The Amplatzer Cardiac Plug (ACP) is the first Amplatzer device that was specially designed by Dr. Kurt Amplatz for percutaneous left atrial appendage occlusion (LAAO). [1] It acquired CE mark in December 2008. The Amplatzer Amulet is a second-generation LAAO device that became commercially available in Europe in January 2013 (AGA Medical, acquired by St Jude Medical, St Paul, MN, USA – now Abbott Vascular, Santa Clara, CA, USA). [2] Currently, the Amplatzer Amulet is one of the most widely used devices for LAAO worldwide but is not yet approved by the US Food and Drug Administration.
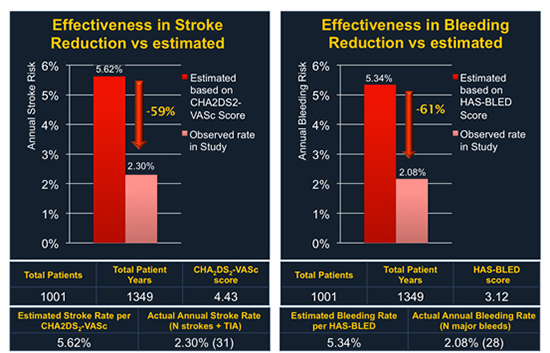
Most of the clinical data for ACP come from the ACP multicenter registry. [3] In this retrospective registry, prospectively collected data on 1047 consecutive patients from 22 centers were transferred in a common database and analyzed. Patients were treated between December 2008 and November 2013 so this analysis included the “learning curves” of all participating centers and operators. A total of 1001 patients who underwent LAAO with the ACP and had complete follow-up were included and were further analyzed for stroke and bleeding reduction by comparing their outcomes with the predicted risks by the CHA2DS2-VASc and HAS-BLED score, respectively. The most common indication for the procedure was previous bleeding (47%) followed by high bleeding risk (35%) and the avoidance of triple therapy for coronary artery disease and stenting (22%). The composite of previous bleeding (major or minor) and high bleeding risk was 73%. Therefore, this study was the first large “real world” report on LAAO in patients with contraindications to oral anticoagulation (OAC) therapy. The mean CHADS2 score was 2.8±1.3 and the mean CHA2DS2-VASc score was 4.5±1.6. The mean HAS-BLED score was 3.1±1.2. A score of ≥3 was present in 742 patients (72%). Procedural success was achieved in 1019/1047 patients (97.3%). A total of 52 periprocedural major adverse events (4.97%) were reported, including 8 procedure-related deaths, 9 strokes, and 13 cardiac tamponades. The average patient follow-up was 13 months, resulting in a total of 1,349 patient years. It should be noted that LAAO with the ACP does not require OAC after the procedure. According to the device manufacturer recommendations, patients were prescribed dual antiplatelet therapy (DAPT) for 1-6 months (average 3 months) and single or no antiplatelet therapy thereafter. The annual rate of systemic thromboembolism in the study (periprocedural and follow-up) was 2.3% (31/1,349 patient-years), which is a 59.1% risk reduction [Figure 1]. The annual rate of major bleeding (periprocedural and follow-up) was 2.1% (28/1,349 patient-years), which is a 61.0% risk reduction. On transesophageal echocardiography (TEE) follow up that was available in 632/1001 of successfully implanted patients, a significant (3-5mm) peri-device leak was found in 12 patients (1.9%) patients, whereas a thrombus related to the device was observed in 28/632 patients (4.4%). This study had a few limitations: it was not randomized without a control group, the TEE follow-up was not available for all patients and the study results were self-reported, without independent adjudication. However, a written summary was provided for all major adverse events.
Figure 1. Effectiveness of LAAO with the Amplatzer Cardiac Plug in reduction of thromboembolism and bleeding. Both periprocedural and follow-up events were included in the analysis.
In order to address the lack of independent TEE adjudication in the ACP registry, a relevant sub-study was performed by Saw et al., entitled “Incidence and clinical impact of device-associated thrombus and peri-device leak following left atrial appendage closure with the Amplatzer Cardiac Plug”. [4] A total of 344 follow up TEEs (performed after a median of 134 days), from 605 consecutive patients were submitted to a core laboratory and reviewed by 2 independent experts for peri-device leak, device-associated thrombus, device embolization, device migration, left atrial appendage thrombus, and left atrial thrombus. Device-associated thrombus was observed in 3.2% and significant peri-device leak in 1.2% of patients. Neither device-associated thrombus nor peri-device leak was associated with an increased risk for cardiovascular events. Independent predictors of device-associated thrombus were smoking (odds ratio: 5.79; p=0.017) and female sex (odds ratio: 4.22; p=0.027). It should be noted that device-associated thrombus is a complex matter and other factors could be independent predictors but were not included in this analysis.
