Influence Of Inter Electrode Atrial Lead Distance On Acapconfirm™ Viability
Lapuerta Irigoyen J.A, Valverde Andre I, Vigil Escalera P, Gonzalez Vasserot M
Cabuenes Hospital, Gijon, Spain.
The AcapConfirm™ feature available with the Zephyr pacemaker family (St. Jude Medical) is designed for monitoring patient’s atrial capture threshold periodically, and automatically adjusting the atrial pulse amplitude. Previous studies showed a relative low proportion of patients at three months follow-up with recommended automatic atrial capture after the AcapConfirm™ viability test. The purpose of the present study is to evaluate the effect of inter electrode distance on the viability of the AcapConfirm™ algorithm.
132 patients (66 woman and 86 men; 71, 08 ± 8, 04 years old) were enrolled into this prospective evaluation. Sixty six bipolar leads (models 1882 (54p) and LPA1200M (12p)) with an inter electrode distance of 10 mm (Group A) were compared with sixty six bipolar leads (model 1999) with an inter electrode distance of 1,1mm (Group B). Set-up test AcapConfirm viability and manual step-down atrial threshold test as well as automatic threshold testing by AcapConfirm™ were performed at 3 months after implantation. A positive viability of the AcapConfirm™ algorithm was much lower in Group B (37, 9%; 95% confidence interval, 10, 3% – 65, 4%) versus thirty two patients (48, 5%; 95% confidence interval, 20, 9% - 76%) in Group A. However, the difference was not statistically significant (χ2=1, 51; p=0, 33). The most frequent reason to reject the AcapConfirm activation was a too small evoked response to polarization ratio (N9).At 3 months, threshold results from the AcapConfirm™ positive test were: 0, 53 ± 0, 13 V in Group B versus 0, 67 ± 0, 18 V in Group A (p< 0, 01). The differences between automatic and manual measurements were ≤0.25V in all patients.
We observed that a short inter electrode distance (1,1mm) is more likely correlated with a lower frequency of AcapConfirm™ viability and threshold that a standard inter electrode distance (10mm). A small evoked response to polarization ratio was the most common cause of a negative test of AcapConfirm™ viability.
Key Words : Acapconfirm, Automatic Threshold, Interelectrode Distance.
Correspondence to: Lapuerta Irigoyen, Cabuenes Hospital, Gijon, 33203 Spain.
The AcapConfirm™ feature available with the Zephyr pacemaker family (St. Jude Medical) is designed for monitoring patient’s atrial capture threshold periodically, and automatically adjusting the atrial pulse amplitude. ACapConfirm™ use pacing depolarization inte¬gral to calculate atrial-evoked response.1 ¬ Previous studies showed a relative low proportion of patients at three months follow-up with recommended automatic atrial capture after the AcapConfirm™ viability test.2
The purpose of the present study is to evaluate the effect of inter electrode distance on the clinical viability of the AcapConfirm™ algorithm. Viability of atrial threshold monitoring algorithm is defined as the percentage of patients who had atrial threshold monitoring enabled.
This observational, prospective data collection study included 142 consecutive patients with right atrial leads (bipolar) and right ventricular leads (bipolar) admitted for dual chamber pacemaker implantation. The intervention was performed by a single operator with experience in atrial and ventricular lead placement, under local anaesthesia and conscious sedation using a combination of intravenous midazolam and fentanyl. All patients received prophylactic intravenous antibiotics just before the procedure. Both leads were inserted via the left or right subclavian venous approach. The atrial and ventricular lead position choice was left to the discretion of the operator. After the device was implanted and before the patient was discharged from the hospital, the pacemaker was interrogated and the patient underwent chest radiography and standard 12-lead electrocardiography. Set-up test AcapConfirm™ viability and manual step-down (2,5 to 0,25V @ 0,4ms) atrial threshold test as well as automatic threshold testing by AcapConfirm™ were performed at implant, and 3 months after implantation. Data from participants who successfully completed both an automatic and manual capture thresholds test during follow-up at three months, were compared.
Continuous variables were expressed as mean ± standard deviation range, while categorical data were expressed as frequency and percentage. To compare the proportion of AcapConfirm™ between the two groups, we performed ᵡ2 test for categorical variables. Continuous variables were compared using t-test for normally distributed data or Mann Whitney U test for nonnormally distributed data as deemed appropriate.
Between July 2011 and August 2014, 142 patients were enrolled. Lead implantation was successful in all patients. The reasons for failure to reach the three months follow-up were atrial perforation (1p), atrial fibrillation (1p) death (3p), and loss (5p).Consequently, 132 patients (66 women and 86 men; 71, 08 ± 8, 04 years old) completed the 3-month follow-up. Regarding to interelectrode bipolar leads distance, patients were divided in 2 groups. Group A (10mm) included 54 patients with models 1882 and 12 patients with model LPA 1200M. Group B (1,1mm) included 66 patients with model 1999. Set-up test AcapConfirm viability and manual step-down atrial threshold test as well as automatic threshold testing by AcapConfirm™ (pulse width setting of 0.4 ms) were performed at 3 months after implantation.
