Superior vena CAVA Isolation by Cryoballoon in Addition to Pulmonary Vein Isolation in Atrial Fibrillation Ablation Patients. A Randomized Trial. CAVAC AF Trial. Study Rationale and Design
Castro-Urda V1, Segura-Dominguez M1, Jiménez-Sánchez D1, Vela-Martín P1, García-Izquierdo E1, Aguilera-Agudo C1, Veloza-Urrea D1, Morillo-Diaz J1, Pham-Trung C1, Hernández-Terciado F1, De Castro Campos D1, Chichakli Cela M1, Mingo-Santos S2, García Rodriguez D1, Toquero-Ramos J1, Fernández-Lozano I1
1Electrophysiology Unit. Cardiology Service. Hospital Puerta de Hierro. Majadahonda (Madrid). Spain.2Cardiac Imaging Unit. Cardiology Service. Hospital Puerta de Hierro. Majadahonda (Madrid). Spain.
Superior vena cava (SVC) has been considered a specific trigger in atrial fibrillation (AF) development. Cryoballoon SVC isolation seems feasible and safe, and has never been compared in addition to pulmonary vein isolation (PVI) to PVI alone.
A unicenter randomized trial, comparing two ablation procedures is proposed. Cryoballoon SVC isolation in addition to PVI is compared to PVI alone in paroxysmal or non long standing persistent AF patients. Patients from 18-80 years old are included. Patients are excluded if there is a previous AF ablation procedure, transvenous pacemaker or defibrillator (ICD) implanted, severe mitral valve disease, left atrium (LA) anteroposterior diameter > 55mm or LA indexed volume > 48ml/m2 in an echocardiogram performed in the last year, left ventricular ejection fraction (LVEF) < 35%, hypertrophic or restrictive cardiomyopathy.
All patients are provided an Alivecor Kardia Mobile device to record an electrocardiogram (ECG) everyday and in case of clinical symptoms to monitor recurrences.
The primary efficacy end point is defined as any AF/atrial flutter/atrial tachycardia recurrence, with a minimal duration of 30 seconds, registered with surface ECG, Holter ECG or Kardia mobile registry during a 12 months follow up period.
Our study will provide data about the efficacy of SVC isolation in addition to PVI compared to PVI alone in a randomized way, in paroxysmal and non long standing persistent AF patients.
Key Words : .
Víctor Castro Urda. MD
Manuel de Falla, 1. 28232. Majadahonda (Madrid). SPAIN
Pulmonary vein isolation (PVI) is the most important aspect of atrial fibrillation (AF) ablation procedure. Ectopic beats originating in PV can promote AF episodes.. Different types of energy are used in AF ablation procedures, where radiofrequency and cryoablation predominate. Both techniques have been considered equivalent regarding efficacy and safety, although procedure time and technical requirements are different. In spite of technical improvements, experience and better tools during recent years, AF ablation success rate still is suboptimal, especially regarding persistent AF.
Other ablation targets have been proposed in addition to PVI, like left atrial lines, scar homogeneization, rotors, vein of marshall ablation, appendage isolation or extrapulmonary foci.In general, ablation of these targets have failed in achieving greater success rate in AF ablation procedures.
Superior vena cava (SVC) has been considered a specific trigger in AF development and it is implicated in about 30% of extrapulmonary foci according to different studies . It has also been involved in the maintenance and as a substrate in AF episodes. SVC isolation has been considered a different target in AF ablation procedures in order to improve success rates. At present, all the available scientific evidence regarding SVC isolation refers to radiofrequency ablation procedures. Initially, only if it was demonstrated that it behaved as an AF trigger, SVC was isolated. Later on, empirical SVC isolation has been proposed in addition to PVI in AF ablation procedures. Three randomized studies and two meta analysis suggest that SVC empirical isolation in addition to PVI, confer some benefit in AF ablation. This benefit seems to be obtained only in paroxysmal AF and not in persistent forms. No significant differences were found regarding procedures times, fluoroscopy time and complications.