Patients with renal insufficiency and AF are usually difficult to manage with OAC therapy. Moreover, these patients are commonly not well represented in large randomized clinical trials. Therefore, Kefer et al. conducted a sub-group analysis of patients with chronic kidney disease (CKD) within the ACP registry. [5] Patients with CKD had at higher risk for stroke and major bleeding compared to patients without CKD (p<0.001 for both). Kaplan–Meier analysis showed a lower overall survival (84 vs. 96% and 84 vs. 93% at 1 and 2 yrs. respectively; p < 0.001) among patients with an eGFR <30 ml/min/1.73 m2. However, procedural success was similarly high (97%) in all stages of CKD, with no significant differences in peri-procedural major adverse events (MAEs; 6.1 vs. 4.5%, p = 0.47). The overall stroke and bleeding reduction was similar among groups (60-62%).
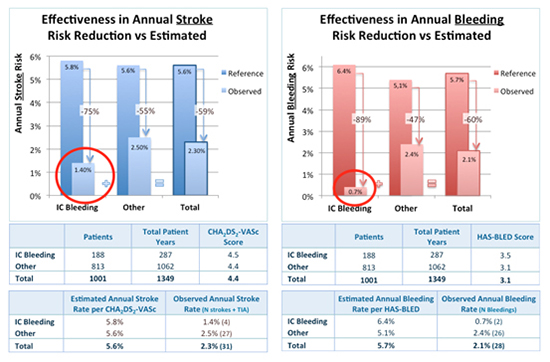
Another important group is patients with previous intracranial bleeding (ICB), who have often an absolute contraindication to OAC therapy. In the ACP registry, a total of 198 patients (18.9%) with previous ICB were identified and were compared with patients without previous ICB. [6] Patients were younger, more commonly male, and had a higher rate of previous stroke. The CHA2DS2-VASc score was similar between groups but the HAS-BLED score was higher in patients with previous ICB. There were no significant differences in peri-procedural MAEs. Patients with previous ICB were receiving more frequently aspirin and less frequently OAC at baseline. In addition, patients with previous ICB were more frequently on single aspirin therapy after LAAO (last follow-up visit: 74.5% vs. 61.3%; p < 0.001). At follow-up (average 1.3 years), the observed annual stroke/transient ischemic attack (TIA) rate (procedure and follow-up) for patients with previous ICB was 1.4% (75% relative risk reduction), [Figure 2]. The observed annual major bleeding rate (procedure and follow-up) for patients with previous ICB was 0.7% (89% relative risk reduction). This study concluded that in patients with previous ICB and AF, LAAO seemed to be a safe procedure and was associated with a significant reduction in stroke/TIA and a remarkably low frequency of major bleeding during follow-up. In a propensity score matched follow-up study from the Nordic countries, Nielsen-Kudsk et al. compared patients with AF and previous ICB who underwent LAAO using the Amplatzer Cardiac Plug or the Amplatzer Amulet to a matched group of 151 patients receiving standard medical therapy. [7] The primary endpoint was a composite of all-cause mortality, ischemic stroke and major bleeding. Patients with AF and a prior ICB treated with LAAO had a lower risk of the composite outcome as compared to patients treated with standard medical care (events/1000 years: 53.3 vs. 366.7; hazard ratio 0.16 [0.07-0.37]). The authors concluded that LAAO was suggested to be of major clinical benefit in AF patients having sustained an ICB. A relevant randomized clinical trial entitled STROKECLOSE (ClinicalTrials.gov Identifier: NCT02830152) started in May 3, 2017. The primary endpoint is a composite of stroke (ischemic or hemorrhagic), systemic embolism, life threatening or major bleeding and all-cause mortality, with a time frame up to 5 years after randomization. The study inclusion criteria are: a diagnosis of paroxysmal, persistent or long-standing NVAF with CHA2DS2VASc score >2, clinical and CT/MRI evidence of ICH within 6 months but not less than 4 weeks prior to enrollment, and age > 18 years. The study exclusion criteria are ICH secondary to vascular malformation or tumors, estimated life expectancy of < 1 year at eligibility assessment, modified Rankin Score > 3 at enrollment, contraindications to LAAO known at the time of enrollment, such as LAA thrombus or systemic infection, prior surgical LAA excision, and planned combined interventional procedures at the time of enrollment.
Figure 2. Reduction in stroke and bleeding after LAAO with Amplatzer Cardiac Plug. Patients with previous intracranial bleeding (IC Bleeding) are compared with patients without IC Bleeding.
The severity of stroke related to AF is a matter of concern as these strokes result in death in 20% or major disability in up to 60% of patients. [8] Freixa et al. investigated all reported cerebrovascular events at follow-up from the ACP registry. [9] There were 9 strokes (0.9%), 9 TIAs (0.9%), and 0 intracranial hemorrhages (0%) at follow-up. After excluding the 2 patients with previous disability, functional assessment showed disabling cerebrovascular events in 3 (19%) of the remaining 16 patients. In the 2 patients with previous disability, no increase in the baseline, in-hospital, and 3-month modified Rankin Score was observed. The median time from LAAO to the cerebrovascular event was 420 days, and 17 patients (94%) were on single-antiplatelet therapy when the event occurred. The authors concluded that cerebrovascular events after LAAO with the ACP system were infrequent and mostly non-disabling. In another relevant analysis form the ACP registry, Freixa et al. investigated the impact of patient age in the overall LAAO outcomes. [10] In this analysis, LAAO with the ACP was associated with similar procedural success in patients aged < 75 and ≥ 75 years. Interestingly, older patients had a higher incidence of cardiac tamponade but at follow-up stroke and major bleeding rates were similar among groups.
