Anesthetic Management In Electrophysiology Laboratory: A Multidisciplinary Review
Mustafa Yildiz1,2, Hulya Yilmaz Ak2, Dogac Oksen1, Sinan Oral3
1Department of Cardiology.2Anesthesiology and Reanimation, Istanbul University Cardiology Institute, Istanbul, Turkey.3Independent Investigator, Istanbul, Turkey.
Many clinical challenges have been encountered in electrophysiology laboratories (EP) while implanting intracardiac defibrillators for lethal arrhythmias, using pacemakers for bradyarrhythmias, placing pacemakers with multiple leads in patients with heart failure and cardiac ablation procedures. In this environment, anesthesiology plays a very critical role to ensure patients comfort, as well as maintains operator’s convenience and facilitate management of undesired situations.
EP laboratories are mostly used for diagnosis of certain heart diseases. Meanwhile, with the exponential increase in interventional procedures in our decade, electrophysiologists’ need to cooperate with the anesthesiologists more frequently.
The literature is still unclear about the effects of anesthetic agents on cardiac conduction pathways, but as we know with our previous data, the most agents we are using currently have more or less effect on the cardiac conduction systems. In this review, we aimed to describe the safe anesthesia methods in cardiac diagnostic procedures and have a closer look up the anesthetic outcomes of these procedures. This article comprehensively reviews the anesthesia practice encountered in electrophysiology laboratories.
Key Words : Electrophysiology, Anesthesiology, Ablation, Device Implantation.
Correspondence to: Dogac Oksen,Cardiologist
Department of Cardiology, Istanbul University Cardiology Institute, Istanbul, Turkey.
An intracardiac demonstrates the electrical activity of heart with providing position and structure, voltage and duration of arrhythmia so as to certifying the accurate diagnosis. Intracardiac records obtain knowledge from high right atrium, coronary sinus, his bundle and right ventricular apex while extracardiac electrocardiography leads are I, II, III, aVL, V1 and V6. Those leads are helpful for planning the diagnosing and guiding strategy while ablation procedure is being performed.
Different mechanisms induce arrhythmias, for example; impairment of impulse generation causes bradyarrhythmias and sick sinus syndrome, sinus arrest, also deterioration of impulse propagation in the cardiac pathway systems known as atrioventricular node block brings indications for pacemakers.
Tachyarrhythmia originates with one of the three certain pathologic mechanisms; the most common one is reentry mechanism, the others are increased automaticity in conduction cycle and triggered activity from another unusual cardiac focus. Reentry causes from an unexcitable area which is surrounded by an electrical pathway circle. Two pathways with different conduction velocities, initial pathway allows retrograde conduction while the other pathway is on refractory period. If anomalous conduction exits between atria and ventricles, that is an example of Wolf – Parkinson –White (WPW) Syndrome which is an anatomical reentry. Supraventricular tachycardia (SVT), WPW Syndrome, atypical atrioventricular nodal reentry tachycardia (AVNRT), sustained monomorphic ventricular tachycardia, atrial fibrillation (AF) and atrial flutter (AFL) is generally caused by reentry mechanism. Variety of metabolic effects, such as; ischemia, hypoxia, increased sympathetic activity, acid base instability, hypokalemia, hypomagnesaemia can be the cause of increased automaticity. Abnormal automaticity originated from electrical discharge of non-pacemaker cells.
Ventricular arrhythmia caused by ischemia or electrolyte imbalance, can be categorized under increased automaticity. QT prolongation, which can lead to polymorphic ventricular tachycardia (VT), is an example to triggered activity. Exceeding after-depolarization can trigger the tachyarrhythmia as well. Calcium and catecholamine imbalance facilitates reaching the threshold potential.
Ablation procedures are potent alternatives of pharmacologic therapy in patients with cardiac conduction problems. These procedures are usually performed under sedation with using the standard monitorization techniques. Any increase in the complexity of procedure, significantly raises the effort of operator, the procedure becomes more time consuming so sedation with more depressed level of consciousness will be required.