SVC isolation using Cryoballoon is feasible, according to a recent study, in which a third generation balloon was used. 30 patients were included, achieving a 89% isolation success rate and one transient phrenic paralysis was reported.
However, as far as we know, the evidence regarding empirical SVC isolation using cryoballon in addition to PVI compared to PVI procedure is lacking.
This is a unicenter randomized trial, comparing the rhythm control effectiveness of 2 ablation procedures: Cryoablation PVI alone or combined to SVC isolation in paroxysmal or persistent AF patients.
The trial was approved by the institutional review boards of the center. All patients provide written informed consent.
Patients are eligible if they are between 18-80 years old and have paroxysmal or short duration persistent (less than 1 year) AF and are scheduled for an Cryoballoon AF ablation procedure.
Exclusion criteria are shown in [Table 1]:
Table 1. Study exclusion criteria
| Exclusion criteria |
| Age < 18 or > 80 years. |
| Previous AF ablation procedure. |
| Pregnancy or probability of it. |
| Life expectancy < 1 year. |
| Unavailability to understand or consent to participate in the study. |
| Reversible AF causes suspected. |
| Transvenous Pacemaker or ICD previously implanted |
| Permanent AF or long persistent duration (> 1 year). |
| Severe mitral valve disease. |
| Left atrium (LA) anteroposterior diameter > 55mm or LA indexed volume > 48ml/m2 in an echocardiogram performed in the last year. |
| Left ventricular ejection fraction (LVEF) < 35%. |
| Hypertrophic or Restrictive cardiomyopathy. |
| Contraindication to the use of antiarrhythmic drugs. |
| Left appendage thrombus presence in transesophageal echocardiogram at the moment of the procedure. |
| Any contraindication to anticoagulant therapy. |
| No “smartphone” available. |
| To be participating in another clinical trial. |
''
Patients are randomly assigned in a 1:1 ratio to either PVI alone or PVI associated to SVC isolation. Randomization is performed using the method of permuted block randomization. The randomization outcome is communicated to the operator. Patients are blinded to randomization outcome, as are the investigators evaluating adverse events and electrocardiographic data. All investigators are blinded to interim analyses.
100 patients are planned to be included, randomized in a 1:1 ratio. Follow up duration is 12 months. 47 patients are necessary in each branch assuming an alfa error of 5% and statistic power of 80%, to obtain a hazard ratio of 0,54 (data obtained from previous RF meta analysis data). 6 patients will be included in addition, in case of loss of follow up and to increase statistic power of the study. [Figure 1]
Figure 1. Flowchart of the trial.
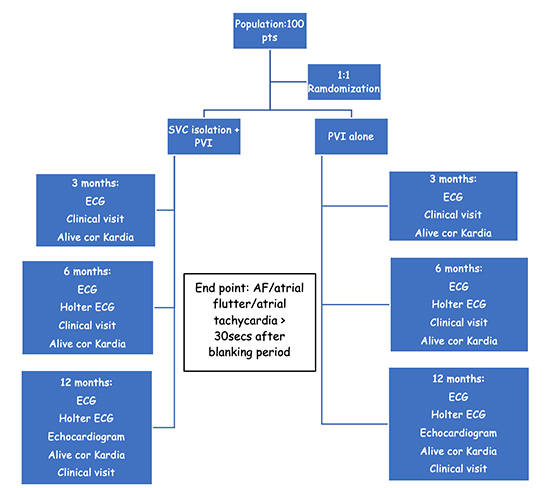
Patients follow up schedule
After ablation procedure three clinical visits are scheduled.
3 months (first visit after ablation). Clinical visit and electrocardiogram (ECG).
6 months. Clinical visit, ECG and 24h Holter recording.
12 months (final visit). Clinical visit, ECG, 24 h Holter recording and echocardiogram.
