Efficacy of Catheter Ablation and Concomitant Antiarrhythmic Drugs on the Reduction of the Arrhythmia Burden in Patients with Long-Standing Persistent Atrial Fibrillation
Atsuhiko Yagishita1, Yasuteru Yamauchi1, Hironori Sato1, Shu Yamashita1, Tatsuhiko Hirao1, Takamichi Miyamoto1, Kenzo Hirao2
1Department of Cardiology, Musashino Red Cross Hospital, Tokyo.2Heart Rhythm Center, Tokyo Medical and Dental University, Tokyo, Japan.
Little is known about the long-term outcome and recurrent form recurrence after catheter ablation of atrial fibrillation (AF) in patients with long-standing persistent AF.
Two hundred thirty-six patients with persistent AF (193 men; age, 61.5±10.7 years) were enrolled, and were classified according to the duration of AF: AF duration of <1 year (group A, n=99), between 1 to 5 years (group B, n=101), and ≥5 years (group C, n=36). The long-term recurrence rate and recurrent form were compared among the groups.
During a median follow-up of 3.7 years, the recurrence rate was significantly worse in group C after the index and multiple procedures (Log-Rank, both for a P<0.001 in comparison to group A). In the multivariate analysis, the duration was an independent predictor of an arrhythmia recurrence (HR, 1.206; 95%CI, 1.053 to 1.381; P=0.007). Recurrent AF became permanent in 8 patients (3.4%), which was not associated with a difference in the groups (Log-rank, P=0.055), while antiarrhythmic drugs (AADs) were continued in 70% of the recurrent patients.
Despite a high AF recurrence rate in the patients with an AF duration of ≥5 years, the majority of the patients with recurrence who continued on AADs had a paroxysmal form of AF. Catheter ablation and concomitant AADs may be effective in reducing the AF burden in such patients with an advanced AF disease stage.
Key Words : Persistent atrial fibrillation, Catheter ablation, Antiarrhythmic drugs.
Correspondence to: Atsuhiko Yagishita, MD. Department of Cardiology, Musashino Red Cross Hospital, Tokyo, Kyonancho 1-26-1, Musashinoshi, Tokyo, Japan.
Catheter ablation is an established therapy for atrial fibrillation (AF). Pulmonary vein isolation (PVI) is the cornerstone of the procedure, particularly in patients with paroxysmal PAF (PAF) [1]. However, the efficacy of PVI alone for the treatment of persistent AF is limited, which has resulted in an increasing demand for additional modification of the atrial substrate, such as targeting complex fractionated atrial electrograms (CFAEs) [2], linear lesions [3], [4], and their combinations. Despite these continued efforts to identify additive strategies, the recurrence rate after catheter ablation in patients with persistent AF has been high compared to that with PAF, and particularly in patients with an AF duration of >2 years [5]. Although it is suggested that the increase in the AF duration is associated with a higher risk of recurrence after ablation, little is known about the recurrence of AF in long standing AF patients post ablation treated with concomitant antiarrhythmic medications. In this study, we sought to determine the long-term recurrence rate in long-standing persistent patients with an AF duration of ≥5 years.
The study was comprised of 236 consecutive patients undergoing catheter ablation of persistent AF at Musashino Red Cross Hospital, Tokyo. There were 193 men, with 137 having long-standing AF, and their mean age was 62±11 years (range 25-82, [Table 1]). This study was approved by the Institutional Review Board, and written informed consent was obtained before the procedure.