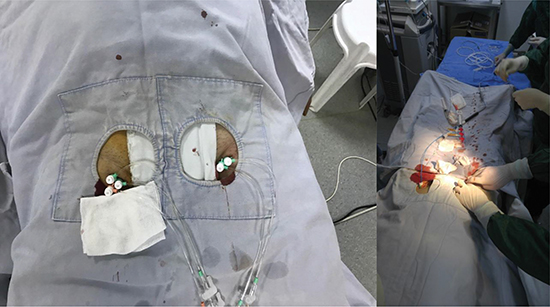
EP laboratories, are used both for diagnostic for certain heart diseases, interventional procedures such as ablation and device implantations. In this risky and stressful environment, anesthesiologists, interventional cardiologist and other staff should work collaboratively and move together (Figure 1A,1B).
Figure 1. View of a electrophysiology laboratory during ablation procedure.
Device implantations including pacemaker, intracardiac defibrillators (ICDs), Cardiac resynchronization therapy (CRT) can be performed under sedation and local anesthetics.
Pacemakers are the backbones of bradyarrhythmia treatment. ICDs, are expected to end certain reentry tachycardias by their anti-tachycardia pacing and expected to terminate malign arrhythmias such as VT and ventricular fibrillation (VF) by sequent defibrillation therapy. The patients require cardiac device frequently eldery and have many comorbidities, history of heart failure, myocardial infarction. Especially, heart failure patients may not stay long time in supine position. Device implantation complications rise along with advancement of device. CRT devices bring more complication rates rather than single chamber pacemakers. Mostly seen complications related with procedure are tamponade and pericardial effusion due to cardiac perforation and myocardial injury, pneumothorax, intrathoracic bleeding. Generally, patients require local anesthetic agents to prevent incisional discomfort and small amount of intravenous sedation. While pacemakers can be implanted under sedation, some studies suggest deeper levels of unconsciousness provided with sedation in the course of intracardiac defibrillator implantation. Particularly, during tunneling of leads and testing the threshold of defibrillation, anesthesiologists perform sedation. Most researches so far, are performed by using midazolam and the extending effect equivalents like propofol. Fentanyl is preferable since it has analgesic effect as well. A remarkable point in here is, with the more extending effect and deeper anesthetic sedation it needed, anesthesiologists will stand for more technical supply and medication, which eventually affect the total cost of procedure (1).
Fox et al (2) mentioned using local anesthetics and light sedation in patients who undergo cardiac implantation is safer than using general anesthesia (GA). Five hundred implantations have been scanned and there were not any significant difference between midazolam and fentanyl doses in biventricular devices with longer procedure times versus others. Only 11% patients described discomfort about procedure.
ICD implantation was performed under conscious sedation achieved with midazolam, morphine, promethazine and etomidate was administered for defibrillation threshold testing (DFT). Conscious sedation with combined hypnotic agents also additional etomidate to ensure sedation was found safe with low complication rates (3). It is feasible for electrophysiologists to implant devices with local anesthetics and sedation upon patient’s requirement. Lately, with the advances in device technology, necessity for DFT has stuck.
Previously, surgically situated pericardial defibrillators with large generators placed in abdomen, left their place to percutaneously implantable defibrillators with smaller generators. Today, subcutaneous cardioverter defibrillators with miniaturized generator sizes have been successfully used. Subcutaneous intracardiac defibrillators (s-ICD) consist of a single tripolar parasternal lead and a generator connected with lead, mostly implanted at left sternal midline intersected at 5th or 6th rib. s-ICDs are especially useful for pediatric cases with congenital heart diseases and cases that have previously failed ICD implantation due to struggles for finding the transvenous access. This procedure has rather less risk in the aspect of pneumothorax and vascular complications than conventional procedures. With EFFORTLESS and IDE studies, the efficacy and safety of s-ICDs were demonstrated (4).
