Predictors of long-term outcome in patients undergoing a first repeat ablation consisting solely of re-isolation of reconnected pulmonary veins.
Juan-Pablo Abugattas 1, Bruno Schwagten1, Jeroen De Cocker1, Hugo-Enrique Coutiño2, Francesca Salghetti2, Varnavas Varnavas 2, Erwin Ströker 2, Juan Sieira2, Michael Wolf1, Pedro Brugada2, Carlo de Asmundis2, Gian-Battista Chierchia2, Yves De Greef1
1
Electrophysiology Unit, ZNA Middelheim, Lindendreef 1, 2020 Antwerp, Belgium.2
Heart Rhythm Management Center, Postgraduate program in Cardiac Electrophysiology and Pacing, Universitair Ziekenhuis Brussel-Vrije Universiteit Brussel, Brussels, Belgium.
To define predictors of long-term outcome of a first repeat ablation solely consisting of re-isolation of reconnected pulmonary veins (PVs).
Three hundred seven patients (age 59 ± 9%, 77% males, non-paroxysmal AF 43%) with recurrent AF after first PVI were studied. Re-isolation of reconnected PVs was guided by a circular mapping catheter and 3D mapping system using RF ablations. A PV was defined as “triggering” in case of spontaneous ectopy or AF paroxysms originating from the PV.
After a mean follow-up of 5.05 ± 2.21 years, 194 (63.2%) patients (73.0% in PAF vs 50.4% in non-PAF, log Rank <0.001) were free from AF. A “triggering” PV was present in 48 (15.6%) during the first PVI and in 52 (16.9%) at repeat. Independent predictors of recurrence were a non-PAF type (HR: 1.814, 95%CI: 1.090 - 3.018, p=0.022) and early recurrence (≤ 3 months) after first PVI (HR: 1.632, 95%CI: 1.091 - 2.443, p=0.017) while a “triggering” PV at first or repeat was a predictor of good outcome (HR: 0.574; 95%CI: 0.344 - 0.959; p=0.034) in the multivariable analysis.
A repeat ablation solely consisting of re-isolation of reconnected PVs results in a high degree of long-term AF freedom, especially in PAF and in case of a PV trigger at index or repeat. Patients with non-PAF or experiencing early AF recurrence after first PVI are less responsive.
Key Words : Ablation, repeat, atrial fibrillation, triggering PV, re-isolation.
Correspondence to: Juan-Pablo Abugattas, M.D.
Electrophysiology Unit, ZNA Middelheim, Lindendreef 1, 2020 Antwerpen, Belgium and Heart Rhythm Management Center, Postgraduate program in Cardiac Electrophysiology and Pacing, Universitair Ziekenhuis Brussel-Vrije Universiteit Brussel, Brussels, Belgium.
Electrical isolation of the pulmonary veins (PVs) is now a mainstay AF treatment obtaining single procedure success rates of 60-80% [1-4]. A repeat ablation is however needed in about 30% of patients [1-6].
PV reconnection (PVR) is believed to be the main cause of recurrent AF after an initial successful PV isolation (PVI); this is supported by the finding of at least 1 reconnected PV during repeat ablation in around 80% of patients [2]. Re-isolation of reconnected veins (re-PVI) has shown to further eliminate AF and improve clinical outcome at short- and midterm follow-up. It constitutes therefore the basis of any repeat AF ablation as recommended by the HRS consensus document 2017 [4]. Additional ablation strategies beyond PV re-isolation are less well defined at the present time. No data exist at the present time on the long-term outcome of a re-PVI only approach. In view of this, the question arises in which patients a re-PVI only strategy would suffice to prevent future AF recurrences on the long-term and on the other hand in which patients additional ‘beyond the PVs’ mapping and ablation would be indicated. In the present article, we aim to (i) study long-term outcome of a repeat ablation solely consisting of re-isolation of reconnected PVs and (ii) to define clinical and procedural predictors of outcome.
In this retrospective, case control study, consecutive patients in the time period from February 2008 to December 2017 from the Middelheim-PVI Registry who underwent a similar ablation trajectory consisting of “PVI only” AF ablation as index procedure and subsequently a “re-PVI only” repeat ablation were analysed.
None of the patients included in the study underwent substrate modification, ablation of complex fractionated electrograms (CFAE) or ablation of non-PV triggers during the index or repeat ablation procedure.
