Safety of Twice Daily Sotalol in Patients with Renal Impairment: A Single Center, Retrospective Review
Abigail Rabatin1, Melissa J. Snider 1, J. Michael Boyd1, Mahmoud Houmsse1
1
The Ohio State University Wexner Medical Center
452 West 10th Ave
Columbus, OH 43210
.
The class III antiarrhythmic sotalol is renally eliminated with a dose-related propensity to cause adverse drug reactions (ADR) potentially leading to life-threatening arrhythmias. Although product labeling recommends once daily dosing in patients with renal impairment, twice daily dosing is commonly utilized. This study evaluates the safety of this practice.
This retrospective, observational study examined renally impaired patients with atrial fibrillation or atrial flutter admitted for sotalol initiation from July 1, 2012 - December 31, 2014, then for up to 20 months after initiation. Primary endpoints included rates of ADR and therapy changes due to ADR. Secondary endpoints included therapy changes due to arrhythmia recurrence, admissions due to arrhythmia recurrence, and therapy changes for any cause.
Analysis included 134 patients with an average creatinine clearance of 51 ml/min, followed over a median of 170 days. Length of stay averaged 3 days withADR occurring in 53.7% of patients, most commonly QT prolongation or bradycardia. Therapy change due to ADR occurred in 45.5% of patients (n=61). Therapy change due to arrhythmia recurrence occurred in 23.1% (n=31), admission due to arrhythmia recurrence occurred in 24.6% (n=33), and therapy change for any cause occurred in 74.6% (n=100).
Initiating sotalol twice daily in renally impaired patients results in ADR and therapy change rates consistent with rates seen in clinical practice for non-renally impaired patients, with minimal length of stay.This practice may be reasonable when initiated in the acute care setting with subsequent outpatient monitoring, however further study is needed.
Key Words : Sotalol, Renal Impairment, Renal Dysfunction, Atrial Fibrillation, Dosing.
Correspondence to: Melissa J. Snider, PharmD, BCPS, CLS, BCACP1
The Ohio State University Wexner Medical Center
368 Doan Hall
410 West 10th Ave
Columbus, OH 43210
Proarrhythmia and sudden cardiac death are significant concerns linked to the use of antiarrhythmic drugs. Sotalol, a Class III antiarrhythmic agent with non-selective beta-blocking properties, is known to effectively treat atrial arrhythmias[1]. However, its dual mechanism of action may cause adverse drug reactions (ADRs) including bradycardia and QT prolongation, which can lead to life threatening arrhythmias[2].Sotalol does not undergo first pass metabolism, and 80-90% of its elimination occurs by renal excretion of unchanged drug[3]. The half-life varies from 7 -18 hours based on renal function. Sotalol has been shown to have dose-dependent proarrhythmic effects,thus it is essential to consider the renal elimination of sotalol when selecting a dosing strategy.
Atrial fibrillation (AF) and atrial flutter (AFL) are two of the most common arrhythmic disorders, leading to significant morbidity and mortality worldwide[4]. Yet, for patients with renal impairment, there is limited data regarding use of antiarrhythmic drugs (AAD). For sotalol specifically, many studies evaluating its safety exclude patients with renal impairment, or do not disclose creatinine clearance (CrCl) of subjects. Studiesincluding renally impaired patients with a CrCl 40-60 ml/min utilized a once dailydosing schedule, with doses ranging from 80-160mg[5,6]. Therefore, the product labelling for sotalolrecommends once daily dosing (specifically 80mg once daily, titrated up to 160mg once daily as tolerated) when sotalol is used to treat AF or AFL for patients with a CrCl of 40-60 mL/min. The product label also recommends monitoring patients within an inpatient setting for the first five doses when starting sotalol[7,8] .However, the ACC/AHA/HRS AF guidelines have differed on their recommendations for sotalol initiation. In 2011, the guidelines recommended outpatient initiation of sotalol in patients with little or no heart disease, QT interval <450 ms, and minimal risk factors for proarrhythmia. The 2014 ACC/AHA/HRS AF Guidelines acknowledge that practice patterns vary widely. Authors note while inpatient initiation should be considered given the package insert warning, there is considerable experience with outpatient initiation as well and the initiation strategy should be individualized.