On the account of large dissection area and tunneling process, most anesthesiologists tend to prefer GA. According to studies, monitored anesthesia care (MAC), which contents sedation or local anesthetic with hemodynamic follow up is also effective, feasible and reliable. MAC does not reduce patients comfort, complicate management of procedure and increase intubation requirements. MAC provides adequate sedation and analgesia in the experienced hands (5).
Re-entrant arrhythmias constitute large majority of arrhythmias and are feasible for catheter ablation with using radiofrequency or cryothermal energy. Energy is conveyed to site by catheter and drives the point electrically inert. Femoral, subclavian, internal jugular and brachial vein accesses are usually used to introduce electrodes and catheters. Left side enterprises are performed with either transseptal approach or retrogradely through aorta.
Ablation procedures require sedation; especially ablation of WPW, atrioventricular nodal reentrant tachycardia (AVNRT), AFL and other reentrant tachycardias can be tolerated by patients while they are under their spontaneous breathing and relatively take shorter time. AF and VT ablations are relatively longer procedures and are required GA or conscious sedation.
AF revealed from ostium of pulmonary veins in the left atrium. Transoesophageal echocardiography is a must before ablation to ensure that there is not any thrombus in left atrial appandage. Access is obtained mostly via femoral vein and catheters are introduced into left atrium by passing through foramen ovale on the interatrial septum after systemic heparinization. Certain techniques are used to ablate culprit tissue. Radiofrequency ablation (RFA) heats myocardial tissue with convection of heat. Extending RFA beyond the myocardium may cause thermal injury to joining tissues like esophagus. This damage may result with left atrial – esophageal fistula formation. Basically, limiting the ablation energy (25 to 35 W ) and restricting duration under 30 seconds are the most common clinical approach to minimize thermal injury. Irrigation with saline from the tip of catheter restricts the ablation area and concentrates the energy to a small endocardial area. Cold flow via catheter reduces the risk of esophageal injury but continuous monitorization of luminal esophageal temperature is strongly recommended to avoid any excess heating of adjacent tissues. Esophageal temperature monitoring is associated with decreased complication rates. There is not any necessity to evaluate esophagus with imaging tools previously, but luminal esophageal temperature probe is better inserted under fluoroscopic guidance, positioned behind posterior wall of left atrium. Optimal contact is required between esophageal wall and the probe to ensure accurate temperature (6).
Unbounded saline infusion may result with pulmonary edema by fluid overload, so the anesthesiologist and electrophysiologist should limit the administered fluid amount and apply intravenous furosemide upon for total balance. Cryoballoon ablation (CBA) is balloon based catheter, balloon is inflated in every pulmonary vein and the conduction of ectopic rhythm is blocked via cryothermal energy given to myocardial tissue. Certain handicaps of CBA are mostly associated with phrenic nerve paralysis and structural damage concluded with pulmonary vein stenosis. Anesthesiologists should avoid using neuromuscular blockade in order to unnoticed phrenic nerve stimulation at ablation site. According to previous studies, GA has better outcomes than conscious sedation or/and MAC. Although, GA costs slightly more than the other anesthetic techniques, GA has better results and more effective in cardiac ablation procedures. GA provides greater comfort for both patient and for operator in this kind of relatively long procedure. It restricts patients movements that may cause an unwanted distortion in critical anatomic plan as well as anesthesiologist can control respiratory rate and depth during the procedure which prevents any uncontrolled respiratory movement (7).
Some anesthetic agents effect the cardiac ionic balance and can change propagation thresholds during process. Propofol, which is the most frequent agent used both in GA and deep sedation, effects R-R interval, atrial electrical activity. In experimental studies, Propofol increases AF cycle length in right atrium, but has opposite effect on left atrium. Additionally, propofol tends to organize right atrium electrical activity but induces chaotic conductions in left atrium (8).
Lastly developed technology, remote magnetic catheter navigation significantly reduces fluoroscopy time and less effected by respiratory movements during procedure. Remote magnetic catheter navigation system minimizes the impacts of respiration so conscious sedation also can be safely and effectively performed comparing to conventional techniques (9).