Also, patients presenting with persistent PV isolation at repeat and those with documented or spontaneously occurring atrial tachycardia or flutter requiring additional ablation ‘beyond the PVs’ were excluded from the study.
Paroxysmal AF (PAF) was defined as AF that terminates spontaneously or with intervention within 7 days of onset. Persistent AF (PersAF) was defined as continuous AF sustained beyond 7 days, long-standing PersAF (LS-PersAF) as continuous AF of more than 12 months duration [4,5].
All patients provided informed consent for the procedure and the study was approved by our local Ethical Committee.
Ablation procedures were performed under therapeutic anticoagulation [uninterrupted warfarin (after 2012) or bridging with low-molecular weight heparin (LMWH)] (before 2012) and general anaesthesia. A 6F decapolar catheter was positioned in the coronary sinus. Immediately prior to transseptal puncture an intravenous loading dose of 10000 IU heparin was administered. Transseptal sheath(s) (TS) were continuously flushed with heparinized saline for the duration of the procedure (10mL/h). After puncture, a continuous infusion of heparin was started to maintain an activated clotting time above 300 seconds throughout the procedure.
The first PVI procedure was performed using different ablation strategies including conventional point-per-point radiofrequency (RF) ablation (CPVI) guided by the Carto system (Biosense Webster, Diamond Bar, CA, USA) in 134 (43.6%) or Ensite system (St Jude Medical, St. Paul, Minnesota, MN, USA) in 37 (12.4%), multi-electrode RF ablation using the PV ablation catheter (PVAC) (Medtronic Inc., Minneapolis, MN, USA) in 84 (27.3%), the high density mesh ablator (HDMA) (Bard Electrophysiology, Lowell, MA, USA) in 34 (11.1%) and cryoballoon ablation (CB) (Medtronic Inc., Minneapolis, MN, USA) in 18 (5.8%). The choice of the technique was at the discretion of the operator. If spontaneous repetitive ectopy or AF paroxysms was observed during ablation, the initiating focus was localized by combining conventional mapping (CS activation pattern) and sequential mapping of the PVs. A pulmonary vein was defined as a “triggering” vein if the earliest local activation was recorded from the PV preceding the onset of the surface P-wave and CS activation. For all techniques, procedural endpoint was defined as LA–PV entry block with elimination of the PV potentials during sinus rhythm and differential pacing.
All re-PVI procedures were performed using point-per-point RF ablation guided by a circular mapping catheter and the Ensite or Carto system. At baseline, all PVs were mapped sequentially with a circular mapping catheter. In case of repetitive ectopy or short runs of AF, mapping of the initiating focus was done as described above, with in case of a PV origin, defining the PV as “triggering”. Subsequently, reconnected PVs were re-isolated by point-by-point RF lesions through localization and closing of conduction gaps at the circumference of the previous ablation circle. Around a confirmed gap, additional RF applications were given for consolidation. In [Figure 1] a representative example of reconnection of a LIPV is given with a conduction gap at the anteromedian region (left panel) and delayed PV potentials (right panel, LIPV pre). Delivery of RF energy at the gap resulted in re-isolation of the LIPV (right panel, LIPV post).
Figure 1
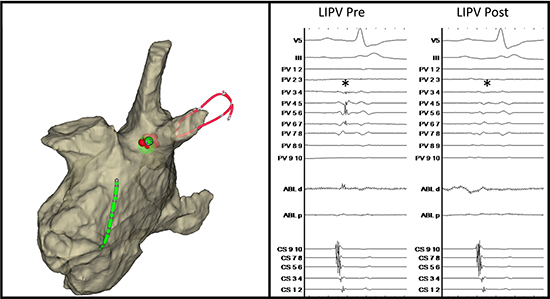
Left panel: representative LA Ensite map after re-PVI only ablation (left lateral view). A conduction gap (green tag) was identified at the anteromedian site of the prior ablation circle. Additional RF applications were given for consolidation reasons (red tags). Right panel: representative tracings of a re-isolation. Surface ECG leads V5/III and bipolar electrograms recordings of the coronary sinus and a decapolar mapping catheter positioned at the ostium of the left inferior pulmonary vein (LIPV) are shown. Ablation resulted in re-isolation.
