Introduction: Pulmonary vein (PV) isolation is the mainstay of catheter treatment of paroxysmal atrial fibrillation (AF). The CoolLoop® cryoablation catheter (AFreeze® GmbH; Innsbruck, Austria) was developed to create wide and complete circular lesions around the PVs. In this study we evaluated feasibility and safety of this novel ablation system in humans.
Methods: 10 patients (6M/4F; 57.6±7.6y) with paroxysmal AF were included in 2 referral centers. The CoolLoop® catheter was positioned at each PV antrum using a steerable transseptal sheath. Subsequently, 2-6 double-freezes over 5min were performed at each vein and PV-isolation was assessed thereafter using a circular mapping catheter. During cryoablation of the right PVs, pacing was used to monitor phrenic nerve function.
Results: The CoolLoop® catheter could be successfully positioned at each PV. A mean of 5.6±1.8 cryoablations were performed in the LSPV, 5.2±1.6 in the LIPV, 6.3±2.5 in the RSPV and 5.4±1.6 in the RIPV, respectively. Mean procedure time was 251±60min and mean fluoroscopy time was 44.0±13.2min. 6 / 10 LSPV, 6 / 10 LIPV, 5 / 10 RSPV and 6 / 10 RIPV could be isolated exclusively using the novel cryoablation system. One patient developed groin hematoma and a brief episode of ST-elevation due to air embolism was observed in another subject. No other clinical complications occurred during 3 months of follow up.
Conclusions: PV-isolation for paroxysmal atrial fibrillation using the CoolLoop® catheter is feasible and appears safe. Clinical long term efficacy still needs to be evaluated and will be compared with established catheters used for AF ablation.
Atrial fibrillation (AF) is the most common sustained arrhythmia and is a major cause of impaired quality of life, hospitalizations, stroke and increased mortality.1,2 In contrast preservation of sinus rhythm by either antiarrhythmic medication or catheter ablation significantly improves quality of life of AF patients.3,4
Pulmonary vein isolation (PVI) - demonstrated as elimination of pulmonary vein (PV) potentials recorded from a circular mapping catheter during sinus rhythm and during PV pacing - has been proposed as the mainstay of catheter ablation in paroxysmal AF.5 Large lesions around the vein antrum rather than ablations within the PVs itself are recommended to avoid PV stenosis and to include more sites of AF initiation and maintenance.5 Indeed, one prospective randomized study showed that isolation of a large circumferential area around both ipsilateral PVs with verification of conduction block is a more effective treatment of AF than ostial segmental isolation.6
Cryoablation catheters work by delivering liquid nitrous oxide under pressure to a tip or balloon, where it turns into gaseous state, resulting in cooling of surrounding tissue. Local tissue injury results from freezing with creation of intracellular ice crystals that disrupt cell membranes and interrupt both intracellular metabolism and electrical activity. In addition, alteration of microvascular blood flow similarly produces cell death.7 PVI with cryoenergy has been shown to be effective in a large prospective multicenter study.8 However, complete PVI was found to be a critical procedural endpoint, since recurrence rates of AF are higher, if PVI was not achieved at the end of the procedure.9
Recently a novel cryoablation system has been developed to create large antral lesions around the PVs using a “single shot” device. The system consists of a cryoablation console, a steerable sheath and a circular cryoablation catheter including a 30mm diameter loop shaped cryo-applicator (CoolLoop®; AFreeze® GmbH; Innsbruck, Austria). In this “first-in-man” study the CoolLoop® ablation system was tested for the first time in patients with paroxysmal AF. In this clinical trial we sought to determine procedural feasibility and short term safety of PVI using the novel CoolLoop® cryocatheter.
Patients were screened and recruited at 2 referral centers for AF ablations between September and December 2012. Only patients with two or more symptomatic, paroxysmal AF episodes despite treatment with at least one antiarrhythmic drug were included. At least one episode of AF within the last 3 months had to be documented by ECG. Exclusion criteria were: LA diameter <50 mm (assessed by transthoracic echocardiography), cardioversion performed later than 7 days after onset of AF, significant structural heart disease (moderate or severe valvular heart disease, congenital heart disease, previous myocardial infarction or coronary artery bypass graft surgery, left ventricular ejection fraction <45%), chronic obstructive pulmonary disease or severe respiratory insufficiency, known bleeding diathesis or intolerance to heparin or oral anticoagulants, previous AF ablation, left atrial thrombus, severe other non-cardiac diseases, hyperthyreosis and pregnant or lactating females.