Table 1. Clinical characteristics
|
Group A:AF duration < 1 year (n=99) |
Group B:AF duration 1-5years (n=101) |
Group C:AF duration ≥ 5 years (n=36) |
P Value |
| Male (%) |
78 (78.8) |
87 (86.1) |
28 (77.8) |
0.321 |
| Age |
63.7 ± 9.7 |
58.8 ± 11.7 |
62.6 ± 9.2 |
0.016 |
| AF history (years) |
2.8 ± 4.4 |
3.0 ± 2.1 |
8.1 ± 2.8 |
<0.001 |
| Duration of persistence (years) |
0.5 ± 0.2 |
2.2 ± 1.1 |
7.8 ± 2.7 |
<0.001 |
| Hypertension (%) |
27 (27.3) |
23 (22.8) |
10 (27.8) |
0.720 |
| Diabetes (%) |
3 (3.0) |
4 (4.0) |
4 (11.1) |
0.130 |
| Prior stroke (%) |
7 (7.1) |
4(4.0) |
4 (11.1) |
0.297 |
| Congestive heart failure (%) |
5 (5.1) |
15 (14.9) |
2 (5.6) |
0.041 |
| CHADS2-Score |
0.5 ± 0.7 |
0.5 ± 0.8 |
0.7 ± 1.0 |
0.503 |
| Cardiomyopathy |
|
|
|
|
| Ischemic (%) |
4 (4.0) |
3 (3.0) |
1 (2.8) |
0.894 |
| Dilated (%) |
0 (0) |
4 (4.0) |
0 (0) |
0.066 |
| Hypertrophic (%) |
3 (3.0) |
1 (1.0) |
2 (5.6) |
0.302 |
| Valvular (%) |
3 (3.0) |
3 (3.0) |
2 (5.6) |
0.738 |
| eGFR |
65.5 ± 17.1 |
70.1 ± 14.5 |
65.0 ± 17.9 |
0.134 |
| BNP (pg/ml) |
121.4 ± 123.8 |
110.1 ± 132.9 |
120.5 ± 200.9 |
0.610 |
| LVEF (%) |
64.1 ± 9.2 |
62.8 ± 13.0 |
68.0 ± 7.8 |
0.121 |
| LVDd (mm) |
47 ± 7 |
49 ± 7 |
48 ± 5 |
0.109 |
| Left Atrial Diameter (mm) |
40 ± 7 |
41 ± 5 |
44 ± 6 |
0.024 |
AF, atrial fibrillation; eGFR, estimated glomerular filtration rate; BNP, brain natriuric peptide; LVEF, left ventricular ejection fraction; LVDd, left ventricular diastolic diameter. Values are given as the mean ± SD.
Persistent AF was defined as continuous AF that was sustained beyond seven days [1], and longstanding persistent AF as continuous AF of greater than a 12 month duration. The duration of persistent AF was determined from the last time when sinus rhythm (SR) was recorded on the electrocardiogram (ECG) to the time during the index procedure. If the AF was found incidentally through a routine health check, the onset was retraced when SR was lastly confirmed on the ECG. Patients were classified by the duration of the AF into three groups: those with an AF duration of less than 1 year (group A, n= 99), those with an AF duration between 1 to 5 years (group B, n=101), and those with an AF duration of more than 5 years (group C, n=36). The long-term outcome after the index and multiple procedures was compared between the three groups. We also investigated the mode of recurrent arrhythmias, namely, whether the recurrent AF became permanent or paroxysmal on antiarrhythmic drugs (AADs). Recurrent AF was defined as permanent if the AF persisted despite multiple procedures and AADs.
All AADs were discontinued for at least 5 half-lives before the ablation. All patients were anticoagulated with warfarin for at least a month before the procedure (target INR 2–3), and the therapeutic anticoagulation was maintained with intravenous heparin after discontinuing warfarin one day before the procedure. Transesophageal echocardiography was performed within 24 hours prior to the procedure. The electrophysiological study was performed under mild sedation with dexmedetomidine hydrochloride. Four vascular sheaths (i.e., one 5-F, and three 8-F) were inserted into the right femoral vein under lidocaine local anesthesia. A 7-F, 20-pole or 14-pole two-site mapping catheter (Irvine Biomedical Inc, Irvin, CA) was inserted through the right jugular vein and positioned in the coronary sinus (CS) for pacing, recording, and internal cardioversion. A 50 IU/kg body weight of heparin was administered after the insertion of the vascular sheaths. A transseptal puncture was performed using a standard needle (Adult BRK 71 cm, St. Jude Medical, Minneapolis, Minnesota). A 50 IU/kg body weight of heparin was re-administered after the transseptal puncture, and heparinized saline was additionally infused to maintain the activated clotting time at 300–350 seconds. After the transseptal puncture, two long sheaths (SL0, SJM, Minneapolis, MN) were introduced into both superior PVs. The pulmonary vein (PV) electrograms were monitored with a decapolar circumferential mapping catheter (Lasso, Biosense Webster, Diamond Bar, CA, USA). A 3.5-mm irrigation catheter (ThermoCool, Biosense Webster, Diamond Bar, CA, USA) was used, under the guidance of a 3-dimensional (3D) mapping system (CARTO3, Biosense Webster). An esophageal temperature probe was