Post-procedural management and follow-up
After the procedure, subcutaneous LMWH was administered to all patients, as well as oral anticoagulation therapy (OAT), either warfarin (target International Normalized Ratio between 2.0 and 3.0) or a NOAC. Antiarrhythmic drugs (AAD) were reinstituted in all patients. After the 1- month blanking period, OAT was continued (unless if a CHA2DS2-VASc score of 0) whereas all AAD were invariably stopped, except for beta blocking agents. All patients underwent follow-up (FU) [questionnaire, physical examination, and electrocardiogram (ECG)] at scheduled (every 6 months during the FU) and unscheduled visits (if symptoms). In case of symptoms, the related arrhythmia was documented either by ECG, Holter monitoring (1–7 days), or event recording.
The primary endpoint was the presence of a recurrence, defined as any episode of AF or atrial tachycardia (AT) of at least 30 seconds after re-PVI only ablation respecting a 3-month blanking period [4].
Continuous variables are expressed as mean ± SD or median and interquartile range (IQR) and significant differences were analysed by Student’s t˗test or the Mann–Whitney U test when appropriate. Categorical data are expressed as number and percentages and compared with χ² test or Fischer’s exact test when appropriate. Event-free survival was estimated by Kaplan–Meier method and compared by log-rank test. Hazard ratios (HR) were calculated using Cox proportional hazards models. For the multivariate analysis, variables with a P value greater than 0.10 were removed from the model. All analyses were performed with SPSS Statistics for Windows, Version 24.0.
Baseline population and anatomical characteristics
In the time period of the study, 2045 patients underwent a PVI only index procedure, of these, 437 (21.37%) patients underwent a repeat ablation procedure. In 130 patients either all PVs were found isolated at baseline, or an atrial tachycardia or flutter was present or documented, requiring additional ‘beyond re-PVI ablations. All these patients were excluded from the study. Finally, 307 patients in whom re-PVI only was performed were studied.
The baseline characteristics are presented in [Table 1]. In brief, the mean age was 59.03 ± 9.14 years (male: 77.2%, Non-paroxysmal: 43.3%). 10.4% of the patients had a CHA2DS2-VASC score of 3 points. The mean LA diameter (LAD) was 42.76 ± 6.69 mm, mean BMI 27.51±4.16 kg/m2.
Most patients had 4 separate veins (N=276, 89,9%), a minority had respectively a left common ostium (LCO) (N=26, 8.5%), a right middle PV (RMPV) (N=4) or a combination of an RMPV and LCO (N=1).
At baseline, out of 1208 studied PVs, 837 (69.29%) revealed recovered LA–PV conduction with a homogeneous spatial distribution of reconnection in between the 4 PVs: LCPV 21/25 (84.0%), LSPV 182/282 (64.5%), LIPV 197/282 (69.9%), RSPV 217/307 (70.7%), and RIPV 220/307 (71.7%). Reconnection of at least one pulmonary vein being a prerequisite was invariably present in all patients with a mean number of reconnected PVs per patient of 2.73 ± 1.01. A “triggering” PV was observed in 48 (15.6%) at the first PVI and in 52 (16.9%) of re-PVI procedures.
Table 1. Baseline clinical, anatomical and procedural characteristics.