The study was approved by the Institutional Ethical Review Board of the Medical University of Innsbruck (AN-4701-312/4.7) and Linz General Hospital (B-28-12) as well as by the Federal Agency for Safety in Health Care (INS-621000-0338) and all participants gave their written informed consent prior to inclusion into the study.
Upon admission to the hospital laboratory blood testing was performed and an ECG was recorded. Left atrial thrombi were ruled out using transesophageal echocardiography and left atrial morphology was visualized by computed tomography and subsequent 3D reconstruction on the day before the procedure.
The CoolLoop® cryoablation catheter consists of a grip, a shaft and a loop shaped cryoapplicator (diameter 30mm) with an eccentric tip (Fig. 1). The catheter was positioned at the PV antrum using standard over the wire technique. Before introduction the catheter was flushed with heparinized saline for at least 60sec (flow: flushing lumen 6ml/min; positioning lumen 85ml/min), thereafter flow rate in both lumina was kept at 2ml/min during the entire procedure.
Figure 1. The CoolLoop® cryoablation catheter: The tip of the catheter is shown including the loop-shaped cryo-applicator (diameter 30mm) and the inner lumen with the introduced guide wire. Please note the eccentric location of the catheter tip.

Left atrial catheter ablation was performed under conscious sedation using a combination of propofol and fentanyl. Continuous recording of ECG, blood pressure using an arterial line, and peripheral oxygen saturation were mandatory.
After a single transseptal puncture the AFreeze® steerable transseptal sheath was advanced into the left atrium under fluoroscopic guidance. Before transseptal puncture 5000 IE of heparin were administered, followed by another 5000 IE as soon as access to the left atrium had been achieved. Activated clotting time was assessed every 30 min. and was maintained between 250 and 350 s using heparin boli. The tip of the CoolLoop® catheter was advanced over a guide wire towards the proximal part of the PV, where the loop-shape of the catheter was formed. By advancing both the steerable sheath and the ablation catheter to each PV sufficient contact with the left atrial wall was achieved. Two consecutive cryoablations for five minutes were performed at each PV and at least two distinct catheter positions had to be delivered by turning the CoolLoop® catheter clockwise for 90-120 degrees after the first ablation.
Before and up to 15 min after the last cryoablation a decapolar circular mapping catheter was placed at each PV ostium via the steerable sheath to assess PV isolation. To measure the degree of PV isolation the percentage of PV signal reduction on the decapolar catheter (signals on post-ablation bipoles / pre-ablation bipoles) was calculated. Furthermore pacing over each bipole of the circular mapping catheter was performed to verify exit block. If electrical isolation could not be demonstrated, additional cryo-applications using the CoolLoop® or another cryo-catheter were performed at the operator’s discretion. At the end of the procedure all catheters and sheaths were removed, sedation was stopped and pericardial effusion was excluded by echocardiography.
Following the procedure patients were monitored by continuous ECG and non-invasive blood pressure measurement for 12 hours. On the day after the catheter ablation phrenic nerve palsy was ruled out by X-ray and spirometry, and oral anticoagulation was started after pericardial effusion had been excluded. Antiarrhythmic drugs, except for ß-blockers, were discontinued following catheter ablation. In case of AF-recurrences antiarrhythmic drugs were re-started upon the operator`s discretion. Follow ups at discharge, as well as 2, 4, 8 and 12 weeks after ablation included medical and clinical history, blood tests and 12-lead ECGs, respectively. Additionally 7-day Holter monitoring and echocardiography were performed at 3-months of follow-up. Oral treatment with Pantoprazol 40 mg per day was continued until 8 weeks after the procedure and oral anticoagulation was mandatory for at least 3 months after ablation.
The primary endpoint of the study was safety and tolerability of cryoablation using the CoolLoop® Cryoablation System. Specifically the following parameters were analyzed: deformation of the catheter loop resulting in entrapment of the catheter in the heart, phrenic nerve palsy, death, pericardial effusion, cardiac tamponade, valve damage requiring surgery and hemorrhage requiring transfusion, atrio-esophageal fistula, sepsis or endocarditis, stroke or transient ischemic attack and PV stenosis until the final visit 3 months after cryoablation.