advanced into the esophagus, and later adjusted to its closest proximity to the ablation sites along the left atrial (LA) posterior wall. The power was limited to 20-25W on the LA posterior wall, and the RF application was truncated when the esophageal temperature rose up to 41 degrees Celsius. Before the initiation of the PV isolation (PVI), up to three internal cardioversions (10J, 20J, and 30J) were administered. If AF persisted after the cardioversion, a PVI was performed during AF. A decapolar, circular catheter (Lasso, Biosense Webster) was used for the bipolar EGM recording at the PV antrum filtered at 30 to 500 Hz and displayed on a commercially available electrophysiological recording system (GE, Houston, TX), and to confirm the PVI, which was defined as PV bi-directional conduction block. After the PVI, internal cardioversion was re-administered when the AF continued. After restoring SR, if the AF was reinitiated, non-PV foci were pursued. Once AF converted into an organized AT, activation mapping on the 3D mapping system, entrainment, and ablation were performed. Roof and floor lines connecting the bottom floor region of the bilateral lower PVs were added in the presence of an LA posterior wall trigger or roof dependent macro-reentrant AT, and a mitral isthmus line was created in the case of macroreentrant perimitral flutter. Cavo-tricuspid isthmus (CTI) ablation was performed with an endpoint of bidirectional conduction block in all patients. Isolation of the superior vena cava (SVC) was only performed when SVC triggered AF was present or the sleeve length of the superior vena cava was ≥ 30 mm [6].
In the repeat procedure, after the isolation of the reconnected PVs, AF or AT was induced by atrial burst pacing in case of SR, using 10-second burst pacing intervals at an output of 10 V starting at a cycle length of 250 ms decreasing down to refractoriness from the right appendage. During native or induced AT, activation mapping on the 3D mapping system, entrainment mapping, and ablation were performed. During native or induced AF, substrate modification with a linear defragmentation method was performed sequentially in a predetermined order [7]. Linear point-by-point lesions guided by the 3D mapping system in the regions were performed in the following order: LA roof, floor lines connecting both PVs, LA septum from the high septum to fossa ovalis, inferior LA along the CS, base of the LA appendage, LA anterior wall, right atrial (RA) septum from the SVC-RA junction to the inferior vena cava (intercaval line), crista terminalis, and base of the RA appendage. The endpoint of the substrate ablation was AF termination. If the AF continued after all the steps, internal cardioversion was performed. If the AF was converted to AT, it was mapped and ablated. If the AF was not induced after the isolation of the reconnected PV, a substrate modification was not performed. An SVC isolation was performed in the patients who underwent it during the index procedure or SVC triggered AF was found during the repeat procedures.
Follow-up after the catheter ablation
The oral anticoagulants were resumed immediately after the procedure, targeting an international normalized ratio of 2 to 3. Patients underwent continuous ECG monitoring as inpatients for two days after the procedure. The first outpatient clinic visit was 3 weeks after the procedure. In all patients, the AADs were continued for at least the first 3 months of the blanking period. An attempt was made to discontinue the AADs 3-6 months following the ablation, except for in the patients with AF or AT recurrences, defined by an episode lasting ≥30 seconds. A repeat procedure was offered if recurrences occurred 3 months following the index procedure. The AADs were continued or resumed in recurrent patients who refused a repeat procedure or recurred after multiple procedures. Oral anticoagulants were essentially discontinued based on the patient’s CHADS2 score [1]. The follow-up visits consisted of a clinical interview, ECG, and 24-hour Holter monitoring every 3, 6, and 12 months. After 12 months, the patients were seen thereafter every 6-12 months at our center. Either the last follow-up entry data set based on the last office visit or a telephone questionnaire were used to assess the long-term freedom from recurrence.