Clinical Characteristics
|
Total(n=307) |
No Recurrence(n=194 (63.2%)) |
Recurrence(n=113 (36.8%)) |
p value |
| Age (years) |
59.03±9.14 |
59.48±9.03 |
58.24±9.30 |
0.252 |
| Gender (Male) |
237(77.2%) |
152(78.4%) |
85(75.2%) |
0.529 |
| Non-Paroxysmal AF |
133(43.3%) |
67(34.5%) |
66(58.4%) |
<0.001 |
| Left atrial diameter (mm) |
42.76 ± 6.69 |
42.02 ± 6.41 |
43.94 ± 7.00 |
0.021 |
| Coronary artery disease |
29(9.4%) |
18(9.3%) |
11(9.7%) |
0.895 |
| Dilated CMP |
4(1.3%) |
2(1.0%) |
2(1.8%) |
0.582 |
| Valvular heart disease |
7(2.3%) |
4(2.1%) |
3(2.7%) |
0.737 |
| Arterial Hypertension |
121(39.4%) |
73(37.6%) |
48(42.5%) |
0.402 |
| Diabetes Mellitus |
17(5.5%) |
10(5.2%) |
7(6.2%) |
0.701 |
| TIA/CVA |
15(4.9%) |
10(5.2%) |
5(4.4%) |
0.775 |
| Endurance sport practice |
62(20.2%) |
41(21.1%) |
21(18.6%) |
0.591 |
| Prior flutter ablation |
54(17.6%) |
30(15.5%) |
24(21.2%) |
0.200 |
| Tachycardiomyopathy |
27(8.8%) |
15(7.7%) |
12(10.6%) |
0.389 |
| Obstructive apnoea |
21(6.8%) |
11(5.7%) |
10(8.8%) |
0.287 |
| CHADS2VASC2 ≥ 3 points |
32 (10.4%) |
21 (10.8%) |
11 (9.7%) |
0.763 |
| Need for > 1 ECV before first PVI |
71(23.1%) |
30(15.5%) |
41(36.3%) |
<0.001 |
| Height (cm) |
176.15±8.71 |
176.03±8.33 |
176.35±9.36 |
0.754 |
| Weight (Kg) |
85.62±15.53 |
84.88±14.86 |
86.88±16.60 |
0.278 |
| BMI (Kg/m2) |
27.51±4.16 |
27.33±4.14 |
27.83±4.18 |
0.311 |
| Obesity(BMI: Kg/m2) | BMI: < 30 |
239(77.9%) |
153(78.9%) |
86(76.1%) |
0.931 |
| Obesity(BMI: Kg/m2) | BMI: 30 - 35 |
50(16.3%) |
30(15.5%) |
20(17.7%) |
0.931 |
| Obesity(BMI: Kg/m2) | BMI: 35 – 39 |
16(5.2%) |
10(5.2%) |
6(5.3%) |
0.931 |
| Obesity(BMI: Kg/m2) | BMI: ≥ 40 |
2(0.7%) |
1(0.5%) |
1(0.9%) |
0.931 |
| BSA (m2) |
2.04±0.22 |
2.03±0.21 |
2.06±0.23 |
0.324 |
| Time to first recurrence: median (IQR), months |
6.13 (2.79 – 18.17) |
6.54 (3.22 – 19.15) |
4.73 (2.40 – 15.84) |
0.116 |
|
|
Anatomical and Procedural Characteristics |
|
|
| PV anatomy 4 separate veins |
278 (90.6%) |
175 (90.2%) |
103 (91.2%) |
0.367 |
| PV anatomy LCO |
24 (7.8%) |
14 (7.2%) |
10 (8.8%) |
0.367 |
| PV anatomy RMPV |
4 (2.1%) |
4 (2.1%) |
0 (0.0%) |
0.367 |
| PV anatomy LCO + RMPV |
1 (0.5%) |
1 (0.5%) |
0 (0.0%) |
0.367 |
| Nº of PV's reconnected 1 |
44 (14.3%) |
28 (14.4%) |
16(14.2%) |
0.628 |
| 2 |
78 (25.4%) |
52 (26.8%) |
26(23.0%) |
0.628 |
| 3 |
103 (33.6%) |
60 (30.9%) |
43 (38.1%) |
0.628 |
| 4 |
82 (26.7%) |
54 (27.8%) |
28 (24.8%) |
0.628 |
| LSPV reconnection |
182 (64.5%) |
117 (65.4%) |
65 (63.1%) |
0.703 |
| LIPV reconnection |
197 (69.9%) |
124 (69.3%) |
73 (70.9%) |
0.630 |
| RSPV reconnection |
217 (70.7%) |
134 (69.1%) |
83 (73.5%) |
0.416 |
| RIPV reconnection |
220 (71.7%) |
141 (72.7%) |
79 (69.9%) |
0.604 |
| LCPV reconnection |
21 (84.0%) |
12 (80.0%) |
9 (90%) |
0.504 |
| Identified PV Trigger at index or repeat |
83(27.0%) |
62(32%) |
21(18.6%) |
0.018 |
| Index Ablation method CPVI |
171 (55.7%) |
117 (60.3%) |
54 (47.8%) |
0.069 |
| HDMA |
34(11.1%) |
17(8.8%) |
17(15.0%) |
0.069 |
| Cryoballoon |
18(5.9%) |
13(6.7%) |
5(4.4%) |
0.069 |
| PVAC |
84(27.4%) |
47(24.2%) |
37(32.7%) |
0.069 |
AF: Atrial fibrillation; CMP: cardiomyopathy; BMI: Body mass index; BSA: Body Surface area; PV: Pulmonary vein; LCO; Left common ostium; RMPV: Right middle PV; LSPV: Left superior PV; LIPV: Left inferior PV; RSPV: Right superior PV; RIPV: Right inferior PV; LCPV: Left common PV; ECV: Electrical cardioversion; IQR: Interquartile range; CPVI: conventional pulmonary vein isolation; RF: Radiofrequency; HDMA: High density mesh ablator; PVAC: PV ablation
Long term Outcome after re-PVI only
After a mean follow-up of 60.60 ± 26.52 months, 194 (63.2%) patients were free from any recurrence [Figure 2]. The median time to recurrence was 8.84 (IQR: 4.16 – 28.14) months. Patients with PAF had a better event free survival than patients with a non-PAF type (73.0% vs 50.4%, respectively; log Rank <0.001).