Secondary endpoints were feasibility (defined by the ability to position the catheter in the proximal part of each PV, forming the cryo-applicator of the catheter to a loop by operating the catheter handle, positioning the loop at the wall of the PV antrum and delivering cryoenergy), immediate efficacy of cryoablation (defined as the percentage of electrodes within the PV without measureable signals), clinical efficacy of catheter ablation (defined by freedom of AF during follow up), procedure and fluoroscopy time (defined as time from introduction until removal of the catheter from the left atrium), cumulative cryoablation time, as well as minor procedural complications (pneumothorax, hematothorax, femoral pseudoaneurysm, arteriovenous fistulae, and hemorrhage not requiring transfusion).
No hypothesis testing and no sample size estimation were performed. Statistical analysis for safety and efficacy was performed according to the intention-to-treat principle. All parameters were summarized using descriptive statistics, i.e. number (n) and percentage of patients of the total cohort (%) for categorical variables and mean values or mean differences ± standard deviations (SD) for continuous variables.
All statistical analyses were performed using SPSS 16.0 statistical software (Chicago, Illinois, USA) and were supported by the Department of Medical Statistics, Informatics and Health Economics at the Medical University of Innsbruck.
10 patients from two Austrian referral centers (6 male/4 female) with exclusively paroxysmal AF were included into the study.
An overview of demographics is given in table 1. The cohort had a long lasting history of atrial fibrillation and a high number (5.4±4.3 per week) of episodes lasting up to 30 hours in average. 5/10 patients had been hospitalized within 12 months prior to ablation, 9/10 had been treated with betablockers and all of them with 1-2 additional antiarrhythmic drugs. Patients’ LA was moderately dilated (42.2±4.0mm), 2 patients had clinically non-significant aortic or mitral regurgitation (table 1). In 2 patients ablation of the cavo-tricuspid isthmus and in one ablation of an accessory pathway had been performed several years prior to the study, one participant suffered from Mb. Bechterew and psoriatric arthritis, one had prior stroke without any neurological residuals, four had hypertension and another one bronchial asthma.
Table 1. Demographics of patients (n=10)
| Variable |
| gender (male/female) |
6/4 |
| age (years) |
57.6±7.6 |
| duration of AF prior to PVI (months) |
82.8±84.5 |
| number of AF episodes / month prior to PVI (n) |
21.7±17.3 |
| maximum duration of AF episodes (hours) |
32.0±18.8 |
| number of hospitalizations due to AF during previous 12 months (n) |
0.7±0.7 |
| left atrial size in echocardiography (mm) |
42.2±4.0 |
| LVEF on echocardiography (%) |
62.0±5.0 |
| BMI (kg/m2) |
25.4±3.4 |
| Hypertension (n) |
4 |
| patients treated with beta-blockers prior to PVI (n) |
9 |
| patients treated with class IC antiarrhythmic drugs prior to PVI (n) |
3 |
| patients treated with class III antiarrhythmic drugs prior to PVI (n) |
9 |
| patients with electrical cardioversion prior to PVI (n) |
4 |
AF – atrial fibrillation; PVI – pulmonary vein isolation; LVEF – left ventricular ejection fraction; BMI – body mass index; values are given in mean ± SD
Three-dimensional reconstruction of CT scans prior to each ablation did not show any PV anomaly, specifically all patients had 4 PVs and no common ostia or additional PVs could be detected.
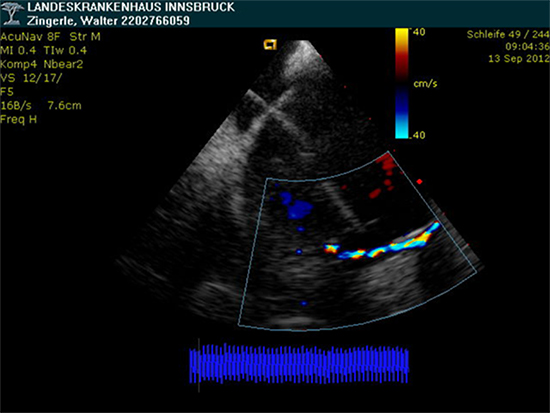
The CoolLoop® catheter could be successfully positioned at each PV ostium in all 10 patients. Furthermore circular contact to atrial tissue could be visualized by intracardiac echocardiography in all subjects (Fig. 2) and cryothermal energy could be delivered at each PV antrum without complications. Moreover the CoolLoop® catheter could be directed to various positions and finally retracted from each atrium through the steerable sheath. An episode of air embolism developed in patient # 2 shortly after introduction of the catheter into the left atrium, leading to asymptomatic transient ST-segment elevations for 3 min on surface ECGs, but without occurrence of AV-block or requirement of vasopressors. Physical examination, ECG and echocardiography after the procedure were unremarkable, thus the patient was not subjected to further cerebral or cardiac imaging. After this event the study protocol was slightly changed and both lumina of the CoolLoop® catheter were flushed with saline even more vigorously prior to insertion of the catheter into the steerable sheath (see methods). No further device related adverse events were observed after these changes of the protocol. Specifically, no peri-interventional stroke, no phrenic nerve palsy and no cardiac tamponade or pericardial effusion occurred during the study. One patient developed groin hematoma during the ablation procedure. The intervention was prematurely stopped (after 4 ablations at each PV using the CoolLoop®) and PVI was not finalized. Surgical revision of the hematoma was performed on the day after ablation.
