First And Second-Generation Cryoballoon Ablation Efficacy Restoring And Maintaining Sinus Rhythm In Patients Electrically Selected And Treated For Long-Standing Persistent Atrial Fibrillation After Acute Complete Electrical Disconnection Of Pulmonary Veins From The Left Atrium Demonstrated
Jesus M. Paylos MD, PhD , Luis Azcona MD, Lourdes Lacal MD, Marisol Paradela MD, Carmen Cilleros MD, Ildefonso del Campo MD, Clara Ferrero RN, Carlos Martínez MD, PhD, Aracelis Morales MD
Cardiac Electrophysiology Lab, Arrhythmia Unit and Atrial Fibrillation Center.Moncloa University Hospital. European University of Madrid, Madrid (Spain).
Introduction Cryoballoon ablation (CB) has proven effective to treatment of patients (pts) with atrial fibrillation (AF). However, the isolated efficacy of CB to treat pts with long-standing persistent atrial fibrillation (LSPAF) is less know. We analyzed the acute results and the long-term follow-up of our pts suffering LSPAF and initially treated with CB. Methods A cohort of 44 pts, 37 male (84%) mean age (60±10 year) suffering LSPAF were treated with first (CB1): 15pts, and second (CB2): 29 pts, generation CB. Eight pts (18.1%) had structural heart disease. Prior to CB, all pts were previously electrically cardioverted (CV) and sinus node and A-V nodal function evaluated at electrophysiological study (EP) once in sinus rhythm (SR) before antiarrhythmic drugs (AAD) load. CB ablation procedure was performed after three months waiting period on AAD following CV/EP drug testing. Result CB procedure was performed in 27 (61.4%) in AF, restoring SR in 8 (18.2%). PV isolation (PVI) was achieved in 95.2%. On follow-up of 30±39 months, 16 pts (40%) had AF recurrence. Second procedure (Redo) was performed in 7 pts. After a single procedure, 24 pts (60%) remain in SR without AAD, after Redo, 29 pts (72.5%), and when AAD added, 31 pts (77.5%) remain in SR. Phrenic nerve palsy (PNP) occurred in 9% of pts (75% with CB2). Conclusion CB technique is safe and useful tool to treat pts with LSPAF with 60% success rate maintaining SR without AAD in a long-term follow-up (30±39 months), up to 72.5% after Redo, and to 77.5% when AAD are added. In the majority of pts maintaining SR (77.5%) CB2 was used in 87% of the cases. Patients without structural heart disease along with those who SR was restored during CB showed the best result.
Key Words : Cryoballoon, Long-Standing Atrial Fibrillation, Ablation.
Correspondence to: Jesús M. Paylos,
Director, Cardiac Electrophysiology, Arrhythmia and Atrial Fibrillation Center
Moncloa University Hospital. HLA-ASISA Group. Avenida de Valladolid 83.
28008 Madrid. Spain.
The electrical disconnection of the pulmonary veins (PV) from the left atrium (LA), by CB catheter ablation, has proven effective in the definitive treatment of paroxysmal atrial fibrillation (PAF). The clinical success rate published being around 70-80% in terms of maintenance of SR, in a short-medium and long-term follow-up.1-4 However, the single efficacy of CB alone, restoring and maintaining SR in pts with persistent atrial fibrillation in less well-known in a medium to long-term follow-up, and unknown to date for LSPAF on a long-term follow-up basis.
In the last few years a CB2 has emerged in clinical practice entailing technical modifications over the former CB1, producing a more homogenous intake and distribution of the refrigerant, resulting in a larger and more uniform zone of freezing on the balloon’s surface, as the cooling zone spans over the entire distal half of its surface, minimizing the impact of balloon orientation on optimal tissue contact, which should induced deeper and more permanent tissular lesions, aiming to improve the procedural outcome in pts treated for AF.