Admission for initiation of an AAD can be a costly endeavor for the patient and health system. A two to three day admission for sotalol initiation may cost over $3,000 [9],
. Patients with renal impairment receiving once daily dosing of sotalol would need to be hospitalized for five days, resulting in a greater cost burden. Due to these concerns as well as clinical experience, patients with a CrCl 40-60 ml/min are often initiated on sotalol twice daily in an acute care setting. This allows patients to remain monitored during their first five doses, while decreasing length of stay. This practice commonly occurs at The Ohio State University Wexner Medical Center (OSUWMC). Patients are often discharged on twice daily sotalol with follow-up in the OSUWMC AAD clinic. Patients with renal impairment presenting to clinic on twice daily dosing are considered for conversion to once daily dosing based on package insert recommendations and/or presence of ADRs.
Although twice daily initiation dosingof sotalol is common in renally impaired patients, a formal evaluation of safety has not yet been conducted. Little is known concerning outcomes of this dosing strategy, or the effect of dose changes in the outpatient setting.The purpose of this study is to describe the safety of sotalol, initiated in the acute care setting at a twice daily dose in patients with renal impairment, during their inpatient stay and outpatient course.
This retrospective, observational study evaluated patients with AF/AFL admitted for initiation of sotalol from July 1, 2012 through December 31, 2014 at the Richard M. Ross Heart Hospital, a 172-bed cardiovascular hospital at OSUWMC (Columbus, OH). Data werecollected on patients seen up to 20 months after sotalol initiation to characterize presence of and response to ADRs, as well as arrhythmia control. Upon discharge, information in the ambulatory setting was obtained from notes written by electrophysiology, cardiology, or AAD clinic visits. The AAD clinic is a pharmacist-run, physician-supervised service and aims to monitor and assess antiarrhythmic therapy [10]. The study was granted exemption from review through the OSUWMC Institutional Review Board.
Participants were identified by a computer generated list of patients with an order for sotalol while admitted to the Ross Heart Hospital with concomitant diagnosis of AF or AFL. Patients were then further evaluated by manual chart review to ensure the sotalol prescription identified was the patient’s first. During admission, sotalol dose was determined based on provider discretion and patient’s QT response after the first dose. All patients continuing sotalol upon discharge were in sinus rhythm at the time of discharge. Patients were eligible for inclusion if all of the following criteria were met: age 18-89 years of age, indication for sotalol of AF or AFL, renal impairment (defined as CrCl 40-60 ml/min) upon admission for sotalol initiation, and sotalol initiated at twice daily. Pregnant women and prisoners were excluded, as well as patients receiving sotalol for other atrial or ventricular arrhythmias. To provide continuity throughout the study, all CrCl calculations were based on clinic protocol. For patients over age 65, if serum creatinine was <1.0, it was rounded to 1.0.Adjusted body weight was utilized in the Cockroft-Gault equation for those with an actual body weight greater than 20% over their ideal body weight.
The primary endpoints of this study included[1] describing the overall proportion of patients who experience ADRs, and[2] the proportion of therapy changes (dose adjustments, drug discontinuations, and additional interventions such as device implantation) related to ADRs. Specific ADRs included: bradycardia (heart rate <50), QT interval prolongation (QT interval >500 ms or >550 ms if ventricular pacing), incidence of torsade de pointes, or other ADR resulting in significant symptoms. Secondary endpoints included proportion of patients with therapy changes due to arrhythmia recurrence, admissions due to arrhythmia recurrence, and therapy changes for any cause (e.g. CrCl).Arrhythmia recurrence was defined as documentation of recurrent AF/AFL after initial discharge requiring intervention such as dose adjustment, or admission for intervention (direct current cardioversion, ablation, or therapy change). Those who failed sotalol at the time of initiation due to ineffectiveness were not included in this definition.
