Alcohol consumption in moderation has been reported to have beneficial cardiovascular protective effect.[1], [2] Recent evidence continues to emerge on the physiologic and genetic mechanisms through which alcohol may reduce the risk of developing CVD.[3] Nonetheless, alcohol intake has been implicated with an increased incidence of atrial fibrillation.[4] The heart holiday syndrome, in fact, was first described in 1978 by Ettinger et al and linked to binge alcohol drinking behavior preceding AF occurrence.[5] Incidence of AF is also elevated in chronic levels of modest alcohol intake.[6] Abstinence from alcohol remains the optimal management of alcohol related heart disease including atrial fibrillation.[7]
Catheter ablation has become the mainstay of therapy in many symptomatic patients with atrial fibrillation. In addition, long-term control of AF requires modification of risk factors and management of associated comorbidities.[8], [9] Identifying predictors of AF recurrence after catheter ablation could help focus the management of these potential risk factors and better select patients who would likely have favorable outcomes. A recent study has shown that alcohol consumption may be an independent predictor of paroxysmal AF (PAF) recurrence after catheter ablation,[10] but the study was small and did not include persistent AF patients. In this study, we further explore the association of different alcohol consumption levels with early and late recurrence rates of AF (both paroxysmal and persistent) and AF-free survival after pulmonary vein isolation by catheter ablation.
Patient Population and Ablation Procedure
Two hundred twenty six consecutive patients with symptomatic, non-valvular AF underwent their first PVI in the period of January 2011 to April 2014. Baseline characteristics of patients in each group are summarized in [Table 1]. The only significant difference was the higher prevalence of diabetes mellitus in the none-rare alcohol group. For all patients, mean age was 62.1 +/- 9.9 years (range 31-84, median=63 years) and 153 (67.7%) were men. PAF was present in 163 (72.1%) patients. The prevalence of PAF was higher as the level of alcohol consumption increased; however, this was not statistically significant among the alcohol groups. Mean AF history was 4.1 +/- 4.9 years (range 2 months to 46 years). Most patients (56.2%) had one drug failure history while 15.0% had 2 or more drug failures.
Table 1. Patient baseline characteristics by alcohol groups
|
Parameter
|
None - Rare
(n=132)
|
Moderate
(n=74)
|
Heavy
(n=20)
|
P value
|
|
Age, years
|
61.3 +- 10.7
|
62.7 +- 9.0
|
64.3 +- 6.9
|
0.361
|
|
Male sex, n (%)
|
86 (65.2%)
|
51 (68.9%)
|
16 (80.0%)
|
0.427
|
|
BMI, kg/m2
|
30.0 +- 6.0
|
28.8 +- 6.6
|
28.0 +- 4.9
|
0.219
|
|
Paroxysmal AF, n (%)
|
89 (67.4%)
|
56 (75.7%)
|
17 (90.0%)
|
0.081
|
|
History of EPS or ablation not for AF, n (%)
|
18 (15.8%)
|
15 (21.7%)
|
2 (12.5%)
|
0.574
|
|
AF history duration, years
|
4.07 +- 4.06
|
3.99 +- 6.52
|
4.98 +- 3.74
|
0.775
|
|
HTN, n (%)
|
79 (59.8%)
|
35 (47.3%)
|
12 (60.0%)
|
0.222
|
|
DM, n (%)
|
28 (21.2%)
|
4 (5.4%)
|
3 (15.0%)
|
0.007
|
|
CAD, n (%)
|
28 (21.2%)
|
10 (13.5%)
|
4 (20.0%)
|
0.397
|
|
CHADS2 score
|
1.4 +- 1.3
|
1.0 +- 1.0
|
1.3 +- 0.9
|
0.060
|
|
Number of failed AADs
|
1.2 +- 0.8
|
1.1 +- 0.8
|
1.4 +- 0.9
|
0.376
|
|
Ablation of additional linear ablation, n (%)
Ablation of additional CFAE, n (%)
Ablation of additional non-PV trigger, n (%)
|
71 (53.8%)
11 (8.3%)
10 (7.6%)
|
40 (54.1%)
6 (8.1%)
9 (12.2%)
|
13 (65.0%)
2 (10.0%)
4 (20.0%)
|
0.693
0.874
0.152
|
|
LAVi, ml/m2
|
31.9 +- 13.3
|
33.0 +- 18.7
|
38.7 +- 15.0
|
0.302
|
|
LVEF > 50%, n (%)
LVEF 41 - 49%, n (%)
LVEF 36 - 40%, n (%)
LVEF < 35%, n (%)
|
98 (89.9%)
6 (5.5%)
1 (0.9%)
4 (3.7%)
|
64 (94.1%)
3 (4.4%)
1 (1.5%)
0 (0.0%)
|
15 (93.8%)
1 (6.3%)
0 (0.0%)
0 (0.0%)
|
0.724
0.894
1.000
0.392
|
|
Early AF recurrence, n (%)
Late AF recurrence, n (%)
|
39 (30.2%)
44 (37.0%)
|
21 (28.8%)
25 (36.8%)
|
5 (26.3%)
4 (22.2%)
|
0.974
0.500
|
BMI: body mass index; EP: electrophysiology study; AF: atrial fibrillation; HTN: hypertension; DM: diabetes mellitus; CAD: coronary artery disease; CHADS2 score: congestive heart failure, hypertension, age, DM, stroke/transient ischemic attack score for the thromboembolic risk assessment; AADs: antiarrhythmic drugs; CFAE: ablation of complex fractionated atrial electrograms; PV: pulmonary vein; LAVi: left atrial volume index; LVEF: left ventricular ejection fraction.