Comparison between CB1 and CB2 showed significant differences at one- year follow-up in the general outcome of pts treated for PAF.5 Some studies6 having reported a success rate of CB ablation on persistent AF using CB1 in 50% of pts at a mean of 18(6-27) months of follow-up.
However CB2 was found to have better outcomes compared to CB1 among pts with PAF and persistent AF.7-8
The first study to report one year follow-up outcome after CB PVI using CB2 was conducted by Metzner et al 9 with a success rate of 77% in a limited number of 14 pts with short-standing persistent AF (< 3 month duration) along with 81% one year clinical success rate for PAF in the same study.
Although the first study focusing solely on persistent AF using CB2 as an index procedure has been described by Ciconte et al10 reporting a success rate of 60.3% of pts in SR at one year follow-up. The largest survey of pts treated with CB2 for persistent AF, including one year outcome has been more recently described by Koektuerk et al10 with 67% of pts in SR at a mean follow-up duration of 10.6 ±6.3 months.
The use of Adenosine (AD) to unmask dormant PV tissue after extensive encircling demonstration of PVI has demonstrated clinical implications after AF ablation procedures and may reduce clinical AF recurrences.12 Some observations having shown the use of AD to guide elimination of dormant PV conduction to increase the success rate of AF ablation procedures.13
We aimed to assess the efficacy of CB catheter ablation in our pts treated with CB suffering LSPAF according to the latest guidelines 14 criteria and initially treated with CB1 and CB2 and elimination of residual conduction (RC) gaps unmasked by AD and guided by bidirectional LA-PV-LA conduction block as the main target end point.
Since November 2008 to November 2015, a cohort of 44 pts, 37 male (84%) mean age: 60±10 year, and 7 female (16%) mean age 64±5 years, suffering from LSPAF were treated with CB1: 15 pts (34%) and CB2: 29 pts (66%).
Clinical and demographic characteristics of the study population are provided in Table 1.
The mean time duration of stable arrhythmia was 4±3 years,(1-13) All pts were previously treated with AAD: BtB: 36pts (81.8%); class III (amiodarone): 3pts (6.8%); class 1C: 31 (70.4%), and BtB+1C: 27 pts (61.3%). Eight pts (18.1%) had structural heart disease: 2 hypertrophic obstructive cardiomyopathy (4.5%); 3 chronic ischemic heart disease (6.8%); 2 dilated cardiomyopathy (4.5%) and 1 non-compaction cardiomyopathy (2.2%). Morphological and structural pts data it’s provided in Table 2.
Table 1. Clinical and demographic characteristics of the study population
| Variable |
|
Pts (n=40) |
| (n=40) |
|
55±10 |
| Male (37 pts) |
|
60±10 |
| Female (7 pts) |
|
64±5 |
| Hypertension |
|
22(50%) |
| Diabetes Mellitus |
|
2 (4.4%) |
| IHD |
|
3 (6.8%) |
| HOCM |
|
2 |
| NCCM |
|
1 (2.2%) |
| DCMP |
|
2 (4.5%) |
| PM |
|
5 |
| LSPAF mean year duration |
|
4+3 (1-13) |
| Follow-up time |
|
2.8 years 30+39 months |
|
Class IC ( Flecainide) |
31 |
|
B-Blocker BtB) |
36 |
| AAD |
Amioradone |
3 |
|
Class IC+BtB |
27 |
IHD: Ischemic heart disease. HOCM: hypertrophic obstructive cardiomyopathy. NCCM: non compaction cardiomyopathy. DCMP: dilated cardiomyopathy. PM: pacemaker
AP: anterior posterior long parasternal axis. SI: supero-inferior. LAT: lateral (transversal). LVEF: left ventricular ejection function. LCT: left common trunk.
Selection of Patients for CB-PV ablation
All pts included in the study provided informed consent prior to the procedure, which was approved by the hospital’s clinical ethics committee.