The categorical variables are expressed as absolute and relative frequencies. Continuous variables are expressed as the mean ± SD or median and interquartile range (IQR) (25th, 75th percentile) as appropriate. A comparison between the groups was performed with a Student's t-test or Wilcoxon rank-sum test, as appropriate. For categorical data, a Chi-squared or Fisher’s exact test was applied, as appropriate. The estimated freedom from arrhythmia recurrence after catheter ablation was calculated using a Kaplan-Meier analysis, and long-lank statistics were used for the overall comparisons among the three groups. In addition, a post hoc pairwise analysis was performed with the use of the Bonferroni method. To clarify the clinical predictors of the outcomes, a univariate Cox proportional analysis was first performed. Sequentially, all variables significant in the univariate analysis were included in the multivariate analysis, and the hazard ratios (HRs) and 95% confidence intervals (CI) were calculated. A 2-tailed p value of <0.05 was considered to indicate statistical significance. All statistical analyses were performed with SPSS statistics software (IBM Corp, Armonk, NY).
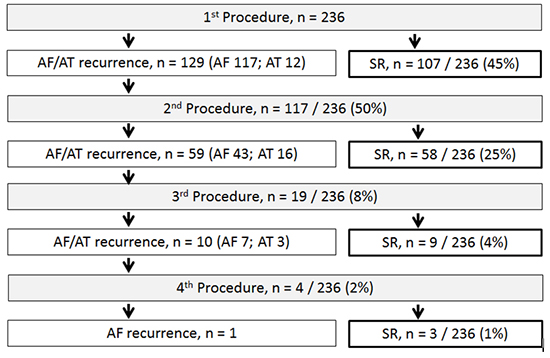
The baseline demographics, clinical characteristics, echocardiographic variables, and biomarkers are shown in Table 1. The age was the highest in the group A and lowest in the group B patients. The duration of AF in the three groups was 2.8±4.4 years, 3.0±2.1, and 8.1±2.8, respectively (P < 0.001). The duration of the persistence was 0.5±0.2 years, 2.2±1.1, and 7.8±2.7, respectively (P < 0.001). Three hundred seventy-eight procedures, that is 1.6 procedures per patient, were performed: 119 had 1, 96 had 2, 17 had 3, and 4 had 4 procedures. [Figure 1] provides an overview of the procedures. After the index procedure, SR was maintained in 107 of 236 (45%) patients. A roof and bottom floor line, mitral isthmus line, and SVC isolation were performed in 38 (16%), 6 (3%), and 22 patients (9%), respectively. Among the 129 patients with a recurrence, 2nd procedures were performed in 117 patients, including 12 (5%) who had only an AT recurrence. During the 2nd procedure, after isolating the reconnected PV, no further AF or AT was induced in 34 patients (29% of the 117 patients), and only AT was induced in four patients (3%). Substrate modification during AF was performed in the remaining 83 patients, and in 11 of whom AF was terminated by ablation (13%). After the 2nd procedures, 59 of 117 patients (50%) presented with recurrences, 16 (14%) of whom had AT. Of the 59 patients, 19 underwent 3rd procedures, and 10 had recurrences, including three with AT recurrences. Among the 10 patients, four patients underwent a 4th procedure, and one patient had an AF recurrence. During a median follow-up of 3.3 (IQR: 1.9, 5.3) years, 177 of 236 (75%) patients remained in SR. AADs were continued in 112 of 236 patients: 72 of 177 patients (41%) without recurrences, and 40 of 59 (70%) with recurrences. Bepridil was used in 77 (mean dose 99 ± 39mg), amiodarone in 11 (mean dose 113 ± 39mg), and class I AADs in 24. Oral anticoagulants were discontinued in 85 patients (48%) without recurrences, and in 19 (32%) with recurrences. Of the 19 patients with recurrences, 18 discontinued their oral anticoagulants due to a CHADS2 score of less than 1. The remaining patient with a CHADS2 score of 2 preferred to discontinue oral anticoagulants.
Figure 1. Flowchart of the outcome after the procedures in the enrolled patients.