Patients who suffered a recurrence had a significantly larger LAD (43.94 ± 7.00 vs 42.02 ± 6.41mm, p=0.021) and more frequently underwent more than 1 electrical cardioversion (ECV) before the first PVI (41 (36.3%) vs 30 (15.5%), p< 0.001). Also, even statistically non-significant, the time to recurrence (TTR) after the first procedure tended to be shorter (4.73 (IQR: 2.40 – 15.84) vs 6.54 (2.79 – 18.17) months; p=0.116) in patients with a recurrence after re-PVI. Other clinical and procedural characteristics were not significantly different between both groups.
Seventy-five out of the 113 (66.4%) patients with a recurrence after re-PVI only underwent a second repeat ablation; of those, 28 (37.3%) patients presented with at least 1 reconnected PV.
Figure 2
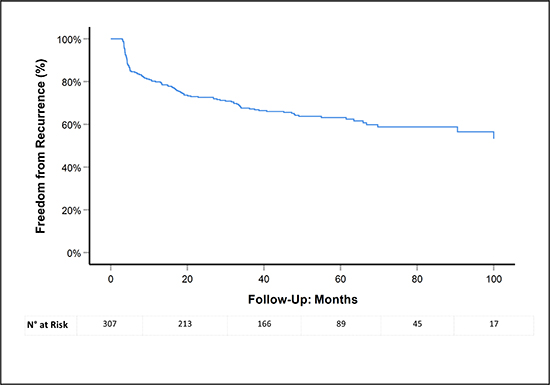
Kaplan-Meier plot representing the time to documented recurrence in the total study population. After a mean follow-up of 5.05 ± 2.21 years, 63.2% patients remained free of AF.
Predictors of recurrences
The results of the univariate and multivariate analysis are summarized in [Table 2]. In brief, in the univariate analysis, the LAD (HR: 1.032, 95% CI: 1.003 - 1.062; p=0.032), the need for more than 1 ECV before first PVI to maintain sinus rhythm (SR) (HR: 2.360; 95%CI: 1.605 - 3.470, p<0.001), a non-PAF type (HR: 2.382; 95%CI: 1.633 - 3.475, p<0.001) and a TTR ≤ 3months after the first PVI (index PVI procedure) (HR: 1.512, 95%CI: 1.027 - 2.228, p=0.036) were associated with an increase in the risk of recurrence after re-PVI. On the contrary, the presence of an identified PV trigger (HR: 0.575, 95%CI: 0.358 - 0.925, p=0.022) at first or re-PVI was associated with a better prognosis. Interestingly, neither the number or type of reconnected PVs nor the PV anatomy was predictive of outcome.
In the multivariate analysis, only the non-PAF type (HR: 1.814, 95%CI: 1.090 - 3.018, p=0.022) and TTR ≤ 3 months after the first PVI (index PVI procedure) (HR: 1.632, 95%CI: 1.091 - 2.443, p=0.017) persisted as independent predictors of recurrence while an identified PV trigger (HR: 0.574; 95%CI: 0.344 - 0.959; p=0.034) remained a predictor of good outcome [Figure 3].
