Role of the Auotnomic Nerves system in the
Creation of Substrate for Atrial Fibrillation
Rishi Arora,MD, Alan H. Kadish, MD
Northwestern Memorial Hospital, Chicago, IL, USA.
Correspondence to: Alan H. Kadish M.D,Northwestern Memorial Hospital,251 East Huron,Feinberg 8-542,Chicago, IL 60611.
Atrial fibrillation (AF) is the most common sustained arrhythmia disturbance and is associated with significant morbidity and mortality. In recent years, the pulmonary veins (PVs) and posterior left atrium (PLA) have been shown to play a significant role in the genesis of AF.
These regions have been shown to possess unique structural, electrophysiological and calcium (Ca2+) handling characteristics, all of which appear to contribute to substrate for AF.
The pathophysiology of AF is complex, and several mechanisms have been thought to contribute to the electrophysiologic and structural substrate for this arrhythmia. These mechanisms include fibrosis, stretch, inflammation and oxidative stress. In addition, neurohumoral factors have also been invoked for their possible contribution to the creation of electrophysiologic substrate for AF.3,4 An important neurohumoral factor that has been studied fairly extensively for its involvement in AF is the autonomic nervous system.
We have recently shown that the PLA is more densely innervated than the rest of the left atrium in normal hearts, and has a unique autonomic profile that appears to contribute to AF substrate.5,6 Recent studies also suggest a role for the autonomic nervous system in the creation of substrate for AF in the setting of heart disease, with animal studies demonstrating an increase in sympathetic as well as vagal nerve firing prior to the onset of atrial arrhythmias in the setting of congestive heart failure.7
In this review, we examine the contribution of both clinical as well as animal studies to our understanding of the role of the autonomic nervous system in AF. Animal models-specifically canine models-have proved invaluable in studies that require pharmacological or surgical manipulation of the autonomic nervous system to better understand the relative role of the sympathetic and/or parasympathetic nervous system in AF. For example, much of our understanding of the contribution of collections of autonomic neurons i.e. ganglionated plexi (GPs) to AF substrate comes from canine studies that ablated these GPs in a systematic fashion.8-14 Animal models also allow the performance of correlative studies that compare electrophysiologic responses with the expression and/or distribution of key signaling molecules in the underlying myocardium.
Potential role of the Autonomic Nervous System in the Creation of Substrate for
Atrial Fibrillation
Despite the heterogeneity of substrate that may contribute to clinical AF, clinical studies suggest that at least in some patients, the sympathetic and/or the parasympathetic nervous system play a role in the genesis of AF.15,16 Autonomic fluctuations before the onset of AF have been recognized in several studies. Although most studies are observational and do not show a clear association between cause and effect, more recent studies suggest that surgical or ablative destruction of neuronal/autonomic fat pads in and around the heart can indeed modify substrate for AF, thereby indicating a more direct role of for the autonomic nervous system in the genesis of AF.11,12
Earlier studies suggested that exercise-induced AF may be sympathetically driven (i.e. Sympathetic AF); in contrast, the parasympathetic nervous system may be contributing to the genesis of AF in young patients ages 30–50 years, who have no structural heart disease, with arrhythmic episodes occurring during rest or night time (i.e. Vagal AF).15,16 Vagal nerve stimulation appears to contribute to AF substrate by non-uniform shortening of atrial effective refractory periods, creating conditions conducive to reentry.17,18 Vagal nerve stimulation can also create conditions for the emergence of focal triggers in the atrium, as suggested by studies conducted by Sharifov et al.19 More recently, both limbs of the autonomic nervous system have been shown to play a role in AF in at least some patients. Amar et al showed that onset of AF was preceded by a primary increase in sympathetic drive followed by marked modulation toward vagal predominance20 Sharifov et al showed that although acetylcholine alone could induced AF in all the dogs that were studied, acetylcholine-induced AF was facilitated by isoproterenol, which decreased the concentration of acetylcholine required for AF induction and maintenance.21 The physiology studies conducted by Patterson et al22-24 further suggest that adrenergic influences may be playing an important modulatory role in creating adequate substrate for AF, helping provide a necessary ‘catalyst’ for the emergence of focal drivers in the presence of an increased vagal tone.