Clinical outcome after the index procedure
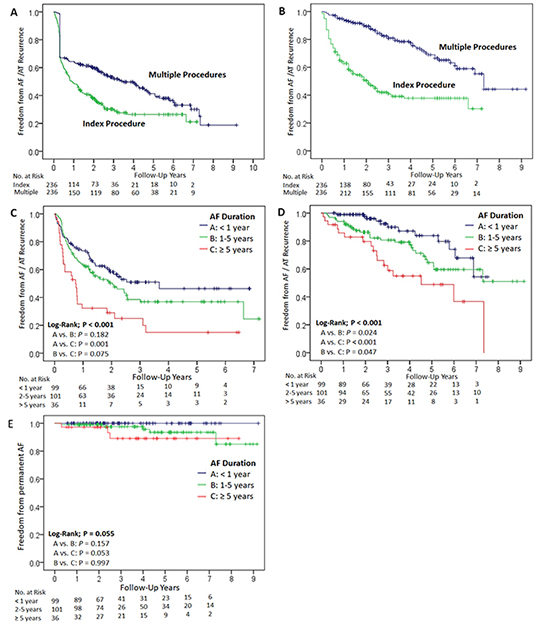
In the Kaplan-Meier analysis, the estimated probability of an atrial arrhythmia-free survival without AADs at 1, 2 and 5 years was 49%, 38%, and 26%, respectively [Figure 2]. With AADs, the estimated probability of an atrial arrhythmia-free survival at 1, 2 and 5 years was 64% (group A 73%, group B 63%, and group C 32%), 51% (group A 58%, group B 50%, and group C 29%), and 37% (group A 46%, group B 37%, and group C 15%), respectively [Figure 2]. There was a significant difference between the groups in terms of the recurrence rate after the index procedure (P<0.001, [Figure 2]). In the three subgroups, there was no difference between Group A and Group B (P=0.182), whereas Group C had a significantly higher recurrence rate than Group A (P<0.001).
Figure 2. Kaplan-Meier event-free survival curve of AF recurrence. The plus sign indicates censored. The numbers at the bottom indicate the patients at risk. AF; atrial fibrillation.
Clinical outcome after multiple procedures
In the Kaplan-Meier analysis, the estimated probability of an atrial arrhythmia-free survival without AADs at 1, 2 and 5 years was 64%, 60%, and 41%, respectively [Figure 2]. With AADs, the estimated probability of an atrial arrhythmia-free survival at 1, 2 and 5 years was 95% (group A 99%, group B 94%, and group C 86%), 90% (group A 98%, group B 86%, and group C 80%), and 69% (group A 84%, group B 65%, and group C 55%), respectively [Figure 2]. There was a significant difference between the groups in terms of the recurrence rate after multiple procedures (P=0.001: P=0.024 for Group A vs. B, P<0.001 for Group A vs. C, and P=0.047 for Group B vs. C).
Factors for arrhythmia recurrence
[Table 2] summarizes the clinical factors associated with the recurrence after multiple procedures. In the univariate analysis, the duration from the time when the patient was initially diagnosed with paroxysmal or persistent AF to the time during the index procedure (HR 1.112; CI, 1.033 to 1.197; P = 0.005), and the duration of persistent AF (HR 1.246; CI, 1.120-1.385; P < 0.005) were predictors of arrhythmia recurrence. In the multivariate analysis, the duration of persistent AF was an independent predictor of arrhythmia recurrence (HR, 1.206; 95% CI, 1.053 to 1.381; P = 0.007).
Table 2. Factors for arrhythmia recurrence after multiple procedures
|
|
Univariable |
|
|
|
Multivariable |
|
|
- |
- |
- |
|
- |
- |
- |
|
HR |
P |
95% CI |
|
HR |
P |
95% CI |
| Male gender |
0.983 |
0.207 |
0.956-1.010 |
|
|
|
|
| Age |
1.251 |
0.586 |
0.560-2.794 |
|
|
|
|
| Duration of AF |
1.112 |
0.005 |
1.033-1.197 |
|
1.054 |
0.299 |
0.954-1.164 |
| Duration of AF persistence |
1.246 |
<0.001 |
1.120-1.385 |
|
1.206 |
0.007 |
1.053-1.381 |
| Hypertension |
0.83 |
0.603 |
0.411-1.675 |
|
|
|
|
| Diabetes |
0.302 |
0.258 |
0.038-2.410 |
|
|
|
|
| Prior stroke |
0.21 |
0.136 |
0.27-1.637 |
|
|
|
|
| Congestive heart failure |
0.675 |
0.494 |
0.219-2.083 |
|
|
|
|
| Ischemic cardiomyopathy |
0.439 |
0.446 |
0.053-3.644 |
|
|
|
|
| Dilated cardiomyopathy |
3.218 |
0.248 |
0.44-23.383 |
|
|
|
|
| Hypertrophic cardiomyopathy |
3.259 |
0.155 |
0.639-16.620 |
|
|
|
|
| Valvular cardiomyopathy |
3.302 |
0.099 |
0.798-13.654 |
|
|
|
|
| eGFR |
1.006 |
0.546 |
0.987-1.025 |
|
|
|
|
| BNP |
0.999 |
0.668 |
0.997-1.002 |
|
|
|
|
| LVEF |
0.983 |
0.223 |
0.957-1.010 |
|
|
|
|
| LVDd |
1.044 |
0.058 |
0.999-1.090 |
|
|
|
|
| Left Atrial Diameter |
1.038 |
0.143 |
0.987-1.091 |
|
|
|
|
AF, atrial fibrillation; eGFR, estimated glomerular filtration rate; BNP, brain natriuric peptide; LVEF, left ventricular ejection fraction; LVDd, left ventricular diastolic diameter. Values are given as the mean ± SD.