Unscheduled visits can be performed during follow up, in case of recurrences of symptoms to modify pharmacologic treatment or to schedule redo procedures. [Figure 1]
All patients are provided an Alivecor Kardia Mobile device to record an electrocardiogram everyday, and in case of clinical symptoms. All tracings are downloaded in a smartphone and forwarded to an email address, that is check in a daily basis by a trained nurse and three electrophysiologists as backup. (MSD, VCU, DJS, EGI). A good monitoring adherence is defined by a threshold of 80% monitored days.
Patients will be contacted in case of absence of tracing sending to increase compliance rate.
The primary efficacy end point is defined as any AF/atrial flutter/atrial tachycardia recurrence, with a minimal duration of 30 seconds, registered with surface ECG, Holter ECG or Alivecor Kardia mobile registry during a 12 months follow up period. All the recurrences in the first three months after ablation are considered in the blanking period and are not considered an end point.
The primary safety endpoint is the presence of any procedure related complications during follow up specially phrenic nerve paralysis and sinus node disfunction.
The secondary end points are atrial fibrillation burden (time in atrial fibrillation divided by monitoring time), total mortality, cardiovascular admission rate, stroke, pacemaker implantation rate, AAD necessity after three months, electrical cardioversion, redo procedures, left atrium remodeling (change in left atrial diameter and indexed volume), left ventricular ejection fraction after 12 months follow up, early recurrence of atrial arrythmias (ERAF) defined as those occurring during the blanking period after ablation, % monitoring adherence, procedural and fluoroscopy time and number of cryoballoon applications.
Age, sex, AF classification, cardiopathy, Hypertension, diabetes, smoking status, AF evolution time, LA diameter and volume, AAD previous use, sleep apnea disorder, renal insufficiency are included as variables to predict ablation success.
All cryoablation procedures are performed in a fasting state and under deep sedation. A transesophageal echocardiogram is performed in every patient, previous to vein access, in order to exclude left appendage thrombi. After transseptal puncture bolus heparin (100mg/kg weight) and infusion is administrated to obtain ACT 300-350 seconds.
All procedures are performed with third generation Medtronic Artic Front AdvanceTM Cryoablation Catheter. Pulmonary vein (PV) potentials are recorded with Achieve AdvanceTM circular mapping catheter. According to the protocol in our center, one 180 seconds application is performed if time to isolation is less than 60 seconds, and one 240 seconds application is performed if time to isolation is between 60-100 seconds. If there is no isolation after 100 seconds of application, this is stopped and the balloon is repositioned. No bonus applications are given. In case of lack of pulmonary vein signals, a 180 or 240 seconds application is given depending on the achieved temperature. Phrenic nerve function is monitored during right veins applications with a catheter located in the right subclavian vein. After last application, entrance and exit isolation is checked in all veins. In case of AF rhythm during procedure, and no RS conversion during applications, a biphasic cardioversion is performed before moving catheters to the right side.
In patients assigned to SVC isolation, this is guided by the presence of SVC signals. Time of application is the time necessary to SVC isolation plus 60 seconds. If no isolation occurred after 100 seconds, application is stopped and the balloon is repositioned. Phrenic nerve function is strictly monitored with a pacing catheter located in right subclavian vein and heart rate is monitored in order to exclude sinus node disfunction. In case of absence of SVC potentials, the patient will be excluded, and ordinary visits are scheduled according to the protocol of the center. In patients assigned to control group, procedure is finished after checking the presence of SVC signals.
Procedures will be performed with no anticoagulation suspension and patients will maintain anticoagulation at least two months after procedure. The decision to keep on anticoagulation after this period is based on the CHA2DS2VASc score. Proton pump inhibitors are prescribed during 15 days to avoid oesophageal complications.
After procedure, a 3 months blanking period is stablished, in which antiarrhythmic drugs (AAD) are prescribed in absence of contraindications.
AAD are recommended to be suspended at 3 month clinical visit although they can be maintained or restarted, during follow up, as at the discretion of the treating physician.
During the blanking period, repeated ablation is allowed but would not reset the blanking period.