Figure 2. Visualisation of the CoolLoop® cryoablation catheter during a freeze cycle by 2-dimensional intracardiac echocardiography (ICE): the loop of the catheter was positioned at the antrum of the LIPV of patient #2 und visualized by ICE; during freezing the catheter could be visualized by color Doppler echocardiography (yellow arrow); note the peri-ostial location of the CoolLoop® catheter tip that even partially overlaps the ostium of the LSPV; LSPV – left superior pulmonary vein; LIPV - left inferior pulmonary vein (green lines indicating PV edges)
Inevitably long procedure durations and large amounts of fluoroscopy were necessary in this first-in-man study. Mean procedure duration was 251±60min, mean fluoroscopy time 44.0±13.2min and total area dose 13267±10163cGy cm2. However, procedure duration and amount of fluoroscopy could be significantly reduced throughout the study (table 2).
Table 2. Procedural parameters of the study patients
| Patient |
Procedure time (min) |
Fluoroscopy time (min) |
Fluoroscopy dose (cGycm²) |
| 1 |
231 |
53.43 |
14969 |
| 2 |
300 |
46.43 |
18127 |
| 3 |
219 |
53.00 |
16789 |
| 4 |
277 |
44.15 |
20829 |
| 5 |
249 |
40.15 |
5162 |
| 6 |
126 |
17.05 |
5742 |
| 7 |
320 |
59.00 |
5832 |
| 8 |
320 |
59.00 |
4165 |
| 9 |
204 |
34.00 |
1217 |
| 10 |
265 |
34.00 |
2243 |
| mean ± SD |
251 ± 60 |
44.0 ± 13.2 |
9508 ± 7318 |
patients 1 - 6 were treated in center #1, 7 - 10 in center # 2
In total 225 ablations with the CoolLoop® were delivered in the 10 study patients. 56 cryoablations were applied at the antrum of the LSPV, 52 at the LIPV, 63 at the RSPV and 54 at the RIPV, respectively. Thus, a mean of 5.6±0.8 cryoablations for 5min each were delivered per PV. 23/40 PVs (57.5%) could be isolated using the CoolLoop® catheter exclusively, whereas 13/40 PVs (32.5%) needed additional touchup-lesions with cryotip (Freezor Max®, Medtronic Cryocath, USA; 9/40) or cryoballoon catheters (Arctic Front Advance®, Medtronic Cryocath, USA; 4/40). The remaining 4 PVs could not be isolated at the end of the procedure for reasons described above. No differences were found in distributions of PVs that could not be isolated (table 3). No early spontaneous PV reconnection within 15min after ablation was observed. Testing for PV reconnection with adenosine (2mg) was performed at the end of the procedure in 5/10 patients. However, no reconnection of PV conduction was recorded in any patient after adenosine testing.(table 2).
Table 3. Pulmonary vein isolation exclusively using the CoolLoop® cryoablation catheter
| Patient |
LSPV |
LIPV |
RSPV |
RIPV |
overall |
| 1 |
1 |
1 |
0* |
1 |
3 |
| 2 |
1 |
1 |
1 |
0* |
3 |
| 3 |
0** |
0** |
0** |
0** |
0 |
| 4 |
1 |
1 |
0* |
0* |
2 |
| 5 |
0* |
0* |
1 |
1 |
2 |
| 6 |
1 |
1 |
0* |
1 |
3 |
| 7 |
1 |
1 |
1 |
1 |
4 |
| 8 |
0* |
0* |
1 |
1 |
2 |
| 9 |
1 |
1 |
1 |
1 |
4 |
| 10 |
0 |
0 |
0 |
0 |
0 |
| overall |
6/10 |
6/10 |
5/10 |
6/10 |
23/40 |
ablation success in each pulmonary vein of each study patient is shown; 1 – PV isolation; 0 – failure of PV isolation; PV isolation was completed by cryo-tip touchup lesions(*) or cryoballoon applications (**) after cryoablation with the CoolLoop® catheter; LSPV – left superior pulmonary vein; LIPV – left inferior pulmonary vein; RSPV – right superior pulmonary vein; RIPV – right inferior pulmonary vein;
In 17 non-isolated PVs signals on the decapolar catheter could be reduced with the CoolLoop® alone by 80% (unaffected PV signal in 1 bipole) in 5 PVs, and by 60% (unaffected PV signal in 2 bipoles) in 8 PVs, respectively. Accordingly PV isolation could be achieved in the majority of patients by additional focal ostial ablations using other cryo-catheters. Importantly all PVs of patient # 5 (of 6) in one center and patient # 3 (of 4) in the other center could be isolated using the CoolLoop® catheter exclusively without any additional touchup ablations, suggesting a fast learning curve of the operators with the new device.
