Post-Ictal Transient Atrial Fibrillation As A Rare Manifestation Of Grand Mal Seizure
Gulshan Man Singh Dangol1,2, David A Hoffman1,2
1Northeast Ohio Medical University. Department of Medicine. Rootstown, Ohio.2St. Elizabeth Health Center, Department of Internal Medicine, Youngstown, Ohio.
Atrial fibrillation (AF) most frequently occurs as a consequence of multiple etiologies including valvular disease, coronary artery disease, hyperthyroidism, alcohol ingestion, and pulmonary embolism. However, on rare occasion transient AF may be a result of generalized tonic-clonic seizures (GTCS). A 33-year-old-man presented to the emergency department following GTCS in AF with rapid ventricular response. He had no previous documented history. Diagnostic evaluation including electrolytes, thyroid function, cardiac enzymes, serum and urine drug screen, and two-dimensional echocardiogram were unremarkable. Diltiazem was initiated for rate control with spontaneous conversion to sinus rhythm with no recurrence. AF post-seizure is a rare phenomenon but should be considered in epileptic patients. Anticoagulation must be considered in AF due to the risk of cardioembolic stroke but should be weighed against the potential risk of head injury and subsequent intracranial bleed in patients with grand mal seizures.
Key Words : Atrial fibrillation, Seizures, Anticoagulation.
Correspondence to: GulshanDangol, MD St. Elizabeth Health Center Department of Internal Medicine 1044 Belmont Avenue Youngstown, Ohio 44501 Email: gulshanms@gmail.com Phone: 202-758-7742 Fax: 330-480-3777
A wide range of autonomic imbalances may result following seizures and can cause various cardiac arrhythmias and repolarization abnormalities.[1] Increased vagal discharges may cause bradycardia and asystole, while increased sympathetic activity results in sinus tachyarrhythmias. Dangerous neurogenic cardiac arrhythmias, such as Atrial Fibrillation (AF) during the peri-ictal period are rare but should be considered in an epileptic patient. We present a case of atrial fibrillation following a generalized tonic-clonic seizure with spontaneous conversion to sinus rhythm with no recurrences.
A 33-year-old obese man (body mass index: 46.18 kg/m2) with a history of depression, and epilepsy maintained on lacosamide (200 mg twice daily) and carbamazepine (200 mg twice daily)presented initially to the emergency department (ED) following a complex partial seizure. He was a former smoker and had never used any alcohol or recreational drugs. On arrival to the ED, he was awake and alert.His heart rate (HR) was 74 beats/min, blood pressure (BP) 119/67 mm Hg, temperature (Temp) 98.1°F and respiratory rate (RR) 18 breaths/min. His oxygen saturation (SpO2) on room air was 99%. His serum electrolytes were within normal ranges. A 12 lead electrocardiogram (ECG) revealed normal sinus rhythm [Figure 1]. No further investigations were obtained,and he was subsequently discharged home.
Figure 1. ECG of the patient during initial presentation demonstrating normal sinus rhythm.

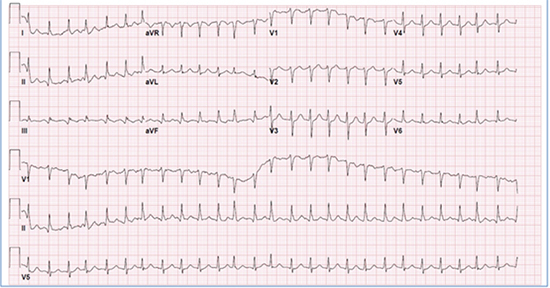
Approximately three hours post-discharge, he was transported back to the ED withwitnessed tonic-clonic seizureaccompanied by fecal incontinence. On presentation, he was in a postictal state. A computed tomography scan of the head was obtained and did not reveal any pathological abnormalities.His BP was 107/59 mm Hg, temperature was 98.4°F, RR 16 breaths/min, and SpO2 on room air was 93%. His pulse was irregularly irregular, and telemetry demonstrated AF with rapid ventricular response.A 12 lead ECG showed AF with a ventricular rate of 160 beats/min [Figure 2].Laboratory values revealed electrolytes within normal ranges; thyroid stimulating hormone (TSH) of 1.14 IU/ml (normal range: 0.27–4.2 IU/ml); negative urine drug screen; and negative cardiac enzymes. Two-dimensional echocardiogram demonstrated left ventricular ejection fraction of 55% with no evidence of cardiac chamber dilatation, pulmonary hypertension, or valvular abnormalities.
Figure 2. ECG of the patient during second presentation demonstrating atrial fibrillation with rapid ventricular response.
A bolus dose of diltiazem (10 mg) followed by a diltiazem drip at a rate of 5 mg/hrwas initiated for rate control, andapproximately 3 hours following the presentation,the patient’s rhythm spontaneously converted to sinus rhythm. His seizures remained controlled withanti-epileptics, and after monitoring for 48 hours, he was discharged home without anticoagulation based on a CHA2DS2VASc score of zero. Since the time of discharge, he has not required any admissions for either seizures or episodes of AF.
The most common peri-ictal arrhythmia is sinus tachycardia, which accompanies 90% of all seizures.Bradycardia and asystole are uncommon and occur in only about 0.5% of seizures.[1]Peri-ictal AF is a rare phenomenon, with only 14 cases reported in the literature to date [Table 1].[2]-[9] Most of the casesin the series were 20 - 40 years of age;the majority were males. Nearly all of the patients suffered GTCS;complex partial seizure was seen in one patient, and seizure type was not reported in one. In most cases, AF was transient with spontaneous conversion to sinus rhythm.One case received digoxin and verapamil for rate control. In two of the cases (case 7 and 9 in the table series), propafenone and amiodarone were used respectively for rhythm control after the second episode of peri-ictal AF. In these two cases, no inter-ictal arrhythmias were reported. Anticoagulation for stroke prevention was not reported in any of the cases and thus CHA2DS2VASc scores were not obtained.