1. Criteria of exclusion for pt to treat included:
1. Prior stroke or TIA.
2. Cryoglobulinemia and hematological or coagulation disorders.
3. Presence of intracavitary thrombi.
4. Significant associated comorbidity.
5. Left atrial size≥50mm.
6.Failed CV to reverse AF.
The CB ablation protocol for this group included the following approach:
All pts underwent to CV and electrophysiological study (EP) once cardioverted in SR to assess sinus node and A-V nodal function in a basal stage, and were reevaluated after intravenous load of AAD adjusted to pt body weight, mostly class I C, flecainide: 1.5 mgr /kg and calcium channel blocker verapamil: 0.15 mgr/kg for pts without structural heart disease, and class III, amiodarone and/or verapamil for pts having structural heart disease, in an attempt to estimate the sinus node and A-V nodal electrical response to the limit for better achieving early electrical stabilization of the remodeled atrium to sustained SR, allowing the pts to perform routine daily physical activity with the least adverse side effects (mostly bradycardia-related) to avoid patient/physician decision for medication to be discontinued.
Any individual pt whose SR was not restored at CV was excluded for CB ablation. Patients recovering SR were discharged from the hospital after CV/EP procedure on oral AAD including verapamil and were follow-up for the next 3 months before CB-PV ablation procedure. Oral anticoagulation with vitamin K antagonist dicumarol, when indicated, was based on CHA2DS2-VASc score targeting an international normalized ratio (INR) within the 2.0-3.0 range.
Previous studies and anatomical approach: CT scan 3D/high resolution /64 slide multidetector (Toshiba Aquilion 64, TSX-101A, Tokyo, Japan) as well as in some alternative cases, RMN (1.5T/ Magnetom Symphony, Siemens, Germany) were used for typification and better definition of the cardiac anatomy, morphology, number, caliber and size of PV as well as endoluminal PV analysis to assess the thickness of the interpulmonary ridge and the morphological shape and size of the PV ostium to choose the optimal CB size and the best orientation to address the balloon wedging at the LA-PV junction level in an attempt to induce the greatest cryo-lesion at the most proximal antral location including the interpulmonary ridge at the carina level in any individual PV faced for CB applications.
Table 2. Morphological and structural patient population data
| Structural Heart disease (n=8)
|
| Diameters (mm) |
LA |
PV (30) |
LCT (2) |
LVEF 61±6
|
| AP |
44±5 (34-48) |
19±6 (14-28) |
26±1 (25-27) |
|
| SI |
61±5 (55-65) |
22±5 (16-32) |
32±3 (30-35) |
mean LA size
(cm2) 29±3
(24-33) |
| LAT |
50±6 (43-60) |
|
|
|
| Non Structural Heart disease (n=36) |
| Diameters (mm) |
LA |
PV (136) |
LCT (8) |
LVEF
66%±7 |
| AP |
40±5 (33-49) |
20±5 (14-30) |
27±4 (25-30) |
|
| SI |
|
|
|
|
|
55±8 (42-70) |
22±4 (9-32) |
30±3 (30-35) |
mean LA size (cm2)
25±5 (14-36) |
| LAT |
47±8 (41-66) |
|
|
|
AP: anterior posterior long parasternal axis. SI: supero-inferior. LAT: lateral (transversal). LVEF: left ventricular ejection function. LCT: left common trunk.
Transthoracic 2D echocardiogram (TTE) in addition to transesophageal echocardiogram was performed on the same day in all cases, to assess cardiac anatomy and to rule out intramural thrombi. All procedures were performed under general anesthesia with orotracheal intubation under propofol for anesthesia induction, cisatracurium for neuromuscular relaxation (only at the time of intubation), continuous perfusion of remifentanil for analgesia and mechanical ventilation maintained with sevoflurane gas.
