Subxiphoid Hybrid Approach for Epicardial/Endocardial Ablation and LAA Exclusion in Patients with Persistent and Longstanding Atrial Fibrillation
Nitish Badhwar1, Ghannam Al-Dosari2,3, Jonathan Dukes1, Randall J. Lee1
1Division of Cardiology, Section of Cardiac Electrophysiology, University of California, San Francisco, San Francisco, CA.2Division of Cardiovascular Surgery, University of California, San Francisco, San Francisco, CA.3Division of Cardiothoracic Surgery, University of Texas Medical Branch, Galveston, TX.
Two patients with long-standing atrial fibrillation (AF) refractory to medical management and with prior pulmonary vein isolation underwent a new hybrid epicardial/endocardial subxyphoid approach for AF ablation and left atrial appendage (LAA) ligation. Pulmonary vein and LA posterior wall isolation, as well as LAA exclusion were achieved in both patients. There were no procedural complications. Both patients remain in sinus rhythm. Both patients are off antiarrhythmic medications.
Key Words : Atrial Fibrillation, Hybrid epicardial/endocardial ablation, LAA ligation.
Correspondence to: Randall J. Lee, MD, PhD
Division of Cardiology, Section of Cardiac Electrophysiology University of California, San Francisco 500 Parnassus Avenue San Francisco, CA 94143
Pulmonary vein isolation (PVI) is a viable alternative for the treatment of paroxysmal atrial fibrillation (AF) and is associated with a high rate of success[1]. However, outcomes for treating persistent and longstanding persistent AF with PVI have been disappointing. Success rates for PVI in patients with persistent and longstanding persistent AF are approximately 45% after one catheter ablation (CA) procedure at 1 year, whereas approximately 65% freedom from AF occurs after multiple procedures [2].
The Cox-Maze III procedure is considered the gold standard for non-pharmacological treatment of AF[1]. It has been associated with a the high rate of maintaining sinus rhythm with a low incidence of stroke in patients with AF [3], but due to the complexity and morbidity of the surgery, few centers perform the operation. Minimally invasive surgical epicardial ablation and hybrid endocardial/epicardial approaches have shown initial promise in maintaining sinus rhythm compared to catheter-based PVI, but are associated with considerably greater number of procedural-related adverse events compared to catheter ablation[4-6].
In this case report, we report on 2 patients with longstanding atrial fibrillation (AF) refractory to medical management and previously failed PVI who underwent a new hybrid subxyphoid epicardial/endocardial approach for AF ablation and left atrial appendage (LAA) ligation.
72 year old woman with a CHADs-VASC score of 4 and long-standing AF refractory to medical management including amiodarone, and a previous PVI underwent a subxyphoid minimally invasive epicardial surgical ablation and LAA closure.
A small subxiphoid incision measuring 6 cm was made. After the xiphoid process was excised and the diaphragm retracted inferiorly with 0 silk suture, the pericardium was entered along the edge of the diaphragm. Increased visualization of the heart within the pericardial cavity was achieved by retraction of the pericardium with stay sutures and a mini thoracotomy retractor with the longer blade on the diaphragmatic surface of the pericardium to inferiorly push the diaphragm down. The pleural space was not entered. The entire inferior surface of the left ventricle and part of the left atrium, the IVC, the right pulmonary veins can be visualized by lifting the heart. Retracting the heart medially with malleable retractor allows for visualization of the left pulmonary veins as well at the LAA.
Isolation of the pulmonary veins were attempted with the Atricure bipolar radiofrequency clamp (AtriCure, Inc, West Chester, OH). Since the Atricure bipolar radiofrequency clamp was designed for a thoracotomy approach, the right angle prevented adequate positioning around the pulmonary veins resulting in partial ablation of the pulmonary veins. Completion of pulmonary vein isolation as well as the posterior left atrium was achieved with the standard Isolator® linear pen probe (AtriCure, Inc, West Chester, OH). Epicardial mapping was performed with the Isolator® linear pen probe to assure pulmonary vein isolation.