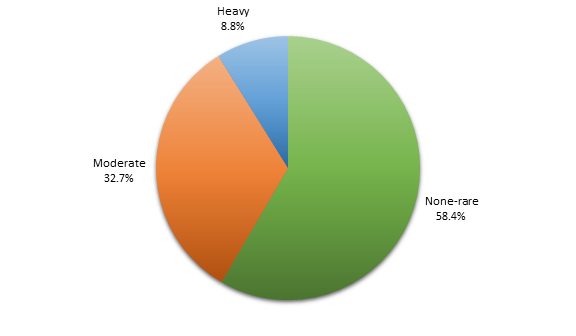
All of the patients were undergoing PVI for the first time; however, prior catheter ablation had been performed in 23 patients for atrial flutter (AFL), 1 for AVNRT and 2 for VT. Eleven patients had a history of sick sinus syndrome. Pacemakers were present in 22 patients (20 dual chamber, 1 single chamber, and 1 biventricular pacemaker). An alcohol use survey on admission showed that the majority of patients, 132 (58.4%), had none to rare (<1 drink per week) alcohol use, while 74 (32.7%) and 20 (8.8%) patients reported moderate (1-7 drink per week) and heavy (>1 drink per week) alcohol use, respectively ([Figure 2]).
Figure 2. Distribution of study population (patients undergoing first time PVI) demonstrated by alcohol consumption levels
Of those presenting for AF ablation, 63 (27.9%) patients had PsAF. PVI was performed using radiofrequency ablation in most patients, except in 9 patients where cryoballoon ablation was used. Additional linear, CFAE and non-PV trigger ablation was performed, in 54.9%, 8.4%, and 10.2% of patients, respectively.
Characteristics of Patients by Alcohol Consumption Levels
Increased alcohol consumption levels are positively correlated with older age, male sex, higher rate of PAF, greater left atrial volume index (LAVi) and further need of additional linear, CFAE and non-PV trigger ablation ([Table 1]).
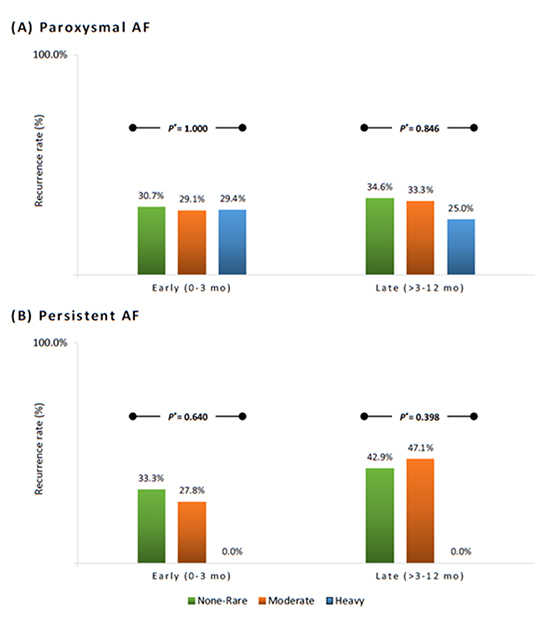
In the blanking period (0-3 months), 5 PAF patients were lost to follow up. In the following 3-12 month follow up period, an additional 12 PAF patients and 4 PsAF patients were lost to follow up. Early and late recurrence rates were 29.1% and 32.2% in the PAF group, and 30.2% and 44.1% in the PsAF group, respectively. For PAF patients, early recurrence rates were 30.7%, 29.1% and 29.4% (p=1.000) and late recurrence rates were 34.6%, 33.3% and 25.0% (p=0.846) in the none-rare, moderate and heavy alcohol groups, respectively ([Figure 3A]). In PsAF patients, early recurrence rates were 33.3%, 27.8% and 0.0% (p=0.640) and late recurrence rates were 42.9%, 47.1% and 0.0% (p=0.398) in the none-rare, moderate and heavy alcohol groups, respectively ([Figure 3B]). Among those patients who had an early recurrence, 68.4% developed late recurrences. The presence of early recurrence was independently predictive of late recurrence after catheter ablation based on multivariable logistic regression models adjusting for linear ablation, AF type, and DM (OR 7.797, 95% CI 3.815-15.937, p<0.001).