Transeptal approach: Seldinger technique was used for all vascular access. A decapolar 6 French electrocatheter introduced through an antecubital vein, was positioned into the coronary sinus (CS) for pacing and anatomical reference purposes. Cuatripolar/6French catheter was positioned at the A-V-nodal-his bundle junction through left femoral vein, for the same anatomical reference purpose, moving later to superior vena cava (SVC) for pacing during CB applications at the right sided PV.
Through right femoral vein, an introducer and fast-cath 8.5 French sheet SLO, (Saint Jude medical, Minnesota, USA), was advanced over a 0,32 mm J typed shape guide wire to the SVC. Then, the guide wire was withdrawn, and a modified Brockenbrough needle (BRKO 71 cm beveled cut 30º/ Saint Jude Medical, MN, USA) is advance through the SLO sheet, and descending the entire transeptal assembly to embed the fossa ovalis.
After gaining left atrial access, a bolus of 10.000 IU of sodic heparin was administered, followed by continuous perfusion as needed to keep the activation clotting time ≥300-350 sec. At the end of procedure, anticoagulation is reversed with protamine and 1grm. of lysine acetylsalicylate given i.v, along with low molecular weight heparine (depending on pts body weight) given subcutaneously, in addition to flecainide (1.5 mgr/Kg) given intravenously in 10 minutes. Continuous intravenous perfusion of sodium heparine adjusted to pt’s body weight is started 4 hours later after removing all catheters from the vascular bed. Twenty four hours later, oral anticoagulation with vitamin K antagonist dicumarol is started targeting an international normalized ratio (INR) within the 2.0 to 3.0 range, in addition to antiplatelet inhibition agent ASA, 100 mgr daily dose.
PV/Cartography/ Mapping: Once in the LA chamber, the long 0’32mm guide-wire is advanced into the left superior PV (LSPV) and selective PV angiogram is performed, and in the same manner for the remaining veins, Left Inferior (LIPV), Right Superior (RSPV) and Right Inferior (RIPV).
After removing the entire transeptal assembly, keeping the guide-wire in the LSPV, and steerable 15F over-the wire sheath (Flex Cath, Cryocath, Medtronic,USA) is advanced and positioned in the LA. Then basal electrical cartography of the veins is obtained with a circular duodecapolar mapping catheter with adjustable diameter (Reflexion spiral, Saint Jude Medical, MN, USA) positioned at the PV-LA junction antrum level, starting on LSPV and followed by LIPV, RSPV and RIPV respectively. After 30 minutes of CB applications all PV were mapping again to assess electrical PV-LA isolation (PVI).
Cryo-Balloon: After withdrawing the circular catheter mapping, a 28 or 23 mm double walled CB catheter (Artic Front, Medtronic, USA) is advanced over the wire up to the LA, inflated and positioned in the PV ostium of each vein and gently pushed against the PV-LA antrum to get a perfect occlusion achieved when selective contrast medium injected (50% ratio with 0.9% saline solution) is fully retained in the vein with no evidence of contrast leakage back into the atrium (grade IV) in keeping with the degree of occlusion classification proposed and used by Neumann et al 2 to grade I with poor occlusion leading to an immediate rapid outflow of contrast medium back into the LA. Until CB2 was commercially available (April, 2013) pts were treated with CB1.
Bidirectional LA-PV-LA block (BB) Protocol
Exit Block: By pacing PV from all 20 poles of the circular catheter mapping at high amplitude voltage (20 mA) with consistent 1:1 PV capture and no evidence whatsoever of any atrial response.
Entry Block: By pacing LA from the CS-catheter at three different cycle lengths (600, 500, 400 mS) with consistent 1:1 LA capture and no evidence whatsoever of any PV electrical activity in any of the 20 poles of circular-catheter mapping placed at the LA-PV junction antral level.
AD Protocol: Included bolus i.v administration of increasing doses (12-18-24… mgrs), and pacing PV/LA when A-V nodal conduction block occurred.
Extrapulmonary muscular connections (EMC)/ rule-out protocol: Included pacing distal vein from the circular catheter mapping after complete BB demonstrated at the LA-PV junction antral level and the demonstration of 1:1 PV-LA conduction reassumed.
