Catheter ablation is a well-established treatment option for patients with atrial fibrillation (AF). AF ablation techniques and technologies have improved greatly over the past 15 years;nevertheless,AF ablation remains a technically challenging procedure with room for improvement in both efficiency and efficacy[1].Reported procedure times arestill lengthy,[2-4] and fluoroscopy exposure remainsa concern for patients, physicians, and electrophysiology (EP) lab staff. Innovative strategies and technologies that increase efficiency while maintaining or optimizing efficacy and safety are thus welcomed by the EP community.
Recent years have seen significant improvements in catheter technology and electroanatomic mapping (EAM) systems. Contact force (CF)-sensing catheters have improved procedural efficiency and efficacy[5].Meanwhile, improvements to EAM systems now allow objective measurement of catheter stability and CF during radiofrequency (RF) energy delivery. As a consequence of these latest technological advances,operators are challengedwitha paucity of data on how to better incorporate them into an effective, efficient, and safe workflow.
The objective of this study is to describe a single-center experience witha workflow developed to integrate and optimize the use of new CFcatheter technologyand compare it with prior technologies.Our goal is toprovide a strategy to help minimize the learning curve required for full incorporation of the new technologies, such that procedural efficiency and effectiveness are optimized, and safety is uncompromised.
This study represents the experience of a real-world paroxysmal atrial fibrillation (PAF) population presenting for an index catheter ablation at a single high-volume EP practice between July 2013 and June 2016. All patients were treated according to standard clinical practice, and were ablated by an operator experienced in both RF and cryoballoon ablation (>100 cases with each technology). Baseline patient characteristics, procedural details, and acute outcomes were collected for all ablations during the study period. Effectiveness outcomes at 12 months were collected systematically beginning inJuly 2014.
Procedural efficiency and safety outcomes were analyzed for ablations performed under the new workflow utilizing the ThermocoolSmarttouchCatheter(Biosense Webster, Inc.) (ST) and CartoVisitag Module (Biosense Webster, Inc.) (Visitag), and were compared with prior and concurrent ablations at the same site utilizing the ThermocoolSF Catheter (Biosense Webster, Inc.)(SF) or the second-generation cryoballoon, the Arctic Front Advance Cardiac Cryoablation Catheter (Medtronicplc).Local institutional review board approval was obtained.
We analyzed the prospectively collected data of all adult PAF patients with index catheter ablations performed by two operators at our center during the 3-year study period. Patients with prior catheter or surgical ablations were excluded from the analysis. Cohorts were defined based on the catheter technology utilized in these procedures. RF ablations were performed with the SF catheter through April 2014, at which time the ST catheter was adopted. Cryoablation (CA)procedures were performed with no technology changebetween July 2013 and March 2016. Data were extracted for the SF and CA cohorts at the end of 2016, and for the ST cohort at the end of 2017. The selection of RF energy versus CA for the index ablation was at the discretion of the operator and shared with the patient.
After obtaining patient informed consent, general anesthesia with endotracheal intubation was administered. The procedure was performed with uninterrupted oral anticoagulation. Most patients were on direct oral anticoagulants (DOACs), which were continued the morning of the procedure without a single dose interruption. For patients on warfarin, INR >2.0 was targeted. A pre-procedure transesophageal echocardiogram was obtained to rule out left atrial appendage thrombus.
A decapolar deflectable catheter was placed in the coronary sinus (CS).Intravenous heparin was administered before and after transseptal catheterization to target an activated clotting time of > 350 seconds. Transseptal catheterization was performed with a Fast-Cath SL2preformed sheath (St. Jude Medical, Inc.) and a Brockenbrough needle, and guided by intracardiac echocardiography (ICE)with minimal or nofluoroscopy, as previously described[6].Whentransseptal access was achieved, a J-tipped wire was advanced to the left superior pulmonary vein (PV), guided by ICE,and the trajectory of the wire across the fossa ovalis was marked with Cartosound Module(Biosense Webster, Inc.) technology. The ablation catheter was then advanced to the left atrium (LA), guided by ICE and Cartosound System, viathe same access site as the initial transseptal access.