In view of the recently discovered role of the PVs and PLA in the genesis of AF, recent studies have also begun to explore the role of the autonomic nervous system in focal AF. Clinical and in vivo animal data also suggest that these PV foci appear to be at least partially modulated by the autonomic nervous system, with sympathetic stimulation (e.g. with isoproterenol) being frequently utilized to “bring out” these triggers in clinical studies. Clinical studies have demonstrated a change in sympathetic-parasympathetic balance-measured by heart rate variability-after ablation of PV triggers.25 Several investigators have also noted significant Bezold-Jarisch-like reflexes during radiofrequency ablation of the pulmonary vein tissues.26 More recently, Pappone et al have suggested that altering vagal input to the left atrium and the pulmonary veins-as measured by the elimination of vagal reflexes on pulmonary vein stimulation-may improve efficacy of ablation procedures for AF.27 Vagal responsiveness also appears to decrease following ablation in the left atrium.28
Recent data from patients undergoing AF ablation suggests that areas in the atrium demonstrating complex fractionated atrial electrograms (CFAE) may represent a suitable target site for ablation; ablation at these sites appears to increase the efficacy of pulmonary vein isolation procedures.29-31 One possible explanation for this improvement in ablation success is that atrial/pulmonary vein sites demonstrating CFAE anatomically overly fat pads containing ganglionated plexi (GP);32 ablation in the vicinity of these GPs would therefore be expected to result in autonomic denervation of the surrounding atrium. In fact, vagal reflexes have frequently been noted in regions demonstrating CFAE, further suggesting that these complex electrograms may indicate the presence of underlying GPs. Data from patients undergoing ablation for AF also indicates that the electrograms in the left atrium with CFAE become more organized following ablation.33
Recent studies suggest that the autonomic nervous system may also be playing a role in the genesis of AF in diseased hearts. Jayachandran et al demonstrated a heterogeneous increase in sympathetic innervation in the atria in dogs subjected to rapid atrial pacing for prolonged periods of time.34 There is also evidence of sympathetic hyperinnervation in patients with AF.35 More recently, Ogawa et al7 have shown increased sympathetic and vagal nerve discharge prior to the onset of atrial arrhythmias in dogs with pacing-induced congestive heart failure. Studies from our own laboratory indicate increased sympathetic as well as parasympathetic nerve growth in the atria of dogs with pacing-induced congestive heart failure.36
Peculiar Autonomic Innervation of the Pulmonary Veins and Posterior Left Atrium
Elegant studies by Armour and Randall several years ago demonstrated the presence of a intricate pattern of autonomic innervation in the heart.37-39 An improvement in our understanding of atrial arrthyhmias in recent years-especially the discovery of the pulmonary veins as being an important contributor to AF-has led to a renewed interest in understanding the detailed anatomy of the cardiovascular nervous system. Armour et al40 showed that autonomic nerves were concentrated in ‘ganglionated plexi’ around great vessels such as the PVs, with the atria being innervated by at least 5 major atrial fad pads, most of which were located on the posterior surface of the atria. More recently, Hou et al9,10 have suggested the presence of an intricate, interconnecting neural network in the left atrium that may contribute to substrate for focal AF. A recent human study described heterogeneity of nerve distribution in the region of the pulmonary veins and surrounding left atrium41 As both parasympathetic and sympathetic activation may have prominent effects on atrial conduction velocity and refractoriness, regional heterogeneities in nerve distribution could thereby be responsible for the heterogeneities in conduction velocity and refractoriness that are necessary for the initiation and perpetuation of AF. Thus, the distribution of autonomic nerves throughout the atrium could represent a substrate for AF, particularly in settings where there are no anatomic abnormalities.
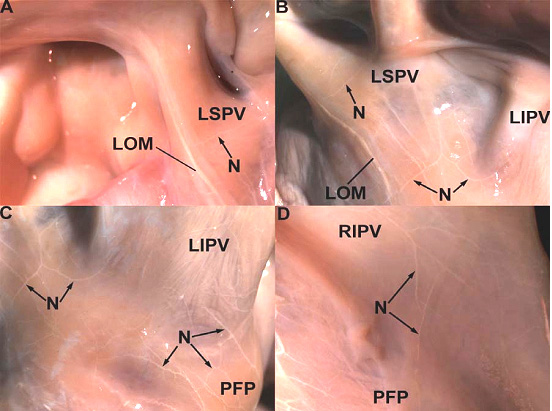
In light of these prior studies, we compared the distribution and physiology of sympathetic and parasympathetic nerves among the pulmonary veins, the posterior left atrium and left atrial appendage.6 We also attempted targeted parasympathetic blockade in the posterior left atrium with a muscarinic receptor blocker and studied in detail the resulting electrophysiologic response of the left atrium. The posterior left atrium was the most richly innervated with nerve bundles containing parasympathetic and sympathetic fibers (see figure 1); nerve bundles were located in fibrofatty tissue as well as in surrounding myocardium. Parasympathetic predominated over sympathetic fibers within bundles (see figure 1). M2 receptor distribution was also most pronounced in the posterior left atrium. Following tropicaimide application locally to the posterior left atrium, vagal-induced ERP shortening was significantly attenuated not just in the posterior left atrium but also remotely from the site of application in the pulmonary veins and the left atrial appendage. AF induciblity decreased significantly after application of tropocamide. In a related study, we discovered a particularly high concentration of parasympathetic fibers in the ligament of Marshall42 (see figure 2). The ligament of Marshall could be traced back to a major branch of the left cervical vagus nerve. Ablation of the ligament of Marshall led to an attenuation of vagal-induced ERP shortening in the left sides pulmonary veins and the rest of the posterior left atrium. The course of the ligament of Marshall along the posterior wall of the left atrium (see figure 2) further highlights the potential importance of this region (i.e. the posterior left atrium) in the creation of autonomic substrate for AF.