Patients were evaluated for the above pre-specified endpoints during 1) hospitalization for sotalol initiation, 2) “1-month clinic visit” occurring 0-3 months after initiation, 3) “6 month clinic visit” occurring 4-8 months after initiation, 4) “12 month clinic visit” occurring 9-14 months after initiation, and 5) “18 month clinic visit” occurring 15-20 months after initiation. For patients who did not have clinic follow-up at every time point but continued on sotalol therapy, dosing data from the prior visit was carried forward. For these patients, it was presumed no significant adverse events or arrhythmia events occurred prompting evaluation.
Baseline demographic and disease-related characteristics were compared across two dose groups (80 mg twice daily and 120 mg twice daily) using t-test, Fisher's exact, and chi-square tests as appropriate. The prevalence of adverse effects leading to changes in therapy, including dosage adjustment, drug discontinuation, and additional intervention, was calculated separately at discharge and post-discharge for the two dose groups. Chi-square tests were performed for comparison. Specific ADRs were described. A two-sided significance level of α=0.05 was used for all tests. Analyses were performed in SAS version 9.3 (SAS institute, Cary, NC).
There were 1,202 patients identified with an active prescription for sotalol during admission from July 1, 2012 through December 31, 2014. After review, 1,068 patients were excluded . The most common reasons for exclusion were CrCl > 60 ml/min (n=664) and sotalolinitiation occurring prior to study period (n=327).Therefore 134 patients were included as initiated on sotalol twice daily during the study period, with 63.5% (n=85) started on sotalol 80 mg twice daily , 35% (n=47) on 120 mg twice daily, and 1.5% (n=2) on 40 mg twice daily. Patients taking 40 mg twice daily were excluded from statistical comparison between these groups, but included in the overall evaluation. The majority of patients were white females, with an average age of 71.9 years and an average CrCl of 51 ml/min [Table 1]. Characteristics were well matched between the two dosing groups, with the exception of age and CrCl.Patients were followed for a median of 170 [IQR: 7-560.75] dayswith 15 patients lost to follow-up after discharge. Length of stay was similar between the two groups, approximately 3 days.
Table 1. Baseline characteristics
|
Total N=132 |
80mg BID n=85 |
120mg BID n=47 |
P-value |
| Age (years), mean ± SD |
71.9 ± 7.1 |
73.5 ± 7.1 |
69.0 ± 6.1 |
0.0004 |
| Male, n (%) |
47 (35.6%) |
35 (41.2%) |
12 (25.5%) |
0.072 |
| BMI(kg/m2), mean ± SD |
29.4 ± 6.9 |
28.8 ± 6.9 |
30.4 ± 6.8 |
0.21 |
| White, n (%) |
122 (93.1%) |
77 (91.7%) |
45 (95.7%) |
0.90 |
| SCr (mg/dl), mean ± SD |
1.14 ± 0.21 |
1.1 ± 0.2 |
1.1 ± 0.2 |
0.51 |
| Number of patients with SCr rounded to 1, n (%) |
58 (44%) |
38 (45.2%) |
20 (43.5%) |
0.94 |
| Calculated CrCl (ml/min), mean ± SD |
50.9 ± 6.5 |
49.5 ±6.6 |
53.3 ± 5.6 |
0.0013 |
| LVEF >40, n (%) |
110 (83.3%) |
72 (84.7%) |
38 (80.9%) |
0.82 |
| Bundle branch block, n (%) |
20 (15.2%) |
10 (11.8%) |
10 (21.3%) |
0.14 |
| Device, n (%) |
56 (42.4%) |
34 (40%) |
22 (46.8%) |
0.45 |
| Permanent pacemaker |
44 (33.3%) |
27 (31.7%) |
17 (36.1%) |
0.84 |
| ICD |
9 (6.8%) |