The LA geometry and voltage were acquired with theCarto(Biosense Webster, Inc.) system using parameters designed for rapid acquisition of geometric and voltage data, as follows:
1. For patients in sinus rhythm, atrial pacing was performed from the CS at 500 ms during geometry and voltage acquisition. For patients in AF,cardioversion was performed beforehand.
2. A Lasso2515 or PentarayCatheter (Biosense Webster, Inc.)was used to create the LA geometry and acquire voltage data with fast anatomic mappingin the Confidense Module ofCarto3 System Version 4 (Biosense Webster, Inc.).
3. For rapid data acquisition, ventilator parameterswere set at 16 breaths per minute ata 1:4 ratio of inspiration to expiration.
The CartounivuModule(Biosense Webster, Inc.) (Univu) was used to integrate still fluoroscopy images as the background for the EAM system. A quadripolar catheter was tied to a standard esophageal temperature probe with silk 2:0 and inserted in the esophagus to monitor location and temperature without fluoroscopy. Wide area circumferential ablations (WACA) around ipsilateral PVs were performed for patients undergoing RF ablation with the SF or ST catheters.
blation workflow for ST ablations
In the ST cohort,the followingworkflowwas followed to optimize the use of the CF and mapping technologies for:
T1. prospective tagging of lesions,
2. real-time monitoring of impedance and CF, and
3. validation of lesion sets.
Initial Visitagparameters included only the stability of 2.5 mm maximum range and 4 seconds minimum timeto quickly obtain tags. Ablation was performed at 40 watts throughout the lesion set. When ablating the posterior wall, RF time was decreased at the operator’s discretion. During ablation, the CF goal was 10 to 20 grams, and Visitagautomatically generated ablation tags for each application meeting the stability parameters. The catheter was moved every 15-20 seconds in the anterior aspects of the ipsilateral veins and every 10-15 secondsin the posterior wall, regardless of whether a tag was obtained. Parameters were chosen to avoid inappropriately prolonging RF delivery while waiting on the appearance of a lesion tag.