Figure 3
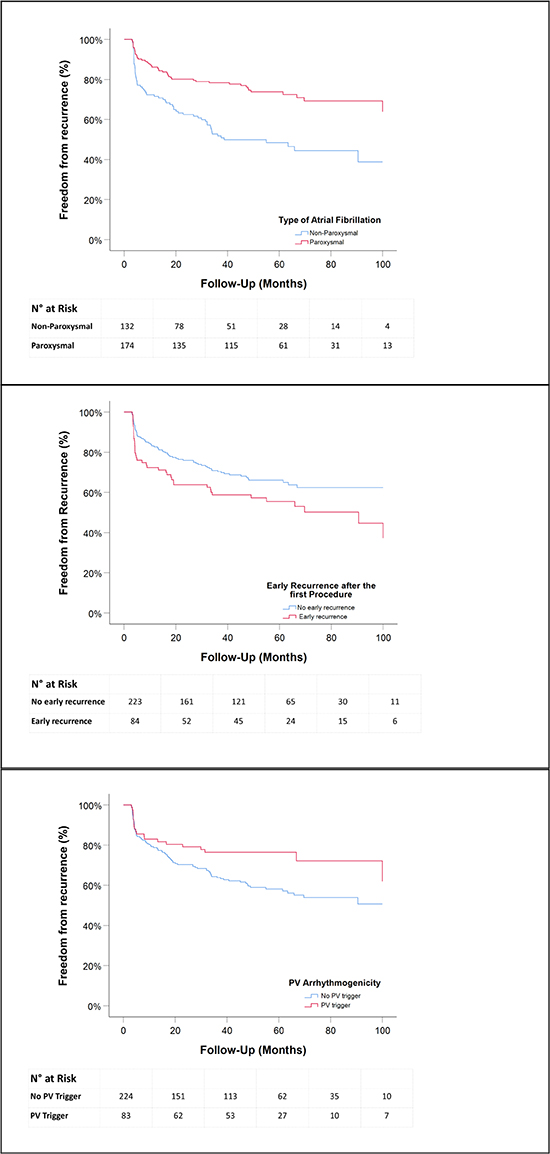
Kaplan-Meier curves representing the time to documented recurrence between PAF and non-PAF patients (upper panel), presence of a PV trigger at index or repeat (middle panel) and early AF recurrence after the first PVI (lower panel) respectively.
Table 2. Univariate and multivariate analysis
|
|
Univariate Analysis |
|
|
Multivariate Analysis |
|
|
p value |
HR |
95.0% CI for HR |
p value |
HR |
95.0% CI for HR |
| Age |
0.240 |
0.988 |
0.969 - 1.008 |
|
|
|
| Gender (F=1) |
0.563 |
1.135 |
0.740 - 1.740 |
|
|
|
| Coronary artery disease |
0.938 |
0.975 |
0.523 - 1.819 |
|
|
|
| Arterial hypertension |
0.396 |
1.176 |
0.809 - 1.708 |
|
|
|
| Diabetes mellitus |
0.577 |
1.243 |
0.578 - 2.672 |
|
|
|
| TIA /CVA |
0.486 |
0.726 |
0.295 - 1.786 |
|
|
|
| Left atrial diameter* |
0.032 |
1.032 |
1.003 - 1.062 |
0.426 |
1.013 |
0.982 - 1.044 |
| Identified PV trigger at index or repeat |
0.022 |
0.575 |
0.358 - 0.925 |
0.034 |
0.574 |
0.344 - 0.959 |
| Need for > 1 ECV before first PVI |
<0.001 |
2.360 |
1.605 - 3.470 |
0.391 |
1.249 |
0.751 - 2.078 |
| Non-paroxysmal AF |
<0.001 |
2.382 |
1.633 - 3.475 |
0.022 |
1.814 |
1.090 - 3.018 |
| Recurrence ≤ 3 months after the first PVI |
0.036 |
1.512 |
1.027 - 2.228 |
0.017 |
1.632 |
1.091 - 2.443 |
| LSPV reconnection |
0.595 |
0.897 |
0.600 - 1.340 |
|
|
|
| LIPV reconnection |
0.990 |
0.997 |
0.652 - 1.526 |
|
|
|
| RSPV reconnection |
0.619 |
1.112 |
0.732 - 1.688 |
|
|
|
| RIPV reconnection |
0.646 |
0.910 |
0.607 - 1.363 |
|
|
|
| Left sided veins reconnection |
0.989 |
0.996 |
0.578 - 1.717 |
|
|
|
| Right sided veins reconnection |
0.475 |
1.244 |
0.683 - 2.265 |
|
|
|
| Superior vein reconnection |
0.788 |
0.924 |
0.518 - 1.648 |
|
|
|
| Inferior vein reconnection |
0.804 |
1.096 |
0.533 - 2.252 |
|
|
|
| Reconnection of all 4 veins |
0.237 |
0.773 |
0.504 - 1.185 |
|
|
|
| Reconnection of ≥ 3 veins |
0.789 |
1.053 |
0.720 - 1.540 |
|
|
|
| Reconnection of only 1 vein |
0.806 |
1.067 |
0.637 - 1.788 |
|
|
|
| Number of PV's reconnected |
0.629 |
0.957 |
0.801 - 1.144 |
|
|
|
| Body mass index |
0.149 |
1.033 |
0.988 - 1.080 |
|
|
|
Increase in risk for every 1 mm increase in the diameter.