5 (5.9%) |
4 (8.5%) |
…* |
| Bi-ventricular ICD |
3 (2.2%) |
2 (2.3%) |
1 (2.1%) |
…* |
| No Device |
76 (57.6%) |
51 (60%) |
25 (53.19%) |
0.32 |
| AARx clinic follow-up, n (%) |
38 (28.8%) |
24 (28.2%) |
14 (30.0%) |
0.85 |
| Length of Stay (days), mean ± SD |
3.09 ±0.03 |
3.08±0.02 |
3.04±0.04 |
0.45 |
| Total Follow up (days), mean ± SD+ |
309.5 ±353.5 |
318.9 ± 362 |
292.5 ± 340.8 |
0.69 |
| Baseline HR (bpm), mean ± SD |
86.5 ± 27.6 |
86.7 ± 27.6 |
86.1 ± 27.9 |
0.92 |
| Baseline QRS (msec), mean ± SD |
99.2 ± 29.6 |
98.3 ± 31.6 |
101 ± 25.9 |
0.61 |
| Baseline QT (msec), mean ± SD |
390.5 ± 58.5 |
390.8 ± 63.7 |
389.9 ± 48.3 |
0.93 |
| Baseline QTc (msec), mean ± SD |
455.8 ± 39.7 |
454.2 ± 38.4 |
458.6 ± 42.1 |
0.55 |
| AF/AFL upon admission, n (%) |
66 (50%) |
41 (48.2%) |
25 (53.2%) |
0.586 |
*Unable to calculate, +Range: 1-600days, AARx= Antiarrhythmic medications; BBB= bundle branch block; CrCl= Creatinine clearance; HR= heart rate; ICD = implantable cardioverter-defibrillator; LVEF = left ventricular ejection fraction; SCr= Serum creatinine
Overall, 53.7% (n=72) patients experienced at least one ADR throughout the study. The most common were QT prolongation (27.6%) and bradycardia (20.2%). There were no reports of torsades de pointes or death due to ventricular arrhythmias. ADRs were further separated into occurrence during admission and post-discharge [Table 2]. While admitted, 37.3% (n=50) of patients experienced at least one ADR with no difference between patients initiated on120 mg twice daily versus 80 mg twice daily (42.6% vs 34.1%, p = 0.34). At time of discharge, 74.6% (n=100) of patients continued on sotalol, and of those, 22% (n=22) experienced ADRs after dischargewith no difference between ADR rate in patients discharged on 120mg versus 80 mg twice daily (19% vs 25%, p = 0.57).
Table 2. Adverse drug reactions on sotalol therapy in renally impaired patients
| ADR, % (n) |
Total (N=134) |
Inpatient course (N=134) |
Outpatient course (n=100) |
| Total ADRs |
53.7% (72) |
37.3% (50) |
22% (22) |
| 1 ADR |
40.1% (54) |
27.5% (37) |
17% (17) |
| 2 or more |
13.6% (18) |
9.8% (13) |
5% (5) |
|
|
|
|
| QT prolongation |
27.6% (37) |
23.1% (31) |
6% (6) |
| Bradycardia |
20.2% (26) |
15.9% (21) |
5% (5) |
| Fatigue |
4.3% (5) |
0 |
5% (5) |
| Dizziness |
2.6% (3) |
0 |
3% (3) |
| SOB |
4.3% (5) |
3% (4) |
1% (1) |
| HF exacerbation |
1.5% (2) |
1.5% (2) |
0 |
| Other |
7% (9) |
3% (4) |
5% (5) |
ADR= Adverse Drug Reaction
Therapy change due to ADR occurred in 45.5% (n=61) of patients throughout the study period with no difference inADR rate betweenpatients on 120mg versus 80 mg twice daily (51.1% vs 41.2%, p = 0.27)and occurring more often inpatient than outpatient [Figure 1]. Therapy change due to ADR during admission for sotalol initiation occurred in 34.3% (n=46) of patients,more often in patients initiated on 120 mg twice daily versus 80 mg twice daily (42.6% vs 29.4%, p=0.10). Of the 100 patients discharged on sotalol, following initiation, therapy change due to ADR for occurred in 15% (n=15) of patients, with no difference between patients on 120mg versus 80mg twice daily (15.8% vs 11%). Outpatient ADRs prompting therapy change included: QT prolongation in 6% (n=6), fatigue in 3% (n=3), bradycardia in 3% (n=3), and other in 3% (n=3).