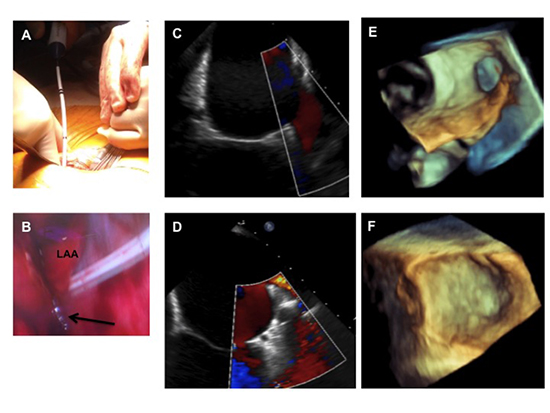
LAA closure was performed with the LARIAT suture delivery device (SentreHeart, Inc, Redwood City, CA). Direct visualization of the LAA was aided by selective lung ventilation, rightward rotation of the bed to lower the right side and rightward traction of the heart with a malleable retractor[Figure 1]. Transesophageal echocardiography (TEE) was used for verification of capture of all lobes of the LAA and complete LAA closure [Figure 1].
Figure 1. LAA ligation. Selective lung ventilation, rightward rotation of the table and gentle medial retraction of the heart with a malleable retractor (panel A) allows for direct visualization of the LAA and passage of the LARIAT snare over the LAA (panel B). The arrow highlights the snare around the LAA. TEE is used to assess capture of all the lobes. Panel C and E represent pre-ligation LAA of 2D and 3D TEE images, respectively. 2-D (panel D) and 3-D (panel F) TEE assessment of LAA closure 7 weeks post-LAA ligation verified permanent LAA closure.
The patient tolerated the procedure without any procedural complications. The patient did develop a pleural effusion on post-operative day 2. During her hospitalization, the patient remained in AF requiring 2 cardioversions to restore sinus rhythm. The patient was discharged in sinus rhythm without any antiarrhythmic medications. The patient remained in sinus rhythm.
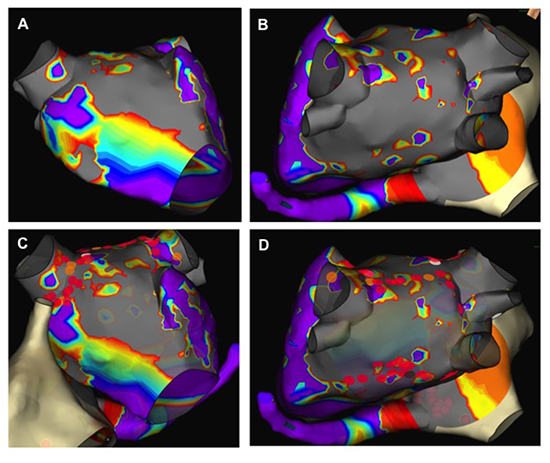
The patient underwent an endocardial electrophysiology study 7 weeks after her surgical procedure to assess for pulmonary vein and posterior LA isolation. In the baseline state, there was electrical connection of the right superior (RSPV), right inferior (RIPV), and left superior pulmonary veins (LSPV) to the left atrium. There was also electrical connection of the posterior wall to the left atrium, though the voltage map demonstrated significant scaring and low voltage throughout the posterior wall [Figure 2]. The spiral catheter (St Jude, Minneapolis, MN) was then positioned in the LSPV and ablation was performed at sites demonstrating electrical connection as demonstrated by local atrial electrograms for pulmonary vein isolation. Entrance block occurred in the LSPV with ablation at the anterior medial aspect of the LSPV. Demonstration of exit block was determined by lack of capture of the LA during pacing around the spiral catheter. Continued ablation in regions of normal voltage around the left superior and inferior veins were performed to complete the left sided WACA. Exit block in the left inferior pulmonary vein (LLPV) was demonstrated.