Figure 1. Co-localization of sympathetic and parasympathetic nerves within a single nerve bundle in the PV. The figures to the left (A,C,E) show a single large nerve bundle stained only with hematoxylin, while the figures on the right (B,D,F) are double-stained with for sympathetic and parasympathetic nerve elements (blue-sympathetic; brown-parasympathetic). A and B (both 2.5x magnification) show that the nerve bundle (arrows) is located in the fibrofatty tissue on the epicardial aspect of the PV. C and D show that same bundle (arrows) at 10x magnification. E and F show the same bundle at 20x magnification. Sympathetic-parasympathetic co-localization is clearly demonstrated in F. The bundle is predominantly composed of parasympathetic nerve fibers (stained brown) with occasional sympathetic nerve fibers (stained blue; see arrows). Panel G shows a bar-graph demonstrating parasympathetic predominance within individual nerve bundles. The bar-graph shows that in each region, within individual nerve bundles that demonstrate sympathetic and parasympathetic co-localization, the proportion of parasympathetic fibers is much greater than the number of sympathetic fibers. Error bars represent the standard error (SE). S=sympathetic fibers, P=parasympathetic fibers. Modified with permission from Arora R, Ulphani J et al. Neural Substrate for Atrial Fibrillation: Implications for targeted Parasympathetic blockade in the Posterior Left Atrium Am J Physiol-Heart and Circ Physiol 2008; 294(1):H134-44
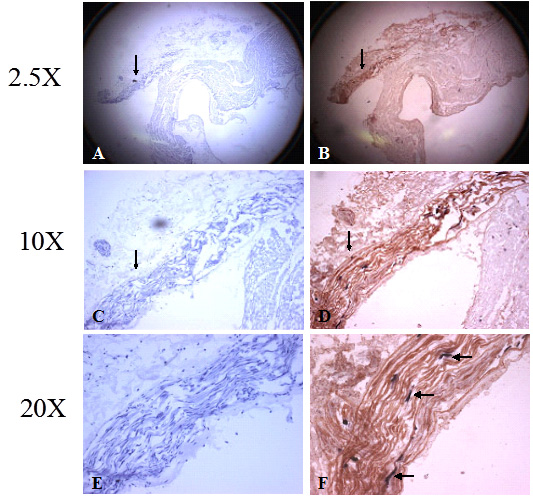
Figure 2. The posterior surface of the left atrium stained for acetylcholinesterase. A. The cholinergic fibers of the vagus nerve are observed to enter the LOM along the left superior pulmonary vein (LSPV). A small nerve branch (N) originating in the LOM is seen to innervate the LSPV. B. White cholinergic fibers originating in the LOM are seen to innervate the LSPV and the left inferior pulmonary vein (LIPV). C. Cholinergic fibers from the posterior left atrial fat pad (PFP) can be seen to innervate the LIPV. D. A small number of cholinergic nerve fibers from the posterior left atrial fat pad can be seen to innervate the right inferior pulmonary vein. Modified with permission from Ulphani J et al. The Ligament of Marshall as a Parasympathetic Conduit. Am J Physiol-Heart and Circ Physiol 2007;293(3):H1629-35
Our immunohistochemical studies are in agreement with clinical studies where Tan et al43 demonstrated co-localization of sympathetic and parasympathetic nerve fibers in nerve trunks in the human left atrium. Although the authors in that study showed a relative predominance of sympathetic fibers (as compared to a preponderance of parasympathetic fibers in our canine study), electrophysiologic studies were not performed to assess the physiologic significance of this nerve distribution pattern. In addition, even though this study contributes significantly to our understanding of the distribution of parasympathetic and sympathetic nerves at the venoatrial junction, a relative comparison of nerve distribution was not performed between different regions of the left atrium; more specifically, the autonomic innervation of the posterior left atrium and pulmonary veins was not compared with the rest of the left atrium. Regardless of the difference in parasympathetic/sympathetic ratio between the two studies (which may be accounted for by inter-species differences in neural innervation between humans and dogs or by differences in sampling between the two studies) the parasympathetic nervous system appears to be a major player in both clinical as well as canine AF.