AFL is a macro-reentrant atrial rhythm, less common arrhytmia than AF and arises from right atrial chamber. AFL can be counterclockwise transmitted around tricuspid annulus also called typical flutter or clockwise direction called atypical (reverse) flutter. A rare form, intra-isthmus flutter usually occurs after AFL ablation (Figure 2). Initial treatment of AFL commonly synchronous DC cardioversion with low energy approximately about 50J and can be gradually increased depending on clinical conditions (Figure 3, Figure 4). Procainamide, amiadarone, diltiazem, verapamide, adenosine are other pharmacologic cardioversion choices which are commonly used in clinics. To interrupt AFL transmission, ablation is performed between inferior vena cava and tricuspid valve. Sedation is adequate in this procedure because the procedure is shorter, much comfortable and less painful than AF ablation. Agitated patients may need deeper sedation with close hemodynamic follow-up. During RFA, patients usually feel a short, mild pain caused by heating catheter. A recent study in the literature compared remifentanyl (a potent, short acting analgesic drug) and propofol (as short acting intravenous anaesthetic, used to induce GA) in the aspect of patients’ anesthetic outcomes who undergo AFL ablation. In the group of patients who subjected to remifentanyl anesthesia suffered from induced muscular rigidity and in this group more patient needed ventilatory support. In contrast with the other group (remifentanyl), the patients subjected to propofol had longer recovery times and faced with decreased blood pressure. Patient/operator satisfaction feedbacks were similar in both groups (10).
Figure 2. Resistant atrial flutter ablation with 3D mapping developed after atrial fibrillation ablation.
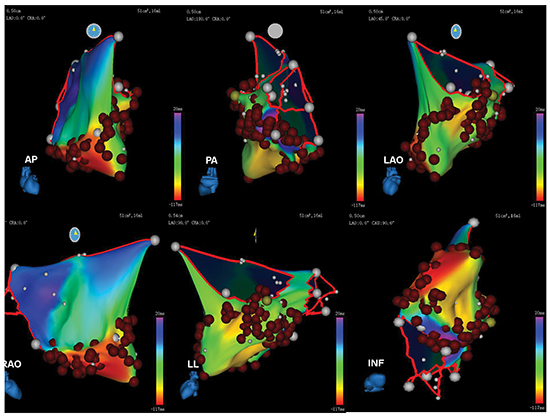
Figure 3. Atrial flutter, seen in ablation procedure .
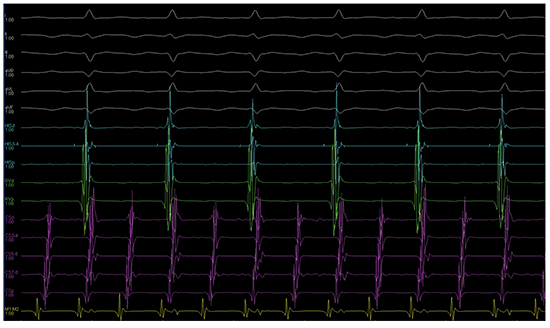
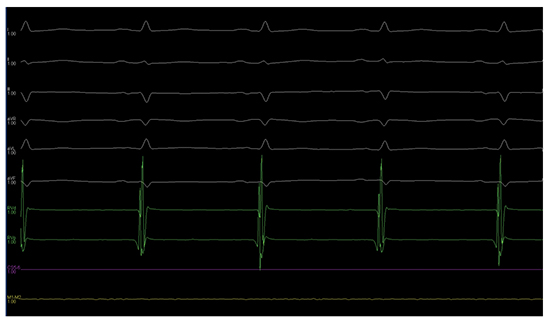
Figure 4. Sinus rhythm restored after procedure.
Inhaled Nitrous Oxide (N2O) is a rarely used potent anesthetic-analgesic and alleviates operation related anxiety as well. N2O causes vomiting attacks but it is still feasible and safe for relieving patients’ discomfort during AFL ablation (11).