Incidence of permanent AF after multiple procedures
A permanent form of AF recurrence occurred in eight of the 236 patients (3%), and the incidence rate was 9.29 cases per 1000 person-years. No patients with AF recurrence in group A had permanent AF. Five of the eight patients were from group B (5% of 101 patients), whereas the remaining three patients were from group C (8% of 36 patients). In the Kaplan-Meier analysis, there was no significant difference between the three groups in terms of the number of patients with permanent AF (P=0.055, [Figure 2]). There was also no significant difference in the incidence of permanent AF between each group (P=0.157 for Group A vs. B, P=0.053 for Group A vs. C, and P=0.997 for Group B vs. C).
Procedural adverse events
Among 378 procedures, there were four major complications (1%) in this study. Transient ischemic attacks occurred in two patients. One patient experienced one shortly after the catheter ablation, and the other experienced one 10 days after the catheter ablation. One patient developed pulmonary edema requiring mechanical ventilation, resulting in a prolonged hospitalization. Cardiac tamponade occurred in one patient. During the follow-up period, two patient died from non-cardiac causes (one patient died from pharyngeal cancer, and the other from pancreatic cancer), while no patients died from cardiac disease. No late thromboembolic events occurred more than one month after the catheter ablation, and no PV stenosis or atrioesophageal fistula occurred.
In this single center study, we found that the AF duration was significantly associated with arrhythmia recurrence after the catheter ablation. However, in conjunction with AADs, the incidence of permanent AF in patients with long-standing persistent AF was similarly low compared to that with an AF duration of <1 year.
Impact of catheter ablation and concomitant AADs on the downgrade of persistent AF
The impact of the AF duration on the arrhythmia recurrence, shown in this study, was in agreement with the previous study. Tilz et al. demonstrated that the arrhythmia-free survival was significantly higher among patients with an AF duration of <2 years than among those with an AF duration of >2 years [5]. In our study, patients with an AF duration of ≥5 years had the highest arrhythmia recurrence rate. However, the number of such patients with a persistent form of AF was fairly low (8%) after multiple procedures, while AADs were continued in 70% of the recurrent patients. In our study, the majority of the recurrent patients having been continued on bepridil, class IV AADs with a multiple ion-channel blocker, was highly effective for the maintenance of SR in patients with persistent AF, compared with amiodarone [8]-[10]. Kondo et al. demonstrated that bepridil reduced the recurrence of AF compared to amiodarone and class I AADs in patients who underwent catheter ablation of persistent AF [11], and a Japanese nationwide survey of catheter ablation of AF revealed that bepridil was most frequently prescribed (17.4%), specifically in patients with persistent AF [12], both of which were in agreement with our results. The annual incidence of permanent AF was 0.929% in this study, which was lower than that for PAF in those treated with bepridil (4%) alone [13], suggesting the additional effect of catheter ablation on bepridil. Importantly, the relatively low dose of bepridil (99±39mg) for AF recurrence was effective for avoiding a permanent form of AF recurrence, while minimizing the risk of QT prolongation associated with high doses of 200mg [14], [15]. Due to the concern over the impairment of the long-term atrial function after extensive substrate modification [16], extensive substrate modification was selectively performed in 35% of the patients (83 of 236 patients) . Considering the limited number of recurrent patients with permanent AF, our stratified approach for catheter ablation of persistent AF, in conjunction with low dose AADs, contributed to the downgrade of persistent AF, likely associated with the reduction in the AF burden.