To validate the lesion set after PVisolation [Figure 1a], force over time (FOT) assessment was used to filter and display only tags for locations where a minimum force of 10 grams was maintained for at least 50% of the time[Figure 1b]. All visual gaps where the CF target was not met were targeted for touch-up. At the end of the procedure, the complete pulmonary vein isolation (PVI) lesion set met stability and CF goals [Figure 1c].
Figure 1a. Procedural Workflow for Pulmonary Vein Isolation with Contact Force Sensing in AF Ablation (a) After Isolation
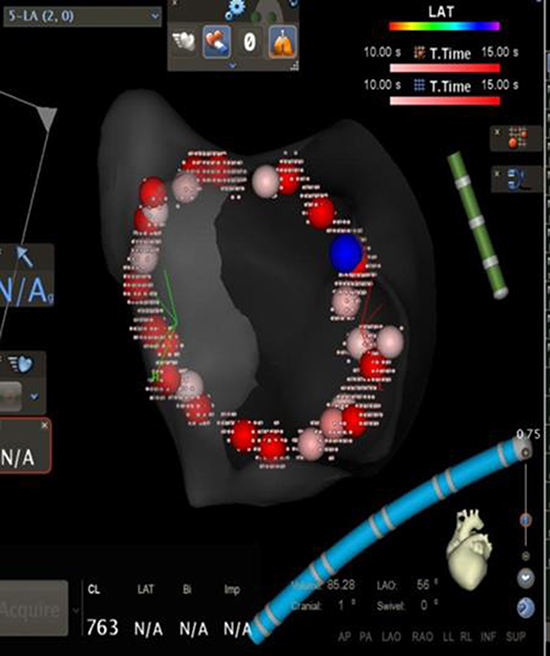
WACA lesion set after PV isolation without force filters; the blue tag denotes isolation
Figure 1b. Procedural Workflow for Pulmonary Vein Isolation with Contact Force Sensing in AF Ablation(b) FOT 50% at 10g
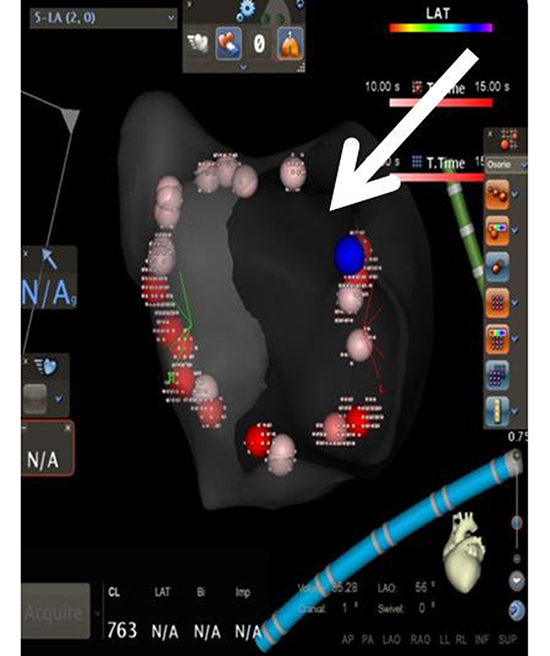
The same lesion set after FOT was used to remove lesion tags where a force >10 grams was not maintained for at least 50% of the time; the arrow shows a gap denoting return of conduction
Figure 1c. Procedural Workflow for Pulmonary Vein Isolation with Contact Force Sensing in AF Ablation (c) After Touch-up, FOT 80% at 10g
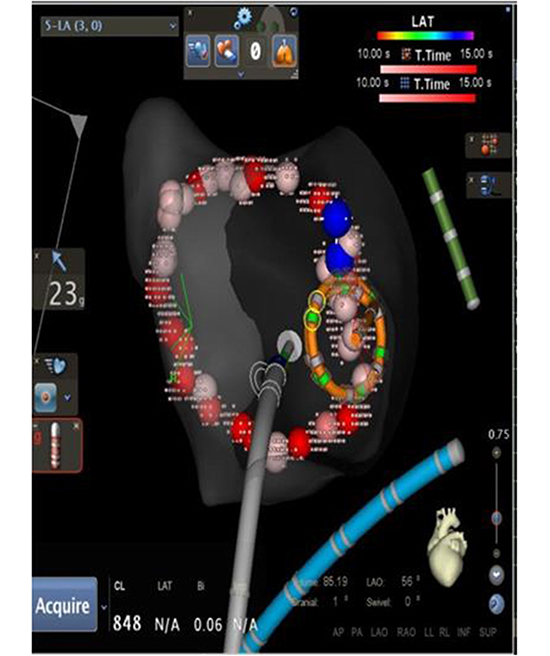
WACA lesion set after touch-up using the FOT filter shows that stable and adequate force was achieved in the entire lesion set
Ablation with SF catheter
The SF ablation procedure was similar, including stability and time criteria for Visitag.The ablation was continued until the local electrogram was abolished or significantly reduced. Univu was not available for the entire span of this cohort.
CA procedures were performed by the standard technique using a second generation cryoballoonwith uninterrupted oral anticoagulation. Single transseptal access was obtained using an SL2 preformed sheath and a Brockenbrough needle guided by fluoroscopy. Neither ICEnor EAM were used. A quadripolar catheter was used to pace the phrenic nerve during ablation of the right-sided veins. The Achieve(Medtronic, plc.) multipolar catheter was used to cannulate the veins and confirm PVI.
PV occlusion was confirmed with pressure monitoring and, if needed, angiography. All veins were isolated using a minimum oftwo freeze-thaw cycles, with freeze durations ofat least three minutes. After isolation of a vein, a bonus freeze was always performed for three minutes.