Figure 2. Voltage maps. Voltage maps prior to endocardial ablation demonstrates areas of residual voltage in the anterior aspect of the LSPV and RSPV (panel A, AP view) and small islands of normal voltage on the posterior aspect of the LA (panel B, PA view). Panel C (AP view) and panel D (PA view) demonstrate the endocardial ablation lesions (2 mm lesions). Red indicates voltage < 0.5 V. Purple indicates voltage >5 V. Gray indicates no signals (ie scar).
The spiral mapping catheter was next placed in the RSPV. The principle areas of continued conduction were in the anterior aspect of the RSPV near the roof. Entrance block was demonstrated, and pacing around the pulmonary vein also demonstrated exit block. Right-sided WACA line was completed by ablating in areas were there was still residual normal voltage (the anterior portion of the vein had significant voltage still present). Next, the spiral mapping catheter was moved to the RIPV where exit and entrance block of this vein was noted.
The spiral catheter was then positioned in the center of the posterior wall. The posterior wall was isolated by completion of the roof line by ablating in the small islands of normal voltage found in the roof between the right and left superior veins. After completion of the roof line, the inferior posterior wall line from the left inferior vein to the right inferior vein was completed. Completed of this line resulted in the LA posterior wall being electrically isolated. Exit block was then demonstrated by pacing around the spiral catheter in the posterior wall.
Atrial pacing was performed after PVI and posterior LA isolation. Typical cavotricuspid isthmus (CTI) dependent atrial flutter was induced. Patient underwent a successful CTI ablation with demonstration of bidirectional conduction block. Total ablation time was 21 minutes including the right-sided CTI ablation. There were no procedural complications.
The patient has not had symptoms of palpitations and has not taken any anti-arrhythmic drugs since her surgery. Extended monitoring for 4 weeks was performed at 6 and 12 months demonstrating sinus rhythm with no recurrence of AF. A follow up ECG during a visit at 14 months revealed sinus rhythm.
71 year-old woman with a CHADs-VASC score of 5, long-standing persistent AF and 2 failed pulmonary vein isolation procedures remained in AF despite continued treatment with amiodarone. She underwent a subxyphoid minimally invasive epicardial surgical ablation and LAA closure as described in case 1. The patient had a successful procedure with demonstration of bilateral pulmonary vein isolation, isolation of the posterior left atrium via epicardial mapping and closure of the LAA with the LARIAT device under direct visualization. There were no procedural complications. The patient had atrial flutter post-operatively with a controlled ventricular rate. The patient subsequently underwent a cardioversion to restore sinus rhythm. The patient remained in sinus rhythm on amiodarone. The planned follow up electrophysiology study was postponed due to the patient requiring an unrelated surgery for her peripheral vascular disease. After the patient’s peripheral vascular surgery, amiodarone was discontinued 1 month after her vascular surgery. The patient subsequently developed atrial flutter. Electrophysiology mapping of atrial flutter demonstrated a left sided atrial flutter involving the left pulmonary veins. Entrainment mapping was performed to delineate the circuit and was successfully terminated with radiofrequency catheter ablation. The placement of a 20-pole Spiral catheter was used to confirm entrance and exit block from all 4 PVs and the posterior LA. The patient has remained symptom free and in sinus rhythm off of amiodarone for 8 months, but has continued oral anticoagulation therapy. Extended 3 week monitoring at 6 months post-EPS study revealed predominantly sinus rhythm with rare episodes of asymptomatic atrial fibrillation of less then one-hour duration and constituting less then 6% burden.