Radiofrequency protocol: Focal RF applications were used for the elimination of RC gaps when evident after single CB application or either after checking for BB, after AD, or when EMC was demonstrated.
Sixty-second “touch-up” of focal RF was used to eliminate all residual gaps only when evident in less than 2 pairs of the circular catheter mapping. Otherwise, when more, a repeated new CB application was performed.
Phrenic nerve physiology control: Phrenic nerve physiology was monitored in all cases during right sided PV/CB applications, by placing the cuatripolar electrocatheter in SVC and pacing at 2.000 mS cycle length, checking the intensity of diaphragm contractions by intermittent fluoroscopy and tactile feedback by placing the operator’s hand on the patient´s abdomen, and immediately stopping freezing when intensity of the diaphragm contraction weakens or is suddenly stopped.
Follow-up Protocol: Before hospital discharge, TTE was performed in all cases to rule out pericardial effusion, in conjunction with chest-X-ray taken in a deep breath, upright position, to confirm normal phrenic nerve physiology.
The immediate follow-up included holter monitoring at 7, 15,30,45,60 and 90 days respectively, and thorax CT-Scan at 30 and 90 days. All pts received AAD, mostly Class IC+BtB and oral anticoagulation with vitamin K antagonist dicumarol is started 1 day after PVI, targeting an INR within the 2.0 -3.0 range for at least three months.
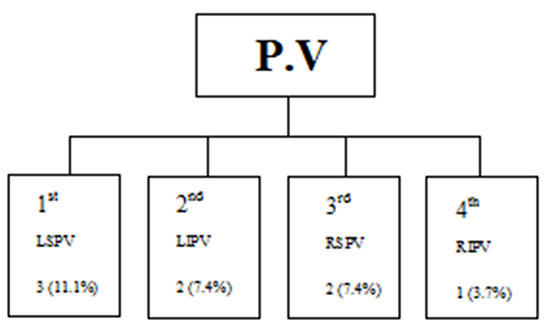
Figure 1 AF reversion/during CB application 8/27 AF pts (29.6 %)
After a 3 month-blanking period on medication, all AAD were discontinued, and follow-up started to count. All pts were monitored by continuous daily transtelephonic information in case of symptoms, monthly ECG holter monitoring having otherwise being routinely done over a mean follow-up of 2.8 years.
Three months after CV, 17 pts (38.6%) remained in SR. The other 27 pts experienced relapses into AF (RAF). The entire group of 44 pts, were included for CB ablation procedure. At the procedure, none of the 27 RAF pts were CV before CB ablation. SR was restored in 8 pts (18.2%) during CB applications at a different PV and numbers Figure 1.
In the other 19 of 27 RAF pts, CV was performed, and all converted in SR, allowing checking for BB.
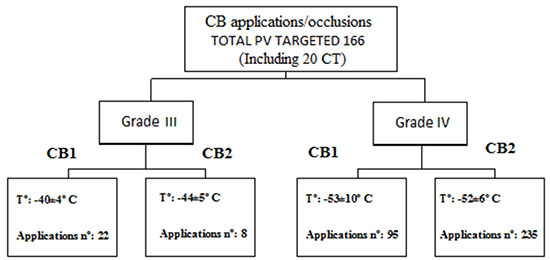
One hundred and sixty six PV including 10 left sided common trunks, (CT) were treated with CB1, 58 PV, (35%) and 108 PV with CB2 (65%) and PVI with complete BB was demonstrated in 158 PV (95.2%).
Three of the others 8 PV, needed to be isolated with RF applications giving that the CB-PV antral anatomical wedging was difficult to approach, not allowing to achieve a complete degree IV CB-PV occlusion.
The other 5 PV showed acute reconnection due to RC gap in LSPV in the inferior segment and in the other 3PV, an extrapulmonary muscular connection (EMC) could be demonstrated. All RC and EMC were eliminated by focal RF applications. None RC were unmasked after AD.