Assessing acute procedural success
After PVI was confirmed in all cohorts,and after all visual gaps in the VisitagFOT tags were ablated in theST cohort, adenosine 18 mg was injected intravenously to assess dormant conduction. If dormant conduction was detected, further ablation was performed until the PVs were re-isolated. Thereafter,continuous isoproterenol infusion was administeredat up to 20 mcg/min to identify any PV reconnectionor non-PV triggersfor a minimum of 20 minutes after the last ablation lesion. If non-PV triggers were identified, they were targeted foradditional RF ablation.
Follow-up visits were performed at 3 and 12 months,with a96-hour Holtermonitoring at 6 and 12 months after the ablation. Event monitors were used as needed for patients with symptoms. Any hospitalizations or unscheduled office visitsthat documented an atrial arrhythmia recurrence were also captured. Though a comprehensive clinical follow-up schedule was in place throughout the study period, standardized data collection forms were only used to capture the effectiveness detail systematically for the ST cohort. Earlier ablation follow-up, including all of the SF and most of the CA ablations, did not capture recurrence information comprehensively or cumulatively.
The primary study endpoints were the procedural efficiency measures of total procedure time and fluoroscopy duration and dose. Procedure time was defined as time from vascular access toremoval of catheters. The safety endpoint was occurrence of anyserious procedure-related complication, including pericardial effusion requiring intervention, periprocedural stroke, death, need for emergent thoracic surgery, oran acute vascular complication. The key effectiveness endpoint wassurvival time without repeat ablation. A secondary effectiveness endpoint, captured only for the later procedures, was treatmentsuccess at the 12-month visit. The 12-month treatment success criteriawas defined as freedom fromany atrial arrhythmia lasting >30 seconds within 3-12 months after the index ablation without antiarrhythmic drugs (AADs),or with an AAD that was previously ineffective or became effective at a lower dose than required previously.
Survival analysis modeling methodology, including Kaplan-Meier and Cox regression, was used to model time to repeat ablation by cohort. Product limit estimates of the proportion of patients who did not requirea repeat procedure were calculated through one year post-ablation for each cohort with Kaplan-Meier survival models. Cox regression models were used to test for the significance of additional patient and procedural characteristics in explaining reablation rates, as well as for calculating hazard ratios to compare reablation risk by cohort. The characteristics tested for significance included age, sex, CHA2DS2-VASc score, congestive heart failure, LA size and volume, left ventricular ejection fraction (LVEF), operator, procedure time, and the ability to pharmacologically induce latent arrhythmia.
All statistical analyses in this study were performed using SAS software, Version 9.2 (SAS Institute, Inc., Cary, NC). All data were de-identified and accessed in compliance with the Health Insurance Portability and Accountability Act.
A total of 323 patients had an index ablation for PAF during the study period and were eligible for inclusion in the analysis. An additional 38 procedures performed within the study period were excluded from the analysis population due to missing catheter information. The RF cohorts consisted of 32 patients with SF ablation from July 2013 to March 2014, followed by 232 patients with ST ablation from April 2014 through June 2016. The CA cohort consisted of 59 patients with ablation dates from July 2013 to March 2016.
The patient population comprised 50% males, with significantly more females (73%) in the CA cohort (chi-square p=0.0006) compared with the SF or ST cohorts (53% and 55%). The mean age was 63 years with similar age distributions across cohorts. CHADS2 scores were also similar across cohorts, averaging 1.4 for the SF and CA cohorts and 1.3 for the ST cohort. Mean CHA2DS2-VASc risk scores were slightly higher in the CA cohort due to the higher percentage of females in this cohort,while prevalence rates of individual comorbidities, LVEF, and LA size(parasternal long axis view) were similar across the three cohorts [Table 1].