Ethics and security aspects
The study is conducted in accordance with the guidelines set out in the Standards of Good Clinical Practice (CPMP/ICH/135/95) and the international ethical recommendations contained in the lasted revision of the Declaration of Helsinki and in the Belmont report. The ethics committee of the Hospital Universitario Puerta de Hierro Majadahonda (Madrid. Spain) has approved the protocol. All study data will be recorded, stored, and processed anonymously. All participants will be informed to the fullest extent possible about the study, in a language and terms that are understable. All participants sign an informed consent at enrolment including the name, and date personally by the subject, and by the person who carry out the informed consent communication.
Shapiro Wilks and Kolmogorov Smirnov test are used to test for normality. Continuous data are described as mean SD if normally distributed and as median (interquartile range) for no normal data. Categorical variables are described as counts and percent. Student t test (Mann-Whitney U test if normality not satisfied) and chi-square test are used to compare groups.
Recurrence free survival is compared by the log-rank test, and Kaplan-Meier curves are generated. Event free duration is defined as time from procedure to occurrence of outcome event (arrhythmia recurrence after blanking period). For patients who are event free at the end of follow up, time to event is censored. Death from any cause within the study period is considered for mortality analysis.
Univariate and multivariate Cox proportional hazard models will be used for identifying significant predictors of AF recurrence. Hazard ratios and 95% confidence intervals (Cis) from the Cox model are reported in the results.
All enrolled patients who undergo the index procedure constitute the intent to treat population and are the subject for safety and efficacy analyses.
All test are 2 sided, and a p value < 0.05 is considered statistically significant. Analyses are performed using IBM Statistics SPSS 25 version.
CAVAC AF is a randomized single blind study that compares PVI associated to SVC isolation to PVI alone, in patients with paroxysmal or non long lasting persistent AF. It differs to previous literature in the technique that is employed. All procedures are performed with third generation Medtronic Artic Front AdvanceTM Cryoablation Catheter.
Cryoballoon therapy to treat AF has been shown to be a safe procedure in real world, across a broad cohort of patients with AF . Serious procedure and device related adverse event rates were only 4,7% and 2% in this registry. Cryoballoon has a better learning curve compared to radiofrequency, and shortens procedure time while fluoroscopy and clinical outcomes are comparable.. This reason makes cryoballoon therapy a very attractive technique to treat AF patients.
PVI has shown suboptimal outcomes in patients with AF, especially in those with persistent form. Adjunctive strategies employed to ablate non PV triggers have shown favorable although non reproducible outcomes.
Cryoballoon SVC isolation has been shown to be a simple, safe and efficacy procedure . According to Campal et al, over 30 patients, success rate was almost 90% and no permanent complications were reported. Only two transient complications occurred (one phrenic nerve paralysis and one sinus node injury).
Atrial fibrillation episodes can often be asymptomatic, even after catheter ablation, creating a disconnect between symptoms and actual arrythmia burden which may alter clinical management . Outcome after ablation depends on the time of monitoring. The more time of monitoring the less efficacy results are reported. Therefore, a strict system of monitoring seems necessary in atrial fibrillation ablation trials at this moment.
Recent technological advancements have facilitated ambulatory electrocardiogram monitoring in the outpatient environment providing continuous, high resolution ECG data streams ranging from days to months at a time.
The likehood of AF recurrence detection, after ablation or cardioversion, was 56% significantly greater in patients randomized to AliveCor Kardia daily monitoring compared to usual care, demonstrating that this strategy of monitoring is mostly beneficial for prompt detection of recurrence after ablation.
The trial´s main limitation is represented by the small number of patients included. Nonetheless, study size has been calculated to obtain significant differences between groups.
Regarding monitoring adherence after ablation, a high level of ECG compliance is necessary to detect AF recurrences. A good adherence is defined by a threshold of 80% monitored days.
Another limitation is the single blinded design, therefore an investigational bias can occur to modify procedural aspects in the SVC isolation group.
Our study will provide data about the efficacy of SVC isolation in addition to PVI compared to PVI in a randomized way, in paroxysmal and non long standing persistent atrial fibrillation patients.