CB1 was used to treat the first 15 pts, until CB2 become commercially available. The number of CB/PV applications, degree of occlusions and temperature reached are provided on Figure 2.
Arrhythmia Recurrence And PV-LA Reconduction
After a three month blanking period on medication, AAD were discontinued and the follow-up started to count. Only 40 pts were included in the follow-up, as the other 4 were still in the blanking period.
In a mean follow-up of 30±39 months (7-84), 16 pts (40%) had recurrence of stable arrhythmia.
Among them, 7 had structural heart disease, meaning 87.5% of the total pts with structural heart disease.
In all recurrence pts, oral AAD was again reinitiated, restoring SR in
Figure 2 CB1-CB2 applications: degree of occlusions and mean nadir temperature reached
7 pts, who allowed for Redo. The other 9 recurrence pts including 7 with structural heart disease refuse Redo and continuing in AF.
Twenty six PV (including 2CT) from the 7 recurrence pts in whom SR was restored after AAD was added, were study again in Redo. In the 2 recurrence pts in whom SR was restored after AAD, all 7 PV (including 1CT) were completely electrically disconnected from the LA.
In the other 5 recurrence pts new CB-PVI was achieved with CB1 in 1pt and with CB2 in the other 4pts.
No RC was unmasked after AD.
In all pts who refused Redo and remained in AF after a single CB procedure, the type of balloon used was CB1.
In summary, in this study population in a mean follow-up of 30±39 months, 24 pts (60%) remain in SR after a single procedure without AAD. After Redo, 29 pts (72.5%) remain in SR, and when medication is added, 31 pts (77.5%) stay in SR.
Side Effects And Complications
Two pts (4.5%) had aphonia. One pt (2.2%) presented groin haematoma due to inadverted arterial puncture resulting in pseudoaneurysm requiring surgical repair. Transient PNP occurred in 4 pts (9%) during CB-RSPV application, recovering full PN function in the course of the procedure. Type of CB used along with mean nadir temperature reached when PNP occurred it’s provided on table 3.
AF is the most disturbing and common clinical arrhythmia encountered in clinical practice with a prevalence of around 1% in developed countries, increasing the risk of heart failure and being responsible for nearly 15% of all strokes in the USA. 15
Although it was unclear why some pts stay in PAF for decades, whereas others with the same characteristics, progress to persistent AF within a few months, the most widely-accepted theory, is based on remodeling concept, yet no fully understood to date.
Atrial stretch, shifts in autonomic tone, or depletion of high-energy phosphates it has been demonstrated do not significantly contribute to atrial electrical remodeling,16,17 wich is mediated by rate-induced intracellular calcium overload. The high rate of electrical activation itself provides the stimulus for the AF-induced changes in shortening the atrial effective refractory period (AERP).18 Electrophysiological changes that contribute to sustain the arrhythmia in a self-perpetuating process supporting the concept of “AF begets AF”.19 Among many others different risk factors to build up the underlying substrate, such as age, hypertension, or structural heart disease, the common factor of tissue substrate is fibrosis wich can be an important factor in the maintenance and progression of AF.20
Table 3. Type of CB used along with mean nadir temperature reached when PNP occurred it’s provided
| Transient Phrenic Nerve Palsy |
|
Tº C |
Seconds |
CB (mm) |
CB Generation |
Mean T
≥60
(75%)
|
| 1 |
-62 |
120 |
28 |
First |
| 2 |
-64 |
123 |
23 |
Second |
| 3 |
-68 |
103 |
23 |
Second |
| 4 |
-54 |
194 |
23 |
Second |
CB2: 75% |
Many questions however still as yet unanswered on the order of explaining the multifactorial aspects involved in the atrial remodeling process and their relationship to the amount of fibrotic tissue related to the arrhythmia “phenotype” (paroxysmal, persistent and long-standing persistent).