Table 1. Baseline Patient Characteristics
| SF (N=32) |
ST (N=232) |
CA (N=59) |
| n |
Percent |
n |
Percent |
n |
Percent |
| Age (mean, SD) |
63.8 |
11.5 |
62.9 |
10.9 |
63.7 |
12.6 |
| Male Sex*** |
17 |
53.1 |
128 |
55.2 |
16 |
27.1 |
| Left Ventricular Ejection Fraction (%) |
| <35% |
2 |
6.3 |
9 |
3.9 |
0 |
0.0 |
| 35-44% |
3 |
9.4 |
14 |
6.0 |
1 |
1.7 |
| 45-54% |
4 |
12.5 |
25 |
10.8 |
9 |
15.3 |
| 55% and higher |
15 |
46.9 |
116 |
50.0 |
35 |
59.3 |
| Unknown |
8 |
25.0 |
68 |
29.3 |
14 |
23.7 |
| Left Atrial Size, cm (mean, SD) |
3.8 |
0.7 |
3.9 |
0.8 |
3.9 |
0.7 |
| Medical History |
| Congestive Heart Failure |
5 |
15.6 |
26 |
11.2 |
9 |
15.3 |
| Hypertension |
22 |
68.8 |
154 |
66.4 |
44 |
74.6 |
| Diabetes |
5 |
15.6 |
50 |
21.6 |
9 |
15.3 |
| Stroke/Transient Ischemic Attack |
4 |
12.5 |
19 |
8.2 |
6 |
10.2 |
| Vascular Disease* |
13 |
40.6 |
57 |
24.6 |
22 |
37.3 |
| Renal Disease* |
3 |
9.4 |
7 |
3.0 |
6 |
10.2 |
| Liver Disease |
0 |
0.0 |
2 |
0.9 |
0 |
0.0 |
| Pre-ablation Medications |
| Antiarrhythmic drugs |
23 |
71.9 |
145 |
62.5 |
50 |
84.8 |
| Beta-blockers |
18 |
56.3 |
131 |
56.5 |
24 |
40.7 |
| CHA2DS2-VASc Score |
| 0 |
0 |
0.0 |
17 |
7.3 |
2 |
3.4 |
| 1 |
6 |
18.8 |
52 |
22.4 |
11 |
18.6 |
| 2 |
9 |
28.1 |
58 |
25.0 |
11 |
18.6 |
| 3 |
6 |
18.8 |
49 |
21.1 |
10 |
17.0 |
| 4 |
8 |
25.0 |
30 |
12.9 |
13 |
22.0 |
| 5 |
1 |
3.1 |
18/ |
7.8 |
6 |
10.2 |
| 6 |
2 |
6.3 |
5 |
2.2 |
5 |
8.5 |
| 7 |
0 |
0.0 |
3 |
1.3 |
1 |
1.7 |
| Follow-up Time After Ablation |
| Patients with ≥ 1 Year |
32 |
100.0 |
232 |
100.0 |
58 |
98.3 |
| Patients with ≥ 2 Years*** |
32 |
100.0 |
145 |
62.5 |
52 |
88.1 |
Statistical tests: ANOVA for numeric variables and chi-squared for categorical variables; significance denoted by * for p<0.05, ** for p<0.01, *** for p<0.001; Note: sample sizes for a particular measurement may be different from the overall sample size due to missing data
Since the SF cohorthad ablations completed during the earliest portion of the study period, all 32 (100%) of this cohort had at least 2 years of follow-up time between their index procedure and the data extraction in December 2016. The ST cohort had the most recent ablations on average, but had a later data extraction date in December 2017, such that 100% of the 232 cases had1 year or more of follow-up time and 62.5% had2 or more years. The CA cohort included 58 (98.3%) patients with at least 1 year and 52 (88.1%) with at least 2 years of follow-up.
Acute procedure-related serious complications were infrequent, and occurred at similar rates between the three cohorts (SF: 2/32 [6%]; ST: 9/232 [4%]; CA: 1/59 [2%]; p=0.5311). The SF events were a case of pericardial effusion requiring treatment and a stroke with complete resolution after 3-4 days. The ST events included six cases of effusion or tamponade requiring treatment or extending the patient’s hospital stay; one vascular access event requiring a blood transfusion; and two cases of transient symptoms potentially indicating a cerebrovascular event, neither of which showed MRI findings nor required treatment. The CA event was a case of pericardial effusion requiring treatment.