Other Supraventricular Tachycardias
SVTs mostly encountered as an episodic attack and comprised of a regular rhythm with narrow QRS complex (except multifocal atrial tachycardia (MAT) and aberrant conduction SVT). SVTs mostly refer atrial tachycardia, AVNRT and atrioventricular reciprocating tachycardia (AVRT). Catheter ablation is an amenable and favorable treatment method for resistent and repetitive arrhythmias. SVT triggered via a reentry mechanism commonly seen in healthy patients, due to premature atrial and ventricular beats arouse tachycardia. AV node blocking agents and other antiarrhythmic medication is convenient as an initial treatment. RFA procedures to cure frequent SVT’s including AVRT, AVNRT and atrial tachycardia are accomplished with increased success rates (12).
A previous study in patients underwent SVT ablation, there was not significant difference in induction rates of propofol and ketamine while stimulating arrhythmia. In patients underwent ketamine seduction has shortened the atrial conduction time, increased the heart rate and with stimulation of sympathetic nervous system enhanced blood pressure. Overall ketamine is suitable for patients with bradycardia and patients who had suffered from hypotensive episodes. Ketamine induces unspecified arrhythmia arising from myocardial tissue; this may facilitate electrophysiological studies and ablations (13).
RFA of preexitation syndromes with accessory pathways have over 90% success rates. An electrophysiologic change with any kind of medication during the ablation procedure reduces transactional success. Sevoflurane is a volatile liquid anesthetic used as an inhaler agent for induction and maintenance of GA. Although sevoflurane has been proven to reduce the sinoatrial conduction time and atrial – His interval time, this effect of sevoflurane has not been encountered clinically for now, both in atrioventricular and accessory pathways (14).
In patients with ventricular arrhythmias typically have a serious cardiac pathology such as ischemic heart disease, congestive heart failure or cardiomyopathy. VT and VF are the most common life threatening arrhythmias, can cause sudden death by progressive hemodynamic deterioration, patients require rapid evaluation.
Premature ventricular contractions (PVCs) are benign ventricular arrhythmias, can also be seen in healthy people. As the frequency increases, PVCs give a feeling of discomfort and palpitations, very frequent ones can cause tachycardiomyopathy. Unifocal early ventricular beats arising from the right ventricular outflow tract have a risk to turn into a malignant arrhythmia such as non-sustained VT, VT or VF. PVCs arising from distal of Purkinje fibers, PVCs in patients with long QT, PVCs in patients with Brugada Syndrome are more likely to trigger a malignant ventricular arrhythmia (15).
Conscious sedation and analgesia are provided with propofol, midazolam, fentanly, ketamine administration. In case of necessity to enhance PVCs; propofol, midazolam sedatives are discontinued. To trigger early beats, isuprel is administered and atrial, ventricular burst pacing are performed during infusion. Isuprel enhances cardiac conduction and used for ventricular arrhythmia induction after standart process of ventricular pacing. Isuprel should be used carefully in patients with coronary artery disease while since isuprel is a cardiac stimulant, enhances blood pressure and augments vasoconstriction. There is not any significant difference between midazolam, propofol, fentanyl and ketamine along mapping and inducing PVCs. Short acting sedatives would better to choose and anesthetics can titrated to low doses or can be discontinued upon adequate PVCs (16).
VT is a fast and improper electrical activity arises from ventricular myocardium, triggered with ischemia, sympathetic activity, heart failure and electrolyte imbalance.
Frequently, underlying reason of the VT is the ischemic heart disease. Heart failure with fibrotic myocardial region, structural heart diseases and idiopathic channelopathies can be the other frequent reasons as well. So far, outcomes of pharmacotherapy in hemodynamically unstable patients with VT and VF have not reached the satisfactory levels. Although antitachycardia pacing and ICD shocks are another favorable and effective way of treatment, patients usually suffer from great discomfort and pain with repetitive shocks. Unresponsiveness to medical treatment and recurrent arrhythmic attacks optimally need to manage with VT mapping and ablation. Focal VT source arises due to triggered activity or enhanced automaticity and frequent sources are right ventricular and left ventricular outflow tract (Figure 5)
(17).