Figure 1. Types of Therapy changes due to ADR on solatol twice daily
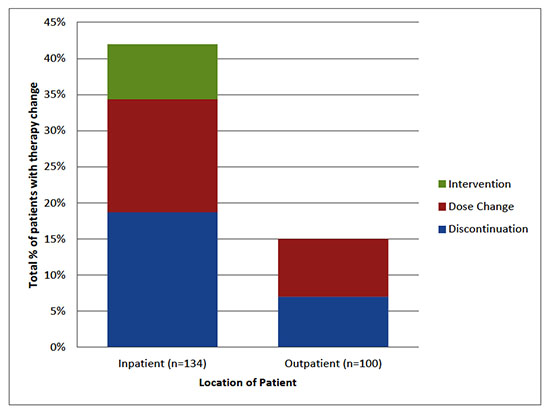
Therapy change due to any cause occurred in 74.6% (n=100) of patients, some with more than one change, including drug discontinuationin 59.7% (n=80), dosage adjustment in 19.4% (n=26), and additional intervention in 9.7% (n=13).Therapy change during admission for sotalol initiation occurred in 47.7% (n=64) of patients with a higher rate noted in patients initiated on 120 mg versus 80 mg twice daily (63.8% vs 40%, p=0.0087).
Therapy changes due to arrhythmia recurrence occurred in 23.1% (n=31) of patients with recurrence less common in patients on 120mg versus 80mg twice daily (4.5% vs. 11.2%).Admission related to arrhythmia recurrence occurred in 24.6% (n=33) of patients for a total of36 admissions. During admission additional rhythm control strategies were utilized such as direct current cardioversion or radiofrequency ablation.
Of the 100 patients discharged on sotalol, 38% (n=38)subsequently established in theAAD clinic.Of those that did not, reasons included: 18% (n=18) discontinued drug before scheduled visit, 17% (n=17) had no appointment, 12% (n=12) cancelled appointment and 15% (n=15) were lost to follow-up. Dose frequency change occurred for 21% (n=8) of these outpatients in the AAD clinic, in these casessotalol was adjusted to once daily. One subsequently discontinued sotalol due to arrhythmia recurrence.
Continuation of sotalol therapy was low overall, with 31.3% of the initial 134 patients (n=45) remaining on drug at the end of the20 month study period.Most common reasons for sotalol discontinuation included: ADR in 23.8% (n=32), therapy ineffectiveness in 17.2% (n=23), stopped after ablation 5.2% (n=7), and worsening renal function in 5.2% (n=7) of patients.
This study establishes a point of reference for the safety and effectiveness of twice daily sotalol in patients with renal impairment in clinical practice.Over half of renally impaired patients started on sotalol twice daily experienced at least one ADR, most commonly bradycardia and QT prolongation. Length of inpatient stay remained minimal with more ADRs and therapy changes occurring in the first days of therapy.Patients on higher sotalol doses experienced more therapy changes during admission anddose changes due to ADR.
Despite dose adjustments and ADRs reported, starting sotalol at twice daily dosing in renally impaired patients did not result in increased length of stay for patients started on either high or lower doses. In order to receive appropriate monitoring utilizing once daily dosing and five doses, length of stay would have been five days at minimum, compared to average of three days seen in this study. Therefore, twice daily dosing did minimize length of stay.
During sotalol initiation, several studies report the rate of discontinuation due to ADRs. One randomized controlled trial of patients with paroxysmal AF/AFL, evaluatedsotalol vs placebotolerability over the course of 12 months. Sotalol was given once daily in patients with CrCl 40-60 mL/min (20% of patients) at doses of 80 mg, 120 mg and 160 mg and twice dailyto non-renally impaired patients. Investigators found discontinuation due to adverse events was dose dependent, occurring in 12%, 18%, and 29% of patients in the 80, 120, and 160mg dose groups (6). Another study of patients admitted for antiarrhythmic drug initiation noted 18% of patients on sotalol experienced a cardiac ADR significant enough to result in discontinuation or intervention (5).Sotalol dose was at the discretion of the treating physician, but most patients received 80mg twice daily. These findings are consistent with the 25.4% discontinuation rate and 18.7% discontinuation rate due to ADR specifically during admission for drug initiationin our study of renally impaired patients on twice daily sotalol.