The main findings of our study are: (1) long-term AF freedom after a repeat ablation solely consisting of re-isolation of reconnected PVs is overall 63.2% going up to 73% in PAF patients, (2) patients with non-PAF and with an early AF recurrence within the first 3 months after first PVI have a higher risk of recurrence while (3) patients with an identified PV trigger at first or repeat PVI have less risk.
Long term Efficacy after re-PVI:
To the best of our knowledge, this is the largest study with the longest follow-up evaluating the efficacy and predictors of outcome following a repeat ablation procedure solely consisting of re-isolation of reconnected PVs. In our study, the incidence of a repeat procedure was 21.37% and therefore in line with the 20-40% of patients needing to undergo a repeat ablation due to AF recurrences as stated in the current guidelines [4].
Previous outcome studies after re-PVI based repeat ablations show varying degrees of success at short-and midterm follow-up from 49% up until 76% [6-9]. In 2004, Callans et al. reported data on 74 patients who underwent a repeat procedure using a re-PVI only strategy.[6] After 9.1 ± 6.7 months, freedom from AF was obtained in 44 (59%) patients. In a comparative study between RF and CB in repeat re-PVI only ablations for PAF, 20 out of 34 (59%) RF-treated patients versus 11 out of 29 (38%) CB-treated patients were free from any recurrence after 1 year follow-up.[7] Fichtner et al. compared in a prospective single center study the efficacy of re-PVI only versus re-PVI plus an anterior line.[8] After 12 months, more patients were in sinus rhythm off antiarrhythmic drugs after re-PVI only versus a combined re-PVI plus anterior line (26 of 41 (63%) versus 18 of 36 (50%), p= 0.26).[8] More recently, De Regibus et al reported the efficacy of the second generation CB for re-PVI.[9] After a mean follow-up of 15 ± 8 months, 76.6% of patients were free from recurrence of AF.
Our results differ from previous studies reporting on a much longer follow-up time of 60.6 ± 26.52 months years and the large number of patients (N 307) all undergoing a similar ablation trajectory (PVI only at index, re-PVI only at repeat). The present study shows that the majority of patients remains AF free using a re-PVI only strategy also on the long-term and supports therefore the primary aim of a repeat ablation to check for PV reconnections and subsequently close reconnection gaps around the previous ablation circle. However, our data also show that despite re-PVI a significant number of patients continue to develop AF recurrences.
Type of atrial fibrillation
Predictors of AF recurrence as AF type, LA size, early AF recurrence and the duration of AF disease are extensively studied and well known for a first PVI procedure [10,11].
Interestingly, some but not all of these predictors could be extrapolated to the present study.
AF type continues to be a significant predictor with a clear better outcome of re-PVI only in PAF (73.0%) versus non-PAF patients (50.4%). This finding reflects a higher ‘sole’ implication of the PVs in PAF, with other mechanisms beyond the PVs being more present in non-PAF patients suggesting the necessity of additional ablation. The latter is however not supported by the STAR-AF II trial showing a similar outcome also after two ablation procedures performed by means of the strategy to what they were initially randomly assigned (only PVI, PVI +lines, PVI +CFAE) [12].
The causative mechanisms of AF recurrence after re-PVI are most probably similar from those described for a first PVI procedure: non-durability of PVI despite re-isolation, the presence of non-PV triggers or a more advanced atrial disease.