Figure 1. Automatic Atrial Threshold ACapConfirm™
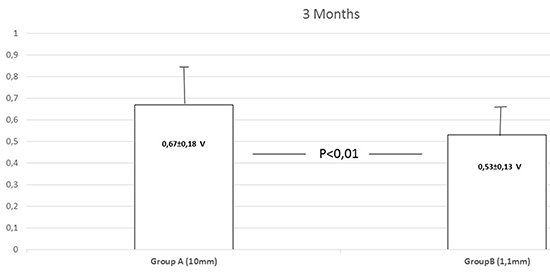
A positive viability of the AcapConfirm™ algorithm was much lower in Group B (37, 9%; 95% confidence interval, 10, 3% – 65, 4%) versus thirty two patients (48, 5%; 95% confidence interval, 20, 9% - 76%) in Group A. However, the difference was not statistically significant (χ2=1, 51; p=0, 33).
The most frequent reason to reject the AcapConfirm activation was a too small evoked response to polarization ratio (100% in Group A vs 87, 8% in Group B). (Table 1). At 3 months, threshold results from the AcapConfirm™ positive test were: 0, 53 ± 0, 13 V in Group B versus 0, 67 ± 0, 18 V in Group A (p< 0, 01). (Fig. 1) The differences between automatic and manual measurements were ≤0.25V in all patients.
Table 1. ACapConfirm not recomended(codes)
|
Group A (1,1 mm distance) |
Group B (10 mm distance) |
| Gain adjustment failed-satured at min gain |
X |
X |
| Gain adjustment failed-ER too small at max gain |
X |
X |
| Capture threshold too high |
X |
X |
| Capture threshold not found |
3p |
X |
| ER too low |
1p |
X |
| ER to polarization too small |
36p |
44p |
| Variability Safety Margin |
1p |
X |
Beat-to-beat verification of atrial pacing is problematic, owing to the small amplitude of atrial-evoked response (AER), which would make an algorithm prone to underdetecting effective stimulation The ACapConfirm™, an atrial threshold monitoring algorithm (Zephyr™ pacemaker family), uses pacing depolarization integral to calculate atrial-evoked response in order to decrease the influence of artifact during measurement1. This optimization of pacing output may increase patient safety and therapy efficacy by ensuring continuous therapy, reduces follow-up burden, allows ambulatory and remote threshold measurement, and may increase battery longevity.3,4,5
Unlike ventricular Autocapture™, ACapConfirm™ algorithm is not beat-by-beat. Once the automatic atrial pulse amplitude is adjusted, it will stay in effect until the next threshold search. Threshold searches are run every 8 or 24 hours, as programmed, and the atrial threshold is determined by overdrive pacing the atrium if necessary. The overdrive rate is determined using the average atrial sensed rate and variance. If the algorithm is set to “ON”, once the threshold is determined, the atrial pulse amplitude is set to a fixed voltage above the threshold, ensuring a safety margin of at least 1.7x until the next threshold search. In order to enable ACapConfirm™, the pacing pulse configuration must be programmed to bipolar and the ACap Confirm setup test must be run. Like ventricular Autocapture™, the setup test will determine the amplitude of the evoked response compared to the measured lead polarization to ensure that there is an appropriate safety margin. If an appropriate safety margin exists, the test will recommend that ACapConfirm™ can be programmed “ON”.6
During an implant procedure, the current of injury associated with lead placement is typically the highest and the measured evoked response is typically lower until the lead matures. Clinical evaluations have shown that the ACapConfirm™ feature is more likely to be recommended as the lead matures, specifically at one and three months.
In our unselected population of patients, a relationship ER to polarization too small in the two groups studied was the most common reason to reject the AcapConfirm™ activation.
We found that a short interelectrode distance (1, 1 mm) can be associated with a lower likelihood (non-statistically significant) of automatic atrial capture than typical interelectrode distance (10 mm) using an evocated response based algorithm (AcapConfirm™). However, the automatic atrial threshold by AcapConfirm™ were significantly lower in the group with short interelectrode distance.
The ability to consistently and accurately detect capture depend on the relative magnitudes of the stimulated cardiac signal (evoked response) and the pacing-induced afterpotential (polarization). Ways to reduce the pacing polarization include increasing electrode surface area, reducing electrode polarization by coating (IROX, Pt Black, or TiN),7,8 or using smaller coupling capacitance in the stimulation circuit.9 When the polarization amplitude was low, the ER and AERI did not change appreciably with stimulus voltage. With a high-polarization electrode, the evoked-response waveform and AERI changes significantly with variations in the stimulus voltage. The stimulus duration does not affect the capture-evoked response, but influences the magnitude of the polarization signal.10
When the polarization signal is large in proportion to that of the evoked response, capture detection is impossible.10 Other factors can influence the viability of AcapConfirm™ feature: a periodic amplitude modulation associated with respiration can affect the IEGM signal and fusion of a paced atrial depolarization with a spontaneous P wave often results in a small or even nonexistent ER, myopotential noise induced by various maneuvers has a demonstrable impact on AER sensing and, for certain exercises, cardiac beats exhibit a signal-to-noise ratio less than 2.11
This study represents observational data from a single centre and thus should be considered exploratory. The major limitation is the small number of patients included, which means that the results will have to be confirmed by other groups as well.
According to our data, a short inter electrode distance (1,1mm) is more likely correlated with a lower frequency of AcapConfirm™ viability and lower threshold than a standard inter electrode distance (10mm). A small evoked response to polarization ratio was the most common cause of a negative test of AcapConfirm™ viability. Larger prospective studies are necessary to confirm our findings.