Contribution of Autonomic Nerves to Atrial Electrophysiology
Despite these initial studies, the complete autonomic profile of the pulmonary veins and the precise role of the autonomic nervous system in the genesis of focal AF are just beginning to be systematically investigated in clinical and animal models. Recent data from our laboratory suggests a differential electrophysiologic response of the pulmonary veins/posterior left atrium from that in the rest of the left atrium in response to autonomic maneuvers.5 In that physiologic study, we demonstrated a greater decrease in refractory periods in the pulmonary veins and posterior left atrium as compared to the rest of the left atrium in response to vagal stimulation. The heterogeneity of vagal responses in the left atrium in this study was found to correlate with differences in the pattern of distribution of IKAch in the pulmonary veins, posterior left atrium and left atrial appendage. We also discovered that with vagal stimulation and/or beta-blockade, there was evidence of regional conduction delay in the pulmonary veins with a significant change in activation direction. Similar activation changes were not seen in the posterior left atrium and left atrial appendage.
Moreover, as mentioned earlier, we have observed that topical cholinergic blockade in the posterior left atrium results in a significant change in vagal responsiveness in the left atrium, confirming the importance of the parasympathetic innervation of the posterior left atrium in the genesis of vagal-induced AF. The diminution of vagal reflexes in the entire entire left atrium in response to topical cholinergic blockade in the posterior left atrium alone suggests that the majority of the parasympathetic fibers innervating the left atrium originate or at least pass through the posterior left atrium prior to innervating the rest of the left atrium. In fact, recent studies9,10 indicate the presence of interconnections between ganglionated plexi in the atria. It therefore appears likely that the remote effects of tropicamide noted in the rest of the left atrium are due to the functional disruption of an intricate and interactive neural network in the left atrium. This hypothesis is further supported by the following observations from our study 6: 1) cardiac ganglia containing neuronal cell bodies are present only in the posterior left atrium and pulmonary veins but were completely absent in the rest of the atrium, 2) the majority of nerve trunks in the left atrium are located in the posterior left atrium and 3) nerve trunks in the posterior left atrium are larger than in the rest of the left atrium. The remote effects of tropicamide on vagal responsiveness in the pulmonary veins and left atrial appendage can therefore be best explained by a positive feedback mechanism that involves muscarinic receptors present on cholinergic neurons;44,45 these receptors are known to participate in negative as well as positive feedback mechanisms that modulate acetylcholine release from neuronal cells.44,45 We postulate that tropicamide, by inhibiting muscarinic receptors present on ganglion cells and nerve trunks in the posterior left atrium, decreases acetylcholine release more distally from neuronal synapses in the pulmonary veins and left atrial appendage.
Selective Parasympathetic Denervation of the Posterior Left Atrium—a new Therapeutic Target for AF?
An ideal therapeutic strategy would be more precise in targeting the nerves involved in the genesis of AF without causing any significant damage to surrounding tissue. Development of such a targeted approach to focal AF hinges on a better understanding of the complexities of neural innervation of the atria.
Elimination of vagal reflexes during ablation lesions has been shown in some studies to be correlated with improved PV ablation outcomes.27,46 However, other attempts at parasympathetic ablation for AF have been less successful.14,47 Current endocardial ablation strategies that target the parasympathetic nervous system of the left atrium are largely empiric, being guided by non-specific electrophysiologic responses such as sinus bradycardia and AV block.27,46 More ‘anatomic’ approaches that surgically target atrial fat pads have also met with somewhat mixed success.47,48 Even though AF inducibility may decrease in the short term following epicardial fat pad ablation, long-term surpression of AF may not be seen.13 An added disadvantage of an anatomic approach is that it inevitably causes transmural atrial tissue damage.13 In addition, the left atrium is innervated by several fat pads, all of which may not be responsible for the innervation of the posterior left atrium and pulmonary veins. Moreover, sympathetic and parasympathetic nerves in the left atrium are nearly always co-localized,6,43 making selective vagal denervation impossible without resulting in collateral damage to neighbouring sympathetic fibres.
The studies discussed above6,42 indicate that the posterior left atrium is the most richly innervated region of the left atrium, with parasympathetic fibers comprising a majority of the nerves supplying this area. Importantly, even though a majority of nerve bundles are located within the fat/fibrofatty tissue itself, up to a third of nerve bundles in the posterior left atrium can be located away from the fat in adjoining/underlying myocardium.6 This finding suggests that ablation strategies directed at atrial fat pads would not be expected to result in complete denervation of the posterior left atrium. Since vagal nerve fibers and M2 receptors are preferentially localized in the posterior left atrium, our experiments demonstrated that topical, pharmacological disruption of cholinergic function in the posterior left atrium alters vagal responsiveness in the entire left atrium. These results suggest that it is indeed feasible to attain selective cholinergic blockade in this region with a non-ablative, pharmacological approach, with a resulting change in vagal-induced AF substrate.