Figure 3. Recurrence rates of PAF (A) and PsAF (B) after catheter ablation in the none-rare, moderate and heavy alcohol consumption groups in the early and late follow up periods. *P value for statistical difference among the three groups of alcohol consumption.
On univariate logistic regression analysis, none of the alcohol groups showed any significant association with early or late AF recurrence ([Table 2]). Among the baseline characteristics, only age and the absence of diabetes mellitus were significantly associated with higher early and late AF recurrence rates, respectively [age (OR 1.032, 95% CI 1.000-1.065, p=0.050) and absence of DM (OR 0.300, 95% CI 0.110-0.818, p=0.019)].
Table 2. Univariate logistic regression analysis of early and late AF recurrence by patient characteristics
|
Parameter
|
0 - 3 month
|
3- 12 month
|
|
|
OR*
|
95% CI
|
P value
|
OR*
|
95% CI
|
P value
|
|
Age
|
1.032
|
1.000-1.065
|
0.050
|
1.019
|
0.989-1.050
|
0.223
|
|
Sex (female)
|
0.889
|
0.477-1.658
|
0.711
|
1.319
|
0.718-2.422
|
0.372
|
|
BMI
|
1.036
|
0.990-1.085
|
0.129
|
1.020
|
0.973-1.069
|
0.418
|
|
AF type (PAF vs. PsAF)
|
0.951
|
0.502-1.801
|
0.878
|
0.603
|
0.324-1.120
|
0.109
|
|
AF history duration
|
1.030
|
0.969-1.094
|
0.344
|
1.032
|
0.957-1.114
|
0.414
|
|
HTN (presence vs. absence)
|
1.010
|
0.565-1.808
|
0.972
|
0.851
|
0.480-1.510
|
0.581
|
|
DM (presence vs. absence)
|
0.842
|
0.370-1.919
|
0.683
|
0.300
|
0.110-0.818
|
0.019
|
|
CAD (presence vs. absence)
|
0.544
|
0.236-1.255
|
0.153
|
1.027
|
0.485-2.173
|
0.945
|
|
CHADS2 score
|
1.062
|
0.840-1.343
|
0.617
|
0.965
|
0.782-1.267
|
0.969
|
|
Number of failed AADs
|
1.036
|
0.717-1.497
|
0.849
|
1.006
|
0.699-1.447
|
0.976
|
|
Alcohol use
None-Rare vs. more
Moderate vs. none-rare
Heavy vs. none-rare
|
0.965
1.100
0.932
0.824
|
0.839-1.110
0.610-1.983
0.496-1.751
0.278-2.447
|
0.617
0.751
0.827
0.728
|
0.950
1.153
0.991
0.487
|
0.828-1.090
0.645-2.063
0.534-1.838
0.151-1.572
|
0.465
0.631
0.977
0.487
|
|
Additional (presence):
Linear ablation
CFAE ablation
Non-PV trigger
|
0.818
2.346
1.319
|
0.458-1.461
0.906-6.078
0.530-3.283
|
0.497
0.079
0.551
|
0.679
1.682
1.127
|
0.382-1.205
0.620-4.566
0.444-2.859
|
0.186
0.307
0.802
|
|
LAVi
|
0.983
|
0.955-1.011
|
0.224
|
1.004
|
0.980-1.029
|
0.723
|
|
LVEF > 50%, n (%)
LVEF 41 - 49%, n (%)
LVEF 36 - 40%, n (%)
LVEF < 35%, n (%)
|
1.883
0.575
-
0.767
|
0.515-6.881
0.118-2.801
-
0.078-7.547
|
0.339
0.494
-
0.820
|
1.196
1.115
-
0.836
|
0.390-3.664
0.302-4.108
-
0.074-9.415
|
0.754
0.870
-
0.885
|
|
Early AF recurrence (presence)
|
|
|
|
7.265
|
3.691-14.299
|
<0.001
|
BMI: body mass index; PAF: paroxysmal AF; PsAF: persistent AF; EP: electrophysiology; AF: atrial fibrillation; HTN: hypertension; DM: diabetes mellitus; CAD: coronary artery disease; CHADS2 score: congestive heart failure, HTN, age, DM, stroke/transient ischemic attack score for the thromboembolic risk assessment; AADs: antiarrhythmic drugs; CFAE: ablation of complex fractionated atrial electrograms; PV: pulmonary vein; LAVi: left atrial volume index; LVEF: left ventricular ejection fraction. *OR refers to odds ratio of recurrence of AF versus remaining in sinus rhythm after catheter ablation.