Electroanatomic mapping studies,21,22 intraoperative obtained specimen analysis,20 post-mortem autopsy findings 23 and late gadolinium delayed enhancement MRI studies 24 have demonstrated a higher mean value of fibrosis detected in pts with persistent AF vs PAF but the variability in the extend of fibrosis is always very high with part of pts with PAF having massive fibrosis an part of persistent AF pts showing mild fibrosis supporting the concept named by Kottkamp 25,26 of fibrotic atrial cardiomyopathy (FACM). According with Kottkamp editorial commentary,27 the structural atrial disease underlying FACM typically does not present with enlarged left atria and is typically undiagnosed before the clinical advent of the first arrhythmia as AF, but a potential sub-diagnoses or disease manifestations are atrial tachycardia, sinus node and or AV node disfunction wich can be severe.
Although some studies28 have demonstrated reverse remodeling of sinus node function after catheter ablation of AF, suggesting that prolonged sinus pauses after PAF may result from depression of sinus node function that can be eliminated by curative ablation of AF. The association of AF and sinus node disfunction, it has being known for a longtime as configuring the sinoatrial disease responsible for brady-tachycardia syndrome as well the evidence of diffuse atrial remodeling and fibrosis in sinus node disease.29-30
Animal experiments have shown16-19 that AF induces shortening of the AERP resulting in an increased vulnerability for reinduction of AF, prolongs intra-atrial conduction time, shortens atrial refractoriness and perpetuates AF,16,17 changes that reverse gradually after termination of AF. In humans, this electrical remodeling was completely reversible within a week after CV, and presumably related to intracellular calcium overload.31 Tachycardia-induced change of AERP in humans has been demonstrated by others 32 and the rate-dependency and effects of antiarrhythmic drugs. Yu et al33 in this study including 70 pts with PAF without structural heart disease and EP performed after pacing-induced AF analyzing the changes on AERP pre and post AAD (class IC, BtB, amiodarone and verapamil) showed the AERP shortening was attenuated after verapamil infusion, but was unchanged after infusion of the other AAD. Furthermore, all these AAD could decrease the incidence and duration of secondary AF. The AERP shortening induced by tachycardia was a rate-dependent response, and verapamil, but no other AAD, could markedly attenuate this effect.
Reversal of atrial electrical remodeling following CV in pts with long-standing (> 6 months duration) (mean 62±36 months) range 16-132 months was demonstrated also by Yu et al33 in 19 pts showing the AERP was gradually prolonged and its rate adaptation response improved after restoration of SR, concluding than restoration and maintenance of SR could reverse these electrophysiological changes. Additionally, the possibility of conversion and maintenance of SR was decreased as the duration of AF increases.34 Clinical observations have demonstrated that the recurrence of AF was frequently clustered in the first month after CV.35
All of these data support the view that AF causes electrical remodeling wich facilitates the perpetuation of AF itself. From the clinical point of view, the early recurrence of AF after successful CV may be due to the adverse effects of electrical remodeling, and calcium-blocker might provide a beneficial effect in this situation. Conversion of AF and maintenance of SR could abolish the adverse effect of electrical remodeling. Wether the atrial remodeling process is reversible after the ablation of AF most likely depends on the degree of remodeling at the time of successful ablation.27
According to all the statements aforementioned, we sought to select among pts with LSPAF: Firstly, does who theoretically involved the least degree of electrical atrial remodeling able to sustain SR after CV. Secondly, among these pts selected, to try to electrically stabilized the atrium with the highest degree of drug tolerance to sustained SR, including verapamil, in an attempt to lengthen the AERP to reverse the acute-rate dependent electrophysiological changes responsible for early AF recurrence within the first week after CV.16-19,30-33 In fact, a pacemaker needed to be implanted in 5 pts due to impaired sinus node and A-V nodal function demonstrated after CV, before giving AAD.