Fluoroscopy times and radiation doses were significantly lower in the ST cohort (ANOVA p-values<0.0001) [Table 2]. Mean procedure times were similar for the SF cohort (92 minutes) and CA cohort (94 minutes), whileRF ablations using the newer ST technology resulted in a mean time savings of 10 minutes over the prior SF technology (82 minutes, ANOVA p-value=0.0201).
Table 2. – Procedural Detail
|
|
SF (N=32) |
ST (N=232) |
CA (N=59) |
| Efficiency Measure |
n |
Mean ± SD |
n |
Mean ± SD |
n |
Mean ± SD |
| Total Procedure Time*(minutes) |
31 |
92.2 ± 25.0 |
222 |
81.9 ± 31.7 |
50 |
93.6 ± 29.5 |
| Total Fluoroscopy Time***(minutes) |
28 |
1.5 ± 1.7 |
229 |
0.2 ± 0.4 |
41 |
12.4 ± 6.9 |
| Radiation Dose*** (mGy) |
30 |
247.7 ± 423.6 |
225 |
9.5 ± 26.0 |
53 |
676.4 ± 949.1 |
| Ablation Time (minutes) |
30 |
35.8 ± 11.1 |
227 |
31.5 ± 11.8 |
24 |
25.0 ± 11.9 |
| Ablation Procedure |
n |
Percent |
n |
Percent |
n |
Percent |
| Additional Non-PVI Triggers*** |
7 |
21.9 |
55 |
23.7 |
0 |
0.0 |
| DC Cardioversion Required |
2 |
6.3 |
10 |
4.3 |
7 |
11.9 |
Statistical test: ANOVA; significance denoted by* for p<0.05, ** for p<0.01, *** for p<0.001
Recurrence of any atrial arrhythmia was captured at 12-month follow-up visits starting approximately at the time of the change from SF to ST technology for RF ablations, and thus was not available for the SF cohort and was only available for CA procedures that occurred after systematic data collection was instituted. Since sample size with 12-month follow-up data was further reduced due to some patients not returning for a 12-month visit, only the ST population had sufficient sample size for reporting this outcome.
As shown in [Figure 2], 186 (80.2%) of the 232 ST patientswith an index ablation during the study period had 12-month follow-up visit data. Of the patients with follow-up, 161 (86.6%) met the 12-month success endpoint and 147 (91.3%) of these were also off AADs. Fifteen (8.1%) of the ST patients with 12 months of follow-up hadreablations within the first year, and another 10 (5.4%) were considered to be failures per the 12-month effectiveness endpoint. Thirteen of the 15 patients with reablations had 12-month follow-up visits after the second ablation, at which time 12 met the success criteria for the effectiveness endpoint.
Figure 2. ST Cohort Disposition
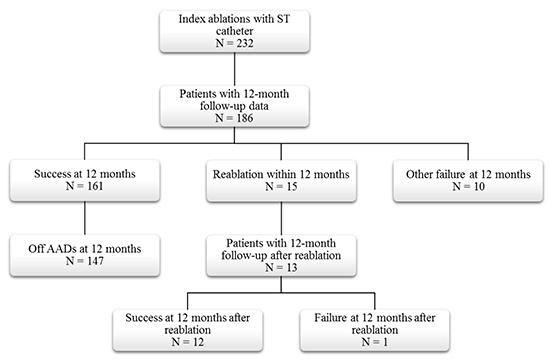
AAD: Antiarrhythmic drug ST: ThermocoolSmarttouchCatheter
Repeat ablation procedures
Kaplan-Meier survival modeling showed that ST ablation technology was associated with a significant reduction in repeat procedure rates (log-rank p-value=0.0194). Product limit survival estimates of repeat procedure rates at one year were 15.6% for SF,13.7% for CA, and 6.5% for ST. This reduction persisted at 2 years with cumulative reablation rate estimates of 21.9% for SF, 20.7% for CA, and 9.0% for ST [Figure 3].