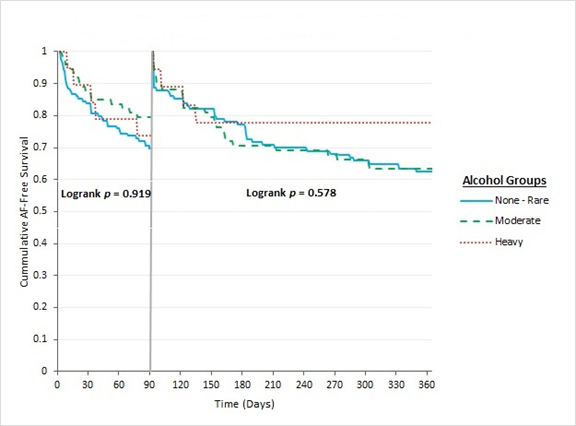
With multivariable regression models, after adjusting for potential confounding covariates, none of the alcohol levels were independently predictive of early or late AF recurrence ([Figure 4]). Paroxysmal AF and the absence of DM were the only two parameters that remained independently predictive of late AF recurrence [PAF (OR 0.410, 95% CI 0.205-0.820, p=0.012) and absence of DM (OR 0.242, 95% CI 0.085-0.688, p=0.008)].
Figure 4. Kaplan-Meier curve demonstrating time to first AF recurrence in early and late follow up periods after catheter ablation according to alcohol consumption groups
In this study, we hypothesized that different alcohol consumption levels will have different effects on outcomes after PVI ablation for AF. However, the main finding of our study was that alcohol consumption was not associated with a higher likelihood of early or late AF recurrence in the first year after pulmonary vein isolation by catheter ablation. To the best of our knowledge, this is the first study exploring the impact of alcohol consumption on early and late catheter ablation outcomes of both paroxysmal and persistent atrial fibrillation.
Cutoffs for the 3 levels of alcohol consumption were chosen to better define terms such as none, rare, moderate and heavy that are used in the literature to describe alcohol consumption behavior. Moderate drinking was defined by National Institute on Alcohol Abuse and Alcoholism (NIAAA) as the following: for women, low-risk drinking is no more than 3 drinks on any single day and no more than 7 drinks per week. For men, it is defined as no more than 4 drinks on any single day and no more than 14 drinks per week. Applying this definition to our study would have required further data collection, which is not feasible in a retrospective study. Therefore, we defined moderate drinking, for both men and women, as 1-7 drinks per week to reflect a weekly habit of drinking but not more than daily.
Our main findings are in contrast to a recently published prospective study that shows a significantly lower success rate after catheter ablation in patients with alcohol use, when compared to the abstainer group, and a possible detrimental dose-dependent effect of alcohol consumption on catheter ablation outcomes of PAF.[10] In their 122-patient study, Qiao et al had a predominantly male population, and the authors used the definition of the NIAAA for daily alcohol consumption levels in their study. This is an important distinction since those patients with rare alcohol use, who would have been classified in the none-rare group according to our definition, were actually included under the moderate alcohol group. Despite this, the size of their moderate alcohol group was smaller (10.6% vs. 32.7%), compared to our study. In addition, the size of their heavy alcohol group was larger (32.0% vs. 8.8%).