Figure 5. mapping and ablation of a right ventricular outflow tract tachycardia.
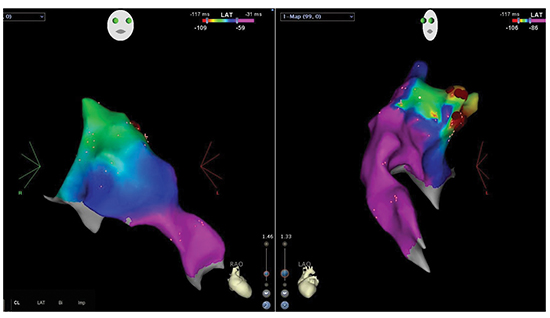
Scar tissue produces reentry in ischemic heart diseases, cardiomyopathies, arrhythmogenic right ventricular dysplasias and able to cause sustained VT which are all feasible for catheter ablation (18).
Retrograde access through aorta or transseptal approach via right atrium is both enable for catheterization of left ventricle. Epicardial access is an option in case of unsuccessful endocardial trial or if the source of VT positioned adjacent to epicardium. Epicardial access is performed percutaneously with subxiphoid approach or the second option preferred in patients whose adhesions make the procedure impossible for surgery with or without sternotomy. Many of institutions perform VT ablations under anesthesiologist's coverage. A preoperative anesthesia consultation is a must, history of illness, history of operations, any kind of anesthesia received, any kind of allergy, chest x-ray, family history, laboratory values, electrocardiography, cardiac functions, vitals, physical examination and airway assessment with Mallampati score comprised preoperative anesthesia evaluation. Anesthesiology and ablation procedure should be planned according to VT etiology since this effects of the choice of hemodynamic expectations and anesthetic agents. Patients’ vitals and hemodynamic changes should be well monitored. Inducing VT by activation mapping may collapse the hemodynamic stability and inotropic support and/or mechanical intervention may be necessary. RFA catheters have a tip that continuously infuses saline which has an approximate rate of 2 ml/min. As length of procedure considered, anesthesiologist should be alert about excess fluid loading and decompansation of heart failure. Urine output must be followed closely and diuretics should be administered according to the fluid balance.
However it is not exactly clear that anesthetics suppress VTs, electrophysiologists prefer patients awake in the course of arrhythmia induction. To maintain patients comfort and serenity, a low dose sedative agent can be administered. To prevent unsuccessful VT stimulation, European Heart Rhythm Association (EHRA) suggests avoiding from GA, deeper sedations. Additionally, neuromuscular blocking agents should be avoided to see any responsiveness of phrenic nerve during procedure (17, 19).
Short – running interventions such as right ventricular and left ventricular outflow tract VT ablation, can be undergo with sedation or MAC, but in long lasting transactions like fibrotic tissue or scar originated VT ablations GA must be the preferred option to maintain patients comfort during process (20).
Electrical cardioversion (EC), is generally used to end up tachyarrhythmia and restore sinus rhytm by electrical shock. EC should be performed under the circumstances of accessibility to advanced life support equipment, anesthetic support.
A Tachyarrhythmia can evolve into a life-threatening situation, frequently with deterioration of hemodynamics and require emergent intervention to maintain hemodynamic stability. Unstable patients should undergo emergent EC. EC is a relatively painful procedure in conscious patient. A short sedation lasted up in a few minutes with slightly losing the consciousness is the aim of anesthesiologist before EC. With ventilation support, patient can be prevented from apnea which is promoted by sedative and anesthetic agents. Oxygen with mask, ventilation bag or nasal cannula during administration helps to avoid hypoxia. Conscious sedation would reduce awareness and improve patients comfort without rendering the patient completely unconscious. Reduced awareness can cause tongue injury which is the most common airway trauma in cardioversions. Basicly, placing a soft bite block before starting cardioversion will help to prevent tongue trauma. Short acting hypnotics like propofol, etomidate dexmedetomidine, thiopentone, diazepam are suitable for setting up the patient. Propofol is the most commonly used agent acting about 5 – 10 minutes with a single rapid injection. Propofol does not have analgesic effect, adding dexmedetomidine to propofol supply analgesia to patient however, additional hypotensive effect complicates usage in hemodynamically unstable cases (21).