After CV/EP AAD testing procedure, the entire groups of pts were discharged from hospital on AAD, including verapamil, after which a three month waiting period commenced during which the pts returned to their normal life.
We did so, on the basis of the aforementioned concepts, that conversion of AF and maintenance of SR could minimize the adverse effects of electrical remodeling, and this probable reverse atrial remodeling most likely depends on the degree of remodeling at the time of successful ablation.27
During this three month waiting prior to CB ablation procedure, 17 pts (38.6%) remained in SR while the rest 27 relapsed into AF, performing the CB ablation procedure being in AF.
We have not found any significant difference between the atrial size and the number of years suffering from arrhythmia in any of the different pt’s group, although the mean years duration of stable arrhythmia and left atrial size was slightly higher (6±5 years) in pts who stay in AF comparing to pts whom SR was restored.
Only one pt with structural heart disease (ischemic) sustained SR on follow-up, having the lowest duration time of stable arrhythmia (1 year).
Type of CB Used/Related Findings
All PV reconduction after CB (5pts) became evident only after CB1 applications. In all 8 pts in whom AF was converted to SR, CB2 was applied in 7 (87.5%). No RC was shown after CB2 applications. All RC showed in Redo pts, the first procedure having being performed with CB1. However, CB2 showed the highest incidence rate of PNP (75% CB2 vs 25% CB1). Both CB1+CB2 totaling 9% of the incidence PNP rate in the entire group, consistent with the PNP rate found by others.36
On the contrary, the lower vascular access incidence complication rate (2.2%), compared to others (10%) 11 might be related to the fact of all procedures having being performed on same operator (JMP).
Aphonia of unexplained etiology was completely spontaneously resolved within less than 72 hours.
No major complications occurred. No PV stenosis. Interestingly, in comparison to previous studies37 and our own experience in 128 pts treated mostly with CB1, RC was unmusked by AD when using CB1 in 4.6% of PV treated,37 and in 4% of PV when CB2 was applied.38 By the contrary, in this study none RC were unmasked by AD when CB2 was used, most probably related with the larger and deeper cryolesions created. According with these findings, we will abandon the use of AD approach due to as the possibility of PV dormant tissue after CB2 applications it will likely being none.
Continuous variables are expressed as mean ± SD.
Categorical variables are expressed as percentages.
From the transeptal approach to withdrawal of LA catheters: 173±47 minutes (95-26)
Mean fluoroscopy time: 43±14 minutes (24-48).
This study has several limitations. First: this is a non-randomized, single center study. Second: this study includes a small sample size. Third: the previous electrical selection of patients likely to limit the outcome that can be extended to the entire LSPAF population group. And, fourth: as a significant number of pts from this group, felt no symptoms, along with the wide interval range (30 days) in between holter ECG recording, the final outcome results might be overestimated.
As a result of this study, we might to conclude: 1. CB technique is a safe and useful tool for treating pts with LSPAF, with a 60% success rate in terms of maintaining SR without medication in a mean follow-up of 30±39 months after a single procedure, increasing the success rate up to 72.5% after a second procedure, and to 77.5% when ADD are added. 2. In the majority of pts maintaining SR (77.5%) (including Redo+AAD), the CB/PVI was achieved with CB2 in 87% of the cases. 3. In all pts who refused Redo (22.5%) having continued in AF, CB1 was used at first procedure. 4. CB2 is better than CB1 to restore and sustain SR after PVI in pts with LSPAF, where bigger antral lesion is needed. However, this entails a higher risk of extracardiac structures damage, mostly PNP (9%). 5. Patients without structural heart disease along with those in whom SR was restored during CB applications, showed the best results. 6. Finally, the main limitation of this study might be due to the small sample size in previously selected pts. Further larger randomized studies are needed to confirm our findings.
We gratefully thank to Dr. Francisco Ivorra Miralles, president of HLA-ASISA group for his continuous support and economical resources in favor of clinical patient care and research.