperature was minus 40 to minus 50 degrees Celsius. Each application lasted for 120 to180 seconds, aiming to achieve PV conduction block within the first 80 seconds.14 The same technique was utilized for the remaining PVs. Prior to CBA for the right-sided PVs, the 10-pole circular mapping catheter was used to pace the SVC at high output to monitor the integrity of the phrenic nerve during cryo applications. Diaphragmatic capture was monitored through compound motor action potentials (CMAP) as previously described15,16 and by manual tactile monitoring of right-sided diaphragmatic stimulation. If the CMAP amplitude diminished or a palpable loss of diaphragmatic contraction was noted, cryotherapy was immediately discontinued.
After CBA, the cryoballoon and 8-pole circular mapping catheter were withdrawn from the patient. The 10-pole circular mapping catheter was withdrawn from the SVC and reinserted into the LA through the transseptal sheath. This catheter was then placed in each of the PVs and used to re-confirm entrance and exit block. After electrical isolation was established, intravenous isoproterenol at up to 20 mcg/min was given to ensure the absence of AF inducibility.
Figure 3. Pressure –guided RLPV occlusion. The pressure wave shows a characteristic pulmonary artery pressure curve with the presence of “V” waves only, indicating complete occlusion of the pulmonary vein
At the end of procedure, intravenous protamine was administered and all catheters were removed. Flowable hemostat (D-Stat, Vascular Solutions, Minneapolis, MN, USA) was administered at the femoral vein puncture sites and manual pressure was applied.
Baseline characteristics of the patients in the study are listed in (Table 1). All patient were in sinus rhythm at the time of the procedure. The average patient age was 59 years and all 5 patients were male. The average LA diameter was 38 mm. Four patients had normal left ventricular systolic function and no symptoms of heart failure while one patient had severely depressed left ventricular systolic function and NYHA Class III symptoms. Only 1 of the 5 patients had undergone a prior PVI, which was over 10 years ago. All 5 patients had recurrent AF despite antiarrhythmic drug therapy. Three patients had a CHA2DS2VASc score of 0 and the remaining two had CHA2DS2VASc scores of 1 and 3 and were on warfarin at the time of the procedure.
Cryballoon Ablation And Acute Procedural Success
Results pertaining to the cryoballoon procedure and outcomes are listed in (Table 2,Table 3). The 28 mm diameter cryoballoon was used for all 5 patients. A total of 20 PVs were targeted for isolation, of which 20 were successfully isolated. An average of 11 +/- 3 cryo applications per patient were used and the average temperature reached during cryo was minus 45.0 degrees Celsius. Procedural success was defined as complete PVI. Following successful PVI, none of the patients exhibited AF that required the creation of additional lines with a focal cryo catheter or RF catheter. Anatomical variants encountered in the series include a patient who had both a left common PV and a right middle PV as well as another patient with core triatriatum sinister.
Table 2. Procedure Details
| Procedure Details and Effectiveness |
N = 5 |
| Procedure Duration * |
181 +/- 42 |
| Fluoroscopy Duration |
0 |
| Total Cryoablation Duration |
30 +/- 2 |
| Time to trans-septal |
25 +/- 10 |
| Average Cryo application per patient |
11 +/- 3 |
| Abnormal Atrial and PV Anatomy |
|
| LCPV |
1/5 |
| RMPV |
1/5 |
| Cor Triatrium |
1/5 |
| Additional Cryo Lines needed |
0 |
| Use of ICE |
5/5 |
| Average Cryoballoon Temperature |
-45.0 C |
* All durations and time units are reported in minutes PV: pulmonary vein; LCPV: left common pulmonary vein; RMPV: right middle pulmonary vein; ICE: intracardiac echocardiography
Table 3. Procedure Outcome
| Procedure Outcomes |
N = 5 |
| Acute Procedural Success |
5/5 |
| Need for additional Cryo lines |
0 |
| Repeat Cryoablation |
0 |
| Need for RF Ablation |
0 |
| Need for DCCV |
0 |
RF: radiofrequency; DCCV: direct current cardioversion • Patients continued their same pre-procedural AF drug therapy for the 3 months blanking period. • All patients were discharged with 3 weeks event monitor.
Results are listed in (Table 2). The average total procedure duration was 181 +/- 42 minutes. The average time from obtaining venous access to completion of the transseptal puncture was 25 +/- 10 minutes. The duration of the procedure was longest for our first patient (233 minutes) and was shortest (117 minutes) in our third patient, who notably had no anatomic anomalies. Cryo was applied for an average of 30 +/- 2 minutes per patient.