Table 1. Literature review of the cases of seizure-induced atrial fibrillation.
| Case |
Year |
Age/Sex |
Type of seizure |
Atrial fibrillation duration |
Recurrence of atrial fibrillation |
Treatment given |
References |
| 1 |
1998 |
74/F |
GTCS |
2 hours |
Not reported |
spontaneous conversion |
Tigaran et al[2] |
| 2 |
1998 |
47/M |
GTCS |
Not reported |
Not reported |
IV digoxin and verapamil, spontaneous conversion |
Tigaran et al[2] |
| 3 |
2000 |
Not reported |
GTCS |
Not reported |
Not reported |
Spontaneous conversion |
Nei et al[3] |
| 4 |
2004 |
Not reported |
GTCS |
>110 sec |
Not reported |
Spontaneous conversion |
Nei et al[4] |
| 5 |
2004 |
Not reported |
Complex partial seizure |
55 sec |
Not reported |
Spontaneous conversion |
Nei et al[4] |
| 6 |
2006 |
Not reported |
Not reported |
Not reported |
Not reported |
Not reported |
Britton et al[5] |
| 7 |
2012 |
45/M |
GTCS |
90 min |
Yes, 1 month later following GTCS |
Propafenone (70 mg in 100 cm3 of saline infusion |
Vedovello et al[6] |
| 8 |
2012 |
41/ M |
GTCS |
2 hours |
None |
spontaneous conversion |
Herskovitz et al[7] |
| 9 |
2012 |
37/M |
Focal seizure with secondary generalization |
2 hours |
Yes, same hospitalization following seizure |
Amiodarone after 2nd AF following a seizure. Patient discontinued medication. |
Herskovitz et al[7] |
| 10 |
2012 |
24/M |
GTCS |
<6 hours |
No |
spontaneous conversion |
Herskovitz et al[7] |
| 11 |
2012 |
21/M |
GTCS |
<6 hours |
No |
spontaneous conversion |
Herskovitz et al[7] |
| 12 |
2012 |
23/M |
GTCS |
~ 25 hours |
Not reported |
spontaneous conversion |
Surges et al[8] |
| 13 |
2012 |
25/M |
GTCS |
>3 hours |
Not reported |
spontaneous conversion |
Surges et al[8] |
| 14 |
2014 |
18/M |
GTCS |
~6 hours |
No |
Synchronized cardioversion |
Singh et al[9] |
| 15 |
2017 |
33/M |
GTCS |
~3 hours |
No |
Diltiazem, spontaneous cardioversion |
Dangol et al(Current case) |
Atrial Fibrillation can be clinically silent in one-third of the cases, and asymptomatic AF preceding the seizure is always a possibility. However, our patient was in sinus rhythm on initial presentation and also denied previous history of AF. Triggering factors, such as valvular disorders, hyperthyroidism, drug or alcohol intoxication, and other medical conditions known to cause AF were absent in our patient. The only identifiable risk was lacosamide. Even though the incidence of lacosamide triggered AF is < 0.5% (typically with a dose of 600mg per day),[10] our patient was not taking the prescribed lacosamide(200 mg twice daily). Thus, given the absence of obvious cardiac diseases and medical conditions associated with AF, and the association of massive catecholamine release following GTCS,[11] seizure-induced AF is a strong possibility in this case. Moreover, the observation of transient AF following GTCS in ten of the fourteen cases (in the table series) supports the pathophysiological relationship between AF and GTCS.
AF is a prothrombotic state and tends to increase the risk of cardioembolic stroke in susceptible individuals. Given the risk for stroke and the potential impairment of cardiac function, AF demands immediate treatment. In hemodynamically unstable patients, synchronized cardioversion is the treatment of choice. Stable patients are managed medically with rate andrhythm control.Anticoagulation for stroke prevention is based on the individual’s CHA2DS2VASc score. The American Heart Association/American College of Cardiology/European Society of Cardiology (AHA/ACC/ESC) recommends oral anticoagulation, either with a vitamin K antagonist (warfarin, to an international normalized ratio (INR) of 2-3) or any of the novel oral anticoagulants (e.g. apixaban, rivaroxaban or dabigatran), in individuals with a CHA2DS2VASc score of ≥ 2. However, this score is weighed against the risk of bleeding.In patients with a high HASBLED score ≥ 3, caution should be applied while prescribing anticoagulation.[12],[13]
This prompts the question of whetherindividuals with seizure-induced AFand aCHA2DS2VASc score of ≥2 should be chronically anticoagulated.It should be emphasized that peri-ictal AF is typically transient, and given the potential risk for fall with subsequent head injury and intracranial hemorrhage, anticoagulation in an epileptic patient should be used with caution. In our opinion, the best approach to such rhythm disturbance is adequate treatment and control of seizures. In cases of recurrent seizures secondary to drug refractoriness or medication noncompliance, the long-term use of appropriate antiarrhythmics to prevent the development of AF may be considered.
AF is a rare consequence of GTCS and is generally transient. The best approach to the management of peri-ictal AF is prevention with adequate seizure control.Given the transient nature of peri-ictal AF and the potential for head injury with subsequent intracranial bleed, anticoagulation for stroke prevention should be cautiously used. Long term use of appropriate antiarrhythmicsmay be an alternative in refractory cases in order to remediate the long term useof anticoagulation.
thank Dr. Patrick A. Litam, MD for editorial assistance.