Figure 3. Freedom from Reablation across Technology Based Cohorts
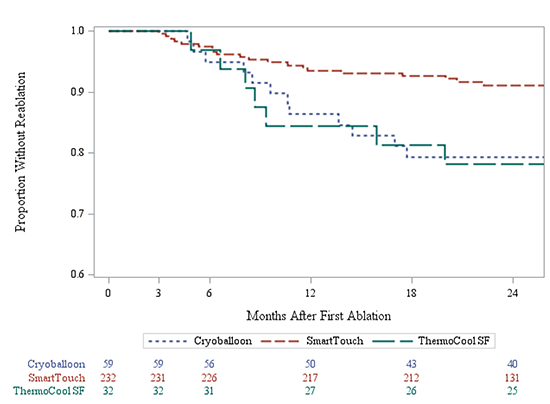
CA: Cryoablation SF: Thermocool SF Catheter ST: ThermocoolSmarttouchCatheter
Cox regression models were used to test for significance of additional baseline patient characteristics on risk of a repeat ablation procedure. None of the available demographics or co-morbidity indicators were significant after adjusting for the catheter cohort. In addition, separate models for each cohort showed no significant effect on need for reablation within any of the technologies.
Our main finding is that CF-sensing catheters, combined with Visitagtechnology andtheproposed standardized ablation protocol could decrease both total procedure and fluoroscopy times in PAF ablation procedures, compared to anon-CF-sensing catheter or cryoballoon. Our study also showed that integrating a CF-sensing catheter (ST) and an automated ablation annotation algorithm with a rigorous workflow led to good clinical outcomes, including a significant reduction in the need for reablation compared to procedures using the SF catheter or second-generation CA.
New technologies for radiofrequency AF ablation
Over the past decade, several technologies have advanced the field of AF ablation. Improvements in EAM systems, such as continuous multi-electrode mapping, have allowed us to obtain more information in a shorter time[7].CF-sensing catheters have also shown great promise in clinical trials, and now allow us to better estimate the quality of lesions delivered, ultimately achieving more durable lesions that can substantially improve procedural outcomes[8].The amount of information available to physicians through these technological advances in AF ablation has increased significantly; nevertheless, there exists no clear consensus on how to use, apply, and combine the new technologies for optimizing procedural outcomes.
A recent study by Anter et al. reported on effectiveness of automated annotation of RF ablation lesions usingVisitag software with different predefined filters and irrigated, non CF-sensing catheter technology[9].These researchers reported a higher PVI success rate after completion of the initial anatomical linewith Visitagand pre-defined catheter stability versus operator-guided ablation (90.5% vs. 66.7%, p=0.0001). This translated to a lower recurrence rate in the Visitagcohort (9.5%) compared to the cohort with empirical lesion tag annotation (23.8%).
Our workflow with CF-sensing technologies (ST cohort) resulted in lower RF time to achieve PVI compared to the Anter study (32 vs. 50 minutes). This reduction is likely due to: a) the addition of CF-sensing minimizing energy delivery when contact is sub-optimal; b) our minimalVisitag filters preventing unnecessarily long ablation times; and c) higher RF power used in our study. The ablation settings of 40 watts for 15 seconds that were used during our study correspond to a common trend toward higher power with shorter duration of energy application that has been proven safe and effective in invivo models.[10,11]
Several studies have also evaluated the synergistic role of Visitag with CF technology. The first study used different stability filters and when CF was not added to Visitag,only 74% of PVs were isolated with the initial PV encirclement, despite open access to real-time CF information[12].When a FOT criterion of 10 g was introduced, and further ablation was performed at the sites of automated annotation visual gaps, the PVI rate increased to 92%. This report did not includelong-term clinical results, but anotherstudy using similar strategiesresulted in clinical success rates of 81% at 12.9 months[13].