An important subgroup of patients in the ACP Registry, those with previous major gastrointestinal bleeding (MGIB) as indication for LAAO, was studied by Lempereur et. al. [11] A total of 151 patients with previous MGIB were evaluated. Periprocedural major bleeding events were more frequent in patients with previous MGIB as compared to those without MGIB (4.0% vs 0.8%, p = 0.001). With an average follow-up of 1.3 years, the observed annual rate of stroke/transient ischemic attack and major bleeding for patients with previous MGIB were 2.1% (61.4% relative reduction according to the CHA2DS2-VASc score) and 4.6% (20.1% relative reduction according to HAS-BLED score), respectively. The study concluded that in patients with NVAF and previous MGIB, LAAO was associated with a low annual rate of stroke/transient ischemic attack. Periprocedural major bleeding events were more frequent in this specific population although the annual major bleeding rate showed a 20.1% relative risk reduction according to the HAS-BLED score.
A very important, partially unexplored field in LAAO therapy is the choice of antithrombotic medication after the procedure. Taking into account that some patients may not tolerate well DAPT, Korsholm et al. investigated the use of single antiplatelet therapy after LAAO with the ACP or the Amplatzer Amulet in 107 patients. [12] With a median follow-up of 2.3 years, device-related thrombosis was detected in 1.9% of cases. Annual stroke reduction was 61% compared to the predicted rate, and annual risk of major bleeding was reduced by 57%. Therefore, the authors concluded that LAAO with the ACP or the Amulet occluder could be safely performed with aspirin monotherapy after implantation, without an increased risk of device-related thrombosis or stroke.
In a study including 500 consecutive patients who underwent LAAO (408 treated with ACP – 92 treated with Amplatzer Amulet) published by Koskinas et al., overall early procedural success was 97.8%, and MAEs occurred in 5.8% of patients, with no effect of device type or size. [13] Independent predictors of MAEs were device repositioning and left ventricular ejection fraction <30%. It should be noted that this center follows fluoroscopy-only guidance for LAAO procedures.
The largest to date report on the Amplatzer Amulet is a global, prospective observational study that enrolled 1088 patients who were treated with this device in 64 clinical sites, worldwide. [14] The majority of patients (82.8%) had a contraindication to OAC therapy. Successful device implantation was achieved in 99.0% of patients. Major adverse events occurred in 3.2% of patients during the proce¬dure and the index hospitalization, including 2 deaths (0.2%), 26 major bleeding events (2.4%), and 2 strokes (0.2%). Patients were discharged on a single antiplatelet agent (23.0%), DAPT (54.3%) or OAC (18.9%). The TEE fol¬low-up 67±23 days post procedure that was available in 673 patients showed adequate (<3 mm jet) occlusion of the left atrial appendage in 98.2% of patients and device thrombus in 10 patients (1.5%), as evaluated by core laboratory analysis. The authors concluded that compared to other recent reports on LAAO, this study showed a high implant success rate (99%) and similar peripro¬cedural risk in a real-world all-comers cohort of AF patients at high risk of stroke and bleeding. Moreover, TEE follow-up data confirmed high closure rates at 1-3 months post implant. Long-term clinical outcome data are being collected and will be reported in the near future.
The Amplatzer Amulet device is currently being evaluated in a randomized controlled trial (Amulet IDE Trial; ClinicalTrials.gov Identifier: NCT02879448). The study is aiming to enroll 1600 patients from 150 centers (100 in the US and 50 worldwide). The Amulet device is compared to the Watchman device (Boston Scientific Inc. Marlborough, MA, US) in a 1:1 ratio. The primary safety endpoint is a composite of procedure-related complications, or all cause death, or major bleeding through 12 months. The primary effectiveness endpoint is a composite of ischemic stroke or systemic embolism through 18 months. A secondary endpoint is device closure, defined as residual jet around the device ≤ 5 mm at the 45-day visit documented by TEE. This study is the largest randomized study on LAAO and the first randomized comparison of two occluders.
An important remaining question in the field is the comparison between LAAO and NOAC therapy. A small, randomized clinical trial from the Czech Republic (PRAGUE-17 study) is currently recruiting patients, aiming to enroll a total of 400 subjects. Patients are randomized to undergo LAAO with either Watchman or Amplatzer Amulet or receive NOAC therapy. The primary endpoint is a composite of “stroke or systemic cardioembolic event or clinically significant bleeding or cardiovascular death or procedure or device-related complications” at one year.
One of the main limitations of LAAO-related clinical studies is the lack of universal definitions for parameters and endpoints to be assessed. A recent publication entitled “Percutaneous LAAO: the Munich consensus document on definitions, endpoints, and data collection requirements for clinical studies” aimed to address this issue. [15] This document proposes a consistent approach in the assessment and reporting of clinical results, including comparisons with other devices and with surgical or pharmacological therapies. It is endorsed by the European Heart Rhythm Association, the European Association of Percutaneous Cardiovascular Interventions, and other societies.