The incidence of ADRs on sotalol therapy during initiation in non-renally impaired patients has been studied. Agusala et al. evaluatedpatients (average GFR of 78.2 ml/min) admitted for initiation of sotalol and dofetilide, looking at risk prediction for ADRs. Of 227 patients treated with sotalol, 43.5% experienced bradyarrhythmia and 56.9% QTc prolongation(defined as increase in 15% from baseline; or exceeding 500 ms) during admission for initiation.Agusala and colleagues were not able to identify any significant predictors of ADRs. The authors concluded ADRs are common during initiation and that these results establish need for close inpatient monitoring [11].The rates of bradycardia and QT prolongation during admission in our study of renally impaired patients (15.9% and 23.1%, respectively) are lower than that reported despite twice daily dosing strategy. Furthermore, sotalol product labeling describes bradycardia incidence of 13.1% [3]. The outpatient rate of bradycardia in our study was lower (5%).
There is currently a lack of literature evaluating outpatient sotalol therapy changes due to ADRs. In theory, the extended half-life of sotalol in renal impairment may result in delay in achieving steady state concentrations by the time of discharge, even after 5 doses. Despite this concern, we found only 15% of patients discharged on sotalol required therapy change due to ADRs in the outpatient setting.
Sotalol therapy is known to have shortcomings in maintaining sinus rhythm. The Sotalol Amiodarone Atrial Fibrillation Efficacy Trial (SAFE-T) assessed amiodarone versus sotalol for treatment of AF, following 655 patients for a minimum of 1 year.Amiodarone and sotalol were equally effective in converting AF to sinus rhythm; however, amiodarone was superior in maintenance of sinus rhythm. On average, 60% of patients in the sotalol group experienced recurrence of AF within 1 year. Weekly rhythm assessments were conducted telephonically [12]. Although our study was not designed to detect all AF recurrence, we found comparable results with 40% experiencing arrhythmia recurrence (based on our definition) and 23.5% had arrhythmia recurrence prompting therapy change over the 20 month study period.Sotalol was discontinued in this population at a high rate of 59.7% with twice daily dosing of sotalol.
During outpatient course, dose frequency change occurred in 21% of patients following in the AAD clinic, with only one patient experiencing arrhythmia recurrence subsequently.Although the low volume of patients makes the safety of this practice difficult to assess, considering the overall number of therapy changes that occurred in our study population, this does not demonstrate a significant trend.
There are several limitations to our study. This study was a single-center, retrospective chart review. Our study was not initially designed to compare the 80mg and 120mg twice daily dosing groups, thus was underpowered to do so . Additionally, the number of patients in the two groups was uneven. Variation in CrCl calculation among practitioners and individual patient characteristics likely impacted dose selection. Confounding variables were not accounted for in assessment, including concomitant medications (such as additional QT-prolonging drugs, or negative inotropes such as non-dihydropyridine calcium channel blockers orbeta-blockers), RR and QT intervals, electrolytes, and device pacing which may have which may have contributed to ADRs .Serum sotalol levels were not measured directly, so the impact of dosing changes on therapeutic drug concentrations is unknown though assessment of changes in the QT interval serves as a surrogate estimate of serum sotalol levels. Another limitation is only 38% discharged on sotalol presented for follow up in the AAD clinic in this study, making it difficult to assess safety in the other 62% after discharge. Lastly, safety and efficacy were not compared directly to once daily dosing in this practice setting.
This study serves to establish a point of reference for the safety of twice daily sotalol in patients with renal impairment.A prospective assessment with a larger sample size, analysis of other contributing risk factors for ADRs, and standardized outpatient monitoring is warranted.
Initiating sotalol twice daily in renally impaired patients results in ADR and therapy change rates consistent with rates seen in clinical practice for non-renally impaired patients, with minimal length of stay. Inpatient monitoring of this practice may be reasonable, further analysis of this patient population in a randomized, controlled study is needed.