No clinical adverse events were observed in follow ups 2, 4, 8 and 12 weeks after ablation. Specifically no nerve palsy, no cardiac tamponade, no clinical sign of valve damage or PV stenosis, no atrial-esophageal fistula, no sepsis or endocarditis, no stroke or transient ischemic attack occurred until the final visit 3 months after cryoablation. Additionally valve damage was excluded by echocardiography and PV stenosis was excluded in CT scans available from 5 patients. Patient # 10 required transfusion and surgical revision due to groin hematoma, but this complication was not found to be device related by the dedicated safety committee of the study and the local institutional review board.
1 to 6 symptomatic episodes of AF were reported in each of the ablated patients, and 1 to 5 symptomatic or asymptomatic AF episodes (>30sec duration) were documented in 12-lead ECGs in each study participant over a follow up period of 3 months. 2 patients had to be hospitalized and underwent electrical cardioversion. The study protocol was not intended to search for asymptomatic episodes of AF, except during 7-day Holter ECG. AF was documented at the end of the study in 7-day Holter ECGs of 6/10 patients (AF-burden 1-18%; cumulative AF 1hr 47min to 30hrs). Of these latter 6 patients, 2 were in stable sinus rhythm at 3 months of follow up under antiarrhythmic medication that had been unsuccessful prior to ablation (1 propafenone, 1 amiodarone), and the other 4 were scheduled for re-ablation.
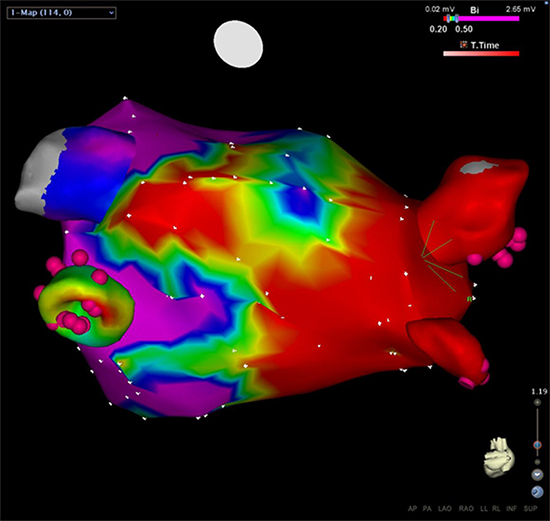
Repeated ablations were performed due to AF recurrence in 4/10 patients 1-3 months after the last follow-up of the study (3-6 months after cryoablation) and in another patient 9 months later. Electro-anatomic mapping in 3/4 patients demonstrated large scars around isolated PVs (Fig. 3). However, segmental reconnections of 2-4 PVs per patient were noticed in each patient, suggesting PV reconnection as the mechanism of AF recurrence. No significant differences were found in locations of conduction gaps during redo procedures. This fact, however, might be due to the limited number of patients included in the current study. These results provide evidence that cryoablation with the CoolLoop® catheter leads to wide antral circumferential lesions, but PV reconnections were observed quite commonly.
Figure 3. Illustration of the extent of lesions around the right PVs induced by the CoolLoop® cryoablation catheter: Patient #10 underwent re-ablation using electro-anatomic mapping (CARTO® 3 system; Biosense Webster; USA) 12 months after the index-procedure due to recurrence of atrial fibrillation. A precise bipolar voltage map was performed to visualize the area around the PVs and to specifically measure the extent of lesions after CoolLoop® ablation. A posterior-anterior projection of the map is shown. Small isolation defects were found in all PVs 12 months after cryoablation, but a large area of scar (defined as bipolar voltage ≤0.20mV; red area) was detected around the right PVs. Pink bullets indicate bipoles of the decapolar circular catheter.