Alcohol Consumption and Risk of Atrial Fibrillation
There is a considerable volume of evidence today to support the observations linking alcohol consumption patterns and levels to cardiovascular effects, including higher incidence of atrial fibrillation. Alcohol-induced cardiac dysrhythmias are supported by several studies demonstrating various electrophysiologic alterations of the cardiac substrate. Since its description in 1978, the “holiday heart” has shed light on the association of alcohol and cardiac dysrhythmias, specifically atrial fibrillation.[5] Although the pathogenesis of (acute) alcohol-induced dysrhythmias remains poorly delineated, direct and indirect effects on heart rhythm disturbances are implicated.[11]-[16]
Chronic alcohol drinking, especially, has been associated with increased risk of AF even with a lack of alcoholic cardiomyopathy.[17], [18] Several pathologic mechanisms and electrophysiologic changes of the underlying cardiac substrate have been described.[19]-[25] Furthermore, chronic alcohol can be associated with other traditional risk factors, such as hypertension, and promote atrial fibrosis and subsequently AF.[26]
Conflicting Evidence Regarding Alcohol Consumption and Atrial Fibrillation
Several studies have shown a deleterious relationship between the increased daily amount of consumed alcohol and the incidence of AF.[27]-[29] Although there is a significant amount of research showing the relationship of binge alcohol drinking and holiday heart syndrome, the relationship for a full range of alcohol consumption with the risk of AF is less certain.[30],[31] A large number of studies have reported that ethanol intake, of various amounts, was not associated with occurrences of AF.[11], [17], [32] In addition, a recent analysis from the Framingham study did not observe a link between alcohol consumption and AF.[33]
The prevalence of moderate-heavy alcohol drinking in this study population is high (41.6%). The lack of association that we have found between alcohol consumption groups and AF recurrence after ablation adds further understanding to this complex relationship between alcohol consumption and incidence of AF. Nevertheless, the influence of alcohol on cardiovascular health, and specifically AF, remains an area of active debate and research.
Catheter Ablation of Atrial Fibrillation in Alcohol Consuming Patients
With increasing advanced therapies for AF, namely catheter ablation, assessing ablation success rates after these procedures is critical for patients, institutions and physicians. Though the outcomes of these procedures are becoming more promising, the success rates are still below what medical providers and patients hope them to be. Several factors can influence procedural success rates including, but not limited to, patient selection, operator-dependent and procedure-related factors, current technology, ablation lesion durability and the underlying AF pathophysiology. As several risk factors have been linked to the incidence of atrial fibrillation, risk factor modification was shown to significantly help manage AF and reduce its recurrence, with or without catheter ablation. Risk reduction after catheter ablation may potentially boost the chances of AF-free survival and greatly reduce the costs associated with recurrences after these procedures. However, from our study, it does not appear that alcohol consumption is a major risk factor after ablation that can be altered to reduce recurrences.
This is an observational retrospective study that investigates the relationship of alcohol consumption behavior reported at the time of the ablation procedure with early and late recurrence following the ablation procedure. Alcohol consumption in the follow up period was not tracked. Hence, we are assuming that the patients’ alcohol consumption patterns remain the same in the follow up period after their ablation. The number of patients in the heavy alcohol group is relatively smaller, and thus the sample size may not have sufficient power to detect differences. Under reporting or recall biases during the alcohol use survey are also possible and are limitations of survey and retrospective studies. In addition, some patients may have a subtype of atrial fibrillation that is more alcohol sensitive. However, the retrospective nature of this study was not able to better define this population in our cohort and may be an area of further research in terms of their outcomes and response to ablation.
Alcohol use is common among patients undergoing catheter ablation of AF. Contrary to known associations of alcohol consumption and incidence of AF, different levels of alcohol consumption were not associated with significantly different rates of AF recurrence after catheter ablation in our cohort of patients undergoing pulmonary vein isolation. Further research is needed to study the effects of alcohol consumption, if any, on outcomes following catheter ablation for atrial fibrillation.
Study data were collected and managed using REDCap electronic data capture tools hosted at the University of Colorado.[34] REDCap (Research Electronic Data Capture) is a secure, web-based application designed to support data capture for research studies, providing: 1) an intuitive interface for validated data entry; 2) audit trails for tracking data manipulation and export procedures; 3) automated export procedures for seamless data downloads to common statistical packages; and 4) procedures for importing data from external sources.
Drs. Sauer and Nguyen receive significant research grants from Biosense Webster and CardioNXT and educational grants from St Jude Medical, Boston Scientific, and Medtronic. Drs. Sauer and Nguyen have a provisional patent on partially insulated focused catheter ablation. Drs. Nguyen and Sauer have non-public equity interests/stock options in CardioNXT.This publication was supported by NIH/NCRR Colorado CTSI Grant Number UL1 RR025780. Its contents are the authors’ sole responsibility and do not necessarily represent official NIH views.