General Management and Considerations
In EP laboratories, patients have wide variety of different characteristics. Physicians deal with young patients presented with AV blocks, accessory pathways and eldery group with many comorbidities, heart failure and mortal arrhythmias. Preoperative anesthesiologist examination is essential to decide anesthesia type, ventilation options and useful to anticipate any kind of potential complications.
Device implantation mostly require local anesthesia and usually supported with sedative agents. For GA and deeper sedation laryngeal mask, airway, suction apparatus and intubation tube should be kept ready in case of any respiratory depression required rapid sequence induction. The monitoring devices are essential to safe procedure. The following utilities must be included: Electrocardiography, non - invazive blood pressure, temperature, pulse oximeter. External defibrillation and pacemaker pads must be placed on chest wall in all cases. Urethral catheterization is a routine medical procedure for prolonged procedures to calculate fluid balance. When neuromuscular blockage is performed while CRT implantation, physician should be alert to avoid pacing of phrenic nerve. Invasive arterial monitorization should be decided upon patients comorbidities abd hemodynamic status. Radial arterial cannulation is more feasible and safe rather than femoral artery for hemodynamic monitorization. Subclavian or internal jugular lines allow central venous pressure measurement used to assess cardiac preload and volume status whether patient has volume overload (22).
Chronic obstructive pulmonary disease, advanced heart failure patients may get worsened due to long procedure times in supine position, also sedation induces respiratory depression may leads fatal consequences. Patients presenting with ventricular arrhythmias are companion with hemodynamic instability. This challenging cases can be supported with inotropic agents, intra – aortic balloon pumps and this kind of cases should be managed with consensus of electrophysiologist and anesthesiologist. Also, during electrophysiological interventions large venous and sometimes arterial sheaths are being used and in some cases septal punctures are created. Multiple catheter usage and prolonged procedure times rise thrombi risk. Despite, sheaths are flushed with heparin, there is still risk of clot formation, thereby can cause cerebrovascular accident. Systemic heparinization is monitored by targeted activated clotting time and is better kept over 300s (22). A cerebral oximeter monitorization facilitates early recognizing of any kind of cerebrovascular accident happens while on ablation procedure.
EP laboratories submit different environment rather than conventional operation rooms. Foreign location brings some difficulties to anesthesia providers. EP laboratories mostly located apart from main operation rooms and intensive care units, therefore there is a limited access to advanced respiratory equipments, some drugs not used commonly in laboratory, support from anesthesia team. C – arm fluoroscopy device, over the patients head makes it difficult for the anesthesiologist to reach the patient' s respiratory tract and may cause delays for urgent interventions. Hybrid EP laboratories are one of the recent developments, this laboratories contain positive ventilation, sterilization and surgical equipments. These labs are expected to reduce mortality and morbidity due to procedural complications and improve outcomes.
Monitorization, Ventilation and Anesthesia
Patients undergoing procedure in EP laboratories may not tolerate long procedure times and painful interventions, thus cases require anesthesia continuum from minimal sedation to GA. Deep sedation is a good alternative for patients who are anxietic or inable to stand still. GA may be required for longer procedures, patients who have significant cardiovascular or pulmonary diseases and intubation necessity.