A more recent study found that use ofaVisitag software algorithm synergistically with CF ablation catheters resulted ina higher PVI rate during the first encircling lesion set compared to no Visitag use (66 vs. 37%, p=0.0006), with a corresponding improvement in freedom from AF at one year (91.8% vs. 76.2%)[14].Our study is the first to compare three widely used technologies, reportingimprovedprocedural efficiency with ST technology combined with a proposed lesion tagging workflow.
Although these technological advances are all intended to increase safety and durability of results, a standardized ablation method has not been validated and adopted widely within the electrophysiology community. The development of Ablation Index (AI) is a recentattempt to provide the missing piece of the equation, a single value thatobjectivelysummarizesthe total energy used. Preliminary studies using AI are promising, revealing improvements in clinical outcomes such as PV reconnection rates and tachyarrhythmia recurrence rates[15]. Our study was conducted prior to publication of the AI literature, but our goal of standardizingRF ablation strategies in order to reduce RF ablation time and the need for repeat ablation procedures was the same.
Cryoablationvs.force-sensing RF technology
Three studies have addressed procedural and clinical benefitsof CF-guided RF ablation versusCA.[16-18]All three studies failed to show a difference in atrial arrhythmia recurrences between the strategies, and they reported conflicting procedural outcomes. Two of the studies reported longer procedural time in the RF group compared to the CA,[16-17]with one suggesting that the reason was time spent generating a reliable 3D reconstruction of the LA geometry[16].Fluoroscopy time in this study, though longer than in our study, was not significantly different between the RF and CA cohorts[16]. Only the study by Kardos used Visitag software in a standardized way, in a non-specified number of patientsspanning approximately 80% of the study duration[17].Although total RF time was not reported, the mean procedural time of 120 minutes was substantially longer than reported in our study[17].Jourda et al. reported shorter procedure time and radiation exposure with CF-guided RF ablation, though values were considerably higher than we observed[18].This could be explained by the lack of standardized lesion tagging, thus supporting our hypothesis that AF ablation procedures could be more efficient with a standardized workflow and our proposed Visitag tagging strategy.
Our study is important as the first to comparethreewidely used AF ablation strategies while standardizing the use of the new CF technologiesin order to achieve reproducible results with minimal variability across physicians and mapping technologist. One clinically relevant benefit to both operators and patients is the minimization of fluoroscopy, particularly with CF-sensing technology where both Visitag and Univu are consistently used, averaging only a few seconds of exposure and mostly lead-free procedures for the operators.
Overall, our automated annotation strategy achieved the desired results of safety, efficiency and effectiveness. Procedural efficiency was achieved throughreducing RF ablation time due to a more lenient initial tag filter criteria. Effectiveness was improved through increasing our ability to identify areas with insufficient CF after the first lesion set, in which further ablation was required, thusresulting in a higher success rate and a lower repeat procedure rate compared to the other two strategies.
The primary limitations of this study are consequences of the non-randomized nature of the retrospective unmatched cohorts. In particular, the timing of SF procedures is earlier than the ST procedures. With rapid advancements in the field, the later procedures incorporate some learnings beyond those attributable specifically to CF technology. Alternatively, the ST procedures include a learning curve for this site on new technology since they represent the first ablations using the new workflow. CA procedures span both SF and ST procedures in time with no significant workflow changes, so theyare unlikely to contribute significantly to any time effects. Arrhythmia recurrence, which was collected only after the switch from SF to ST, could not be compared across technologies.
Another limitation is the potential for selection bias between RF ablation and CA. However, it is reasonable to assume that any selection bias would persist over the entire study timeframe, thus would not explain differences seen between the SF and ST cohorts. Additional limitations include potential confounding by unmeasured variables and the possibility of insufficient statistical power to detect significant differences for factors with low prevalence.