The CoolLoop® first trial reports our initial experience with left atrial catheter ablation using a novel circular cryoablation catheter in humans. Our results show that positioning, freezing and retraction of the CoolLoop® catheter is feasible and PVI can be achieved in the majority of subjects. Even more importantly the procedure with the novel circular cryocatheter system appears to be safe in this small cohort of patients with paroxysmal AF.
Feasibility Of Left Atrial Ablation Using CoolLoop®
The novel cryoablation catheter could be successfully introduced into the LA and positioned at the antrum of each PV of all patients. Subsequently 22.5±3.8 cryoablations were performed per patient. Finally the CoolLoop® catheter could be retracted from each LA through the steerable sheath without technical problems.
As expected due to the “first-in-man” design of the trial, extended procedural and fluoroscopy durations were needed in this study. Especially in the first 2-4 procedures at each center, procedure duration and fluoroscopy time were longer than those observed in a recent AF ablation registry in Germany.10 However, a steep learning curve was found, since fluoroscopy time could be reduced from 53 to 17min from the 1st to the 6th patient in center #1 and from 59 to 34min from the 1st to the 4th patient in center #2. Likewise total freezing time could be reduced by 12% and catheter handling time by 18%, when patients 1-5 were compared with patients 6-10 of the study (data not shown).
23/40 (57.5%) PVs could be isolated by the investigational device, which is comparable to the first PVI results of the Medtronic/Cryocath Arctic Front cryoballoon.11 The majority (13/17; 76%) of PVs that were not successfully treated by the CoolLoop® catheter could be subsequently isolated using 2-3 focal cryoablations. Finally, PVI in 2 of the last 4 patients of the study could be achieved using the CoolLoop® catheter only, again suggesting a steep learning curve of the operators. Thus, results of PVI with the novel CoolLoop® device were found to be adequate in the first 10 treated patients and promising for the future as experience with the new device will grow and improvements of catheter design will follow.
AF recurrence rates in this trial need to be interpreted with caution. Existing evidence shows that early recurrences of AF are common after catheter ablation. However, only 50% of patients with episodes within 3 months after ablation also develop recurrent AF later on.12 Therefore all occurrences of AF within 3 months after ablation in this study cannot be interpreted as PV reconnection and ablation failure. It still has to be mentioned that recurrences of AF were documented in 6/10 of patients after LA ablation with the new device, suggesting an increased rate of recurrences due to PV reconnections compared with prior larger multicenter studies using cryo-9 or RF-energy for PVI.13 However, this study was not designed to evaluate long-term recurrence rates of AF after LA ablation. A larger multicenter trial with a longer follow-up is currently ongoing to assess long-term performance of an improved version of the device.
Safety Of Left Atrial CoolLoop® Ablation
Safety of the CoolLoop® ablation system, although derived from a small cohort, appears to be reassuring. We did not find evidence of any clinical neurologic complication or any other thromboembolic event and pericardial effusion, valve damage or PV stenosis could be ruled out clinically and by echocardiography. Furthermore narrowing of the PV diameter or frank PV stenosis could be excluded in 5 patients, who underwent repeat ablation after closure of the clinical study (data not shown). Finally no phrenic nerve palsy was observed in the current study, whereas 13.5% of patients suffered from this complication in a larger cohort included into a prospective randomized trial analyzing the Medtronic/Cryocath cryoballoon.9 Although incidence of this most frequent complication of cryoablations in the left atrium varies10 and mainly depends on the size of the right pulmonary veins, the results of this small trial with the CoolLoop® catheter are promising.
Size And Durability Of Lesions Created By The CoolLoop® Catheter
The CoolLoop® catheter tested in the current study creates large antral lesions around the PVs (Fig. 3), whereas lesions created by the the Medtronic/Cryocath cryoballoon do not include antral portions of the LA.14 Given the 30mm diameter of the cryo-applicator and the eccentric location of the catheter tip of the new device this behavior of the new catheter has been aimed for and was also visualized by intracardiac echocardiography (Fig. 2) as well as electro-anatomic voltage mapping during a repeat ablation (Fig. 3). If myocardial lesions created using the novel device are found to be complete and durable in future studies, it would be tempting to hypothesize that paroxysmal as well as persistent AF can be treated using the CoolLoop® catheter ablation system.