The subxyphoid epicardial approach was attempted as an alternative to the bilateral thoracic or the transabdominal transdiaphragmatic minimally invasive surgical approach for epicardial AF ablation[5,6]. Although the bilateral thoracic approach has been reported to be more effective then CA for freedom of AF, the bilateral thoracic surgical approach was associated with a significant increase in adverse events compared to CA (23.0% vs 3.2%)[3]. The majority of adverse events associated with the bilateral thoracic approach were major bleeding and pneumothorax. Disadvantages of the transabdominal transdiaphragmatic single port approach is that epicardial LAA closure cannot be performed due to the posterior entry into the pericardial space, and the high rate of complications including those arising from entering the abdominal cavity[6]. The initial attempt of epicardial ablation and LAA exclusion via the subxyphoid approach was found to be feasible and relatively well tolerated.
A subxyphoid approach for the treatment of persistent or longstanding persistent AF allows for epicardial ablation of the posterior aspect of the LA and LAA exclusion with less potential for atrioesophageal fistulas. The addition of LAA exclusion and isolation of the posterior aspect of the LA combined with PVI begins to emulate the Cox-MAZE III procedure which has been shown to be effective in the treatment of persistent AF[7]. The STAR-AF and Chase-AF trial suggests that PVI alone is as effective as PVI plus additional ablation lines or complex atrial fractionated electrograms for the prevention of recurrence of atrial arrhythmias in patients with persistent AF [8,9]. However, these trials were performed in patients with no prior ablation procedure. In patients that have failed PVI, surgical approach including epicardial LAA exclusion and isolation of the posterior LA combined with PVI has greater freedom from recurrence of AF compared to PVI alone [3]. Additionally, LAA ligation is thought to improve freedom from AF recurrence in nonparoxysmal AF due to electrical isolation of the LAA with elimination of potential LAA triggers, LA debulking resulting in electrical remodeling of the LA and allowing for a more complete ablation procedure[10].
In contrast to the transabdominal transdiaphragmatic or bilateral thoracic approach, the subxyphoid approach is familiar to cardiac surgeons who use this approach for pericardial windows. Subxyphoid pericardial access is also familiar to cardiac electrophysiologists who perform epicardial VT ablations or the LARIAT procedure. Direct visualization of the posterior LA and pulmonary veins via a subxyphoid approach or introduction of a space creating device to separate the esophagous from the posterior LA[11] should potentially decrease the potential of atrioesophageal fistulas and allow for a more consistent approach to isolate the posterior LA and pulmonary veins. The potential of creating an atrioesophageal fistula with endocardial ablation has become a deterrent to many cardiac electrophysiologist in creating ablation lesions on the posterior left atrium.
The subxyphoid approach is particularly attractive for a hybrid epicardial/endocardial ablation approach. The surgeon can visualize the posterior aspect of the left atrium and pulmonary veins to perform posterior isolation of the left atrium and linear posterior lines around the pulmonary veins. The cardiac electrophysiologist can perform limited endocardial ablation to complete the isolation of the pulmonary veins and posterior aspect of the left atrium [Figure 3]. The anterior aspect of the LSPV and RSPV, and roof of the LA may be difficult to access epicardially with the current epicardial ablation tools, thus leaving gaps [Figure 3]. However, anterior endocardial ablation of the anterior aspect of the LSPV and RSPV are easily approachable, as well as producing a LA roof line. Producing epicardial ablation lesions from the transverse sinus will allow for an epicardial LA roof line and anterior access to the LSPV and RSVP. A potential epicardial gap would still persist at the transverse recess which would need to be completed via endocardial ablation or via dissection of the transverse recess. Another advantage of the subxyphoid approach is that the LAA can be ligated under direct visualization. If the LAA cannot be visualized, then the LARIAT procedure can be performed by the standard method via the subxyphoid pericardial access and transseptal catherization[12].
Figure 3.

This report is the first demonstration of the feasibility of the subxyphoid pericardial access approach for epicardial ablation and LAA ligation and its use as a hybrid epicardial/endocardial approach for persistent and longstanding persistent AF refractory to medications and/or endocardial ablations. As technology advances, the possibility of a complete minimially invasive “MAZE” procedure via the subyxphoid access approach or a completely percutaneous “MAZE” procedure should be possible.
R.J. Lee is a consultant and equity holder in SentreHEART, Inc.