In this study, the LA diameter differed among the groups, suggesting the correlation between the duration of AF and degree of atrial structural remodeling. The LA diameter has been shown to be a predictor of recurrence after catheter ablation [1], but it was not a predictor of recurrence in the univariate analysis in this study, possibly due to our limited sample size and patient selection bias that patients with severely dilated LA may have been excluded.
AF burden, thromboembolic events, and mortality
The AF burden is highly associated with the incidence of thromboembolic events, which are one of major adverse events associated with AF. Recent studies with a large number of AF patients has shown an increased risk of thromboembolic events in patients with persistent AF as compared to PAF [17]-[20]. Takabayashi et al. reported that the annual incidence of thromboembolic events was 1.4 and 3.0 per 100 person-years in patients without a progression from PAF to persistent AF, and those who progressed to persistent AF, respectively [19]. In the study of the ENGAGE AF-TIMI 48 trial, strokes/systemic embolic events were lower in those patients with PAF (1.49%/year), compared to that in persistent (1.83%/year) and permanent AF (1.95%/year). Furthermore, the all-cause mortality was lower in those with paroxysmal (3.0%/year) than in those with persistent (4.4%/year) and permanent AF (4.4%/year) [20]. Other reports also demonstrated a higher risk for all-cause mortality in patients with persistent AF than in those with PAF [17], [18]. Interestingly, the majority of deaths in a large anticoagulated AF population were not related to strokes [21]. Marijon et al. reported that cardiac deaths accounted for 37.4% of all deaths (persistent AF was 67.2% of 18113 patients), whereas stroke and hemorrhage related deaths represented 9.8%. Those results suggest that the increased mortality may not only be related to an increased risk of thromboembolic events, but to an impairment of the cardiac function associated with AF persistence in patients with a high AF burden. There have been previous studies demonstrating a low incidence of thromboembolic events after a successful catheter ablation of AF [22]-[24]. More recently, a propensity score-matched analysis revealed that the adjusted HRs for strokes and mortality were lower in patients undergoing catheter ablation than in those who did not [25]. Therefore, catheter ablation of AF may have an impact on the decreased risk of thromboembolic events and mortality, possibly due to the beneficial effect of the reduction in the AF burden.
First, our single center study had no control group without catheter ablation to compare the incidence of thromboembolic events and the cardiovascular mortality. Furthermore, there was a lack of evidence to support the favorable effect of downgrading AF on the clinical outcomes due to the relatively small number of patients. To address that issue, lager and randomized trials are required. Second, almost half of the patients continued on AADs after the procedures. Although there were no patients with adverse events from the AADs in this study, the long-term safety of AADs should be cautiously monitored in our patients. Third, in the absence of the continuous monitoring devices, it was likely that some of the patients we regarded as being free from AF had undetected arrhythmia recurrences. These false negatives, if detected, should have been included in the patients without arrhythmia recurrences. Nevertheless, the undetected PAF would not have affected the clinical relevance of our study. Fourth, the symptomatic severity was not systematically assessed during the enrollment of the patients. The efficacy of catheter ablation in patients with symptomatic persistent AF has been widely recognized, while it has not fully investigated in asymptomatic patients. Recently, we conducted a study to determine the efficacy of catheter ablation in patients with asymptomatic persistent AF, and found that maintenance of SR after catheter ablation was associated with an improvement in quality of life, exercise performance, and plasma B-type natriuretic peptide level [26]. Interestingly, there was a significant linear correlation of an increased duration of exercise and plasma BNP decrease. It is suggested that catheter ablation is effective for the maintenance of SR irrespective of symptoms in patients with persistent AF’.
The risk of AF recurrence increases as the duration of persistent AF increases. However, the majority of recurrent patients with long-standing persistent AF continued on AADs and continued to have a paroxysmal form of AF. In conjunction with AADs, catheter ablation may have an impact on the reduction in the AF burden in such patients with an advanced AF disease stage.