Trentman et al (23) reported 186 patients who were underwent various kinds of ablations with planned MAC anesthetics. Twenty of them converted to GA via endotracheal tube insertion or laryngeal mask airway, 54 of them required insertion of oral-pharyngeal airway or nasal airway. Thus, 40% (74/186) of the patients received MAC anesthetics required an airway intervention. Consequently, according to these results, entirely from sedation to GA, all kind of anesthetic interventions should be performed by anesthesia providers. Laboratory must be prepared for any kind of emergent airway intervention.
Oxygen supplementation may be received by nasal cannulas, face masks with 2 – 4 L /min. Motionless is essential for ablations especially for AF ablation. GA is a better selection in such cases. GA can be maintained with inhalation, intravenous anesthetics or combination of them. Respirations of patients are provided with positive pressure mechanical ventilation and during ablations, 30 – 60 seconds temporary arrests can be necessary to ensure stability. EP operators may request breath holding to maintain chest stiffness during ablation process. Cerebral oximeters provide monitorization of cerebral perfusion, hemodynamic status and increased arterial carbon dioxide levels.
High frequency jet ventilation (HFJV) is a type of mechanical ventilation which increases respiration rate over 100 breaths per minute, also known as lung protective ventilation used frequently in acute lung injury. Anesthesiologists are using this technique in EP laboratory to prevent rapid and excess movements of chest wall. Tidal volume in HFJV significantly lower than conventional ventilation, end tidal carbondioxide monitorization has an appearance like saw tooth. During critical periods requiring absolute stability, HFJV lessens chest walls movement. While using HFJV, another intravenous anesthetic may be necessary to maintain awareness because of changed breathing circuit and also an arterial line will help close monitoring of arterial blood gases, oxygenation, acidity and carbon dioxide level. In case of hypercarbia, frequencies of respirations are reduced to conventional rates. Trapped air in distal alveolar areas increases the distal airway pressure, may cause barotrauma (24). HFJV may stimulate pulmonary reflexes and cause coughing. To suppress coughing reflexes, depth of anesthesia would better be increased to an adequate depth. HFJV is a reliable alternative in atrial septum puncture, AF ablation.
A retrospective study included 188 cases underwent pulmonary vein isolation to treat AF under HFJV. All cases underwent ablation under GA and intubated before procedure. 13 of them converted to conventional positive pressure ventilation, because of increased pCO2 or decreased PaO2 (25).
Propofol is the most preferred agent provides both sedation and GA in ablation procedure however; it has minimal effects on QT interval and conduction system. It also represses catecholaminergic activity, inhibits ion channels of myocardium.
Compared to propofol, volatile anesthetics such as sevoflurane has been showed that has some prolongation effects on QT interval and co-use of it with propofol will reverse its effect on QT interval (26). Volatile anesthetics have variable effects on rhytm however opioids have limited effects in terms of electrophysiology. Enflurane is the most potent volatile anesthetic that has affects on accessory pathways.
A reduction of ischemic and reperfusion arrhythmia was showed in dogs underwent anesthesia with isoflurane, desflurane and sevoflurane. Desflurane and sevoflurane significantly reduces arrhythmia incidence when compared with halothane (27).
A study in pediatric population, compared desflurane and fentanly as an anesthetic for RFA of supraventricular arrhythmias. According to study both agents do not have significant different effects on electrophysiological measurements and SVT was inducible in both groups. Dexmedetomidine is a sympatholytic agent blocks a2 receptors. It has shown that dexmedetomidine may suppress supreventricular arrhythmias (28). Desflurane reduced mean arterial pressure and enhanced heart rate (29).
Patients in EP laboratories, have comorbidities like congestive heart failure, ischemic heart disease, arrhythmias etc., so each case needs to be individually planned for appropriate anesthesiology. Mostly, electrophysiology catheterizations laboratories are not particularly meet the needs of anesthesia department; anesthesiologists have limited access to support devices in case of any emergent situation. As a result of technical limitations, cases should be well planned; anesthesiologist plays a crucial role on procedure. Cardiac anesthesia departments should develop a special concept for qualifying their approach in EP laboratories and work for the advance adaptation to this totally new environment.