Procedure-related adverse events are listed in (Table 4). There were no major complications. The only complication noted was the incidental finding of an asymptomatic pseudoaneurysm, which spontaneously resolved after warfarin was withheld for 4 days.
Table 4. Adverse Events
| Procedure Adverse Events |
N=5 |
| Strokes |
0 |
| TIA |
0 |
| Tamponade |
0 |
| Myocardial Infarction |
0 |
| Hemorrhage Requiring Transfusion |
0 |
| New Atrial Flutter |
0 |
| Atrial Esophageal Fistula |
0 |
| Death |
0 |
| Arteriovenous Fistula |
0 |
| Pseudoaneurysm * |
1/5 |
| Phrenic Nerve Palsy |
0 |
| PV Stenosis |
0 |
TIA: transient ischemic attack; PV: pulmonary vein * Patient’s pseudoaneurysm was incidentally discovered when patient underwent a diagnostic CT abdomen and pelvis for suspicious hepatic mass discovered on ICE during the ablation procedure. The pseudoaneurysm spontaneously resolved on follow up ultrasound 4 days later.
A nonfluoroscopic approach for radiofrequency ablation of a wide variety of arrhythmias has been previously described.5-7,17-23 To our knowledge, this series is the first to describe an entirely nonfluoroscopic approach to CBA of atrial fibrillation.
The patients in this series were an unselected group of 5 consecutive patients with symptomatic drug-refractory AF and included 1 patient who had recurred AF despite a prior RF ablation. Pre-procedural CCT or CMR was not performed on any of the patients as real time ICE imaging during the procedure was felt to adequately delineate LA and PVs anatomy. Despite the absence of pre-procedural CT or CMR, PVI was successfully completed on all 5 patients without the use of fluoroscopy, even when anatomical variants such as a left common PV, right middle PV, and cor triatriatum sinister were encountered. Furthermore, no additional applications were required to achieve complete PVI or eliminate spontaneous or isoproterenol-induced AF.
The learning curve was steep for nonfluoroscopic CBA. As our institution has adapted nonfluoroscopic catheter ablation for all arrhythmias since 2010, many of the steps in the procedure, such as ICE and EP catheter positioning, EAM geometry collection, and transseptal puncture, were already a routine. One challenge unique to nonfluoroscopic CBA is the need to maintain proper visual alignment of the balloon and sheath via the use of ICE instead of fluoroscopy. As contrast and fluoroscopy were not used, adequate PV occlusion was confirmed by hemodynamic pressure monitoring and color flow Doppler on ICE. The avoidance of contrast-induced renal injury is an additional benefit of the nonfluoroscopic approach.
The average procedure duration was reasonable at 181 +/- 42 minutes, especially as this series represents our first experience with CBA. The improvement in procedural time, from 233 minutes in the first patient to117 minutes in the third patient, probably reflects a growing proficiency derived from procedural experience.
This series demonstrates that nonfluoroscopic CBA can be performed safely and effectively. With the exception of one small pseudoaneurysm that spontaneously resolved, there were no other procedure-related adverse events. Phrenic nerve injury did not occur in this small series, perhaps due to utilizing CMAP technique.
While the study demonstrates that nonfluoroscopic CBA can be safely and effectively performed in a small number of patients, the extent to which these results can be broadly applied will require future multicenter randomized controlled trials comparing the long term outcomes of fluoroscopic versus nonfluoroscopic approaches to CBA. Also, as this was the initial experience with the nonfluoroscopic CBA, there is insufficient long-term data on the durability of PVI or the late-appearance of adverse events such as atrial-esophageal fistula, esophageal injury, or PV stenosis. Finally, a cost analysis was not performed so it is unclear if the nonfluoroscopic cryoballoon approach would be cost-effective for centers that do not routinely use 3D EAM systems or ICE during CBA procedures.
Radiation risk to patients and EP staff during catheter ablation procedures is considerable and efforts should be made to try to eliminate unnecessary exposure whenever possible. Because the ablation of AF often involves longer procedure times and hence prolonged fluoroscopy duration, employing a nonfluoroscopic approach for ablation of AF is of particular importance and benefit. As the use of CBA for the treatment of paroxysmal AF becomes more widely accepted, the number of patients who undergo this technique will likely increase. Our initial experience with nonfluoroscopic CBA demonstrates that this approach is feasible and can be performed safely and effectively. This nonfluoroscopic approach eliminates the risks associated with radiation to both the patient and the medical staff as well as the risk of renal injury related to the use of contrast.