Despite re-isolation of reconnected PVs at first repeat ablation, recovery of PV-LA conduction is not an uncommon finding in third and even in fourth ablations. Lin et al analysed 169 patients undergoing 3 or more AF ablation procedures.[13] PV re-reconnection was seen in 156 (92%) with as much as 41% presenting with all 4 PVs re-reconnected. Similar results were reported by Tilz et al[14] with PV re-reconnection found in 77.0%, 28,6% and 0% at the first, second and third repeat procedures. In our study, 75 patients underwent a third procedure and at least 1 reconnected PV was found in 28 patients (37.3%). Our study further highlights the difficulty in achieving durable PVI even after 2 ablations.
As for non-PV foci, the reported incidence has varied between 10 and 28% [15,16]. Although it is assumed that also in non-PAF the PVs harbour the majority of triggers, there is some discrepancy about their prevalence in comparison with PAF patients. Bhargava et al [16] reported a higher prevalence of non-PV foci in patients with LS-PersAF and PersAF than in PAF patients (19.1% and 8.2% versus 2.9% respectively, p<0.001). In contrast, Santangeli et Al [15] elicited non-PV triggers in only 11% of patients with a similar prevalence across the different types of AF (165 (11%) PAF, 54 (11%) PersAF, and 15 (11%) LS-PersAF; p=0.996).
Although evidence shows that not been able to locate and ablate a trigger (PV or non-PV) is associated with a worse outcome [17], a routine search for non-PV triggers was not incorporated in the present study. A future comparative trial between a re-PVI only repeat ablation versus a re-PVI plus actively searching and ablating non-PV triggers could give more insight in the prevalence of non-PV triggers at repeat ablations and differences in outcome.
Poor response to PVI and re-PVI only might also be explained by a more advanced structural atrial disease with enhanced fibrosis and low voltage areas. The presence of left atrial scarring in patients undergoing AF ablation is a known powerful, independent predictor of procedural failure [18].
Not only the presence but also the extent of atrial fibrosis is predictive of ablation outcome. Khurram et al [19] studied the association between late gadolinium enhancement (LGE) in the LA on MRI as a marker of fibrosis and AF recurrence after ablation in a cohort of 165 patients. Regardless of the type of AF, patients with LGE >35% had a higher rate of AF recurrence in the first year after ablation in comparison to patients with LGE ≤35%.
These studies support a more patient-tailored approach to target low LA voltage areas to improve ablation outcomes. Voltage mapping at baseline could have offered an insight to explain outcome after re-PVI only but was not systematically performed.
Time to recurrence after the first PVI
Our data shows that ‘early’ recurrences (within the first 3 months) are not only strongly predictive of ‘late’ AF recurrence after the initial PVI but also after a re-PVI only repeat procedure. This strengthens their significance as a marker of non-response to PVI rather than the subsequence of the healing process post-ablation [20,21]. Mechanisms related to ER are the presence of incomplete ablation lesions with early PV reconnection, untreated non-PV triggers, LA enlargement (>40mm), arterial hypertension and permanent AF [22].
These 3 later data as markers of more advanced structural atrial disease might suggest that also the presence of atrial fibrosis could be related with the pathogenesis of ERs.
The potential significance of ER as a non-response to PVI is in line with the results of a sub-study of the STAR AF [23] studying the impact of different ablation strategies (only PVI, only CFAE, combined PVI +CFAE) on early and late AF recurrences. Adding CFAE to PVI increased not only long-term success but resulted in rates of ER that were lower than observed with CFAE ablation or PVI alone. This implies that the persistence of non-PV triggers or unmodified arrhythmia substrate after limited PVI could be responsible for ER. Adding CFAE to PVI potentially result in targeting some non-PV triggers or additional substrate modification explaining the lower incidence of ER. The potential relevance of non-PV triggers to explain ERs is further supported by Themistoclakis et al [24] showing that the lack of isolation of the SVC was an independent predictor of ERs after AF ablation.
Our data show that not the number nor the type of PVs found reconnected at repeat but rather the observation of spontaneous electrical activity of the PVs is a key determinant for outcome after re-PVI only repeat. It supports that when a PV trigger is observed at index or repeat ablation most probably the arrhythmia is solely PV-mediated and durable PVI achieved by PVI or re-PVI suffices to achieve a good outcome.
In a previous publication, we described that the presence of a triggering PV during the initial PVI is associated with a higher risk of AF recurrence [25]. The triggering PV as the cause of AF was supported by a 100% reconnection rate with still signs of electrical activity (ectopy, bursts of AF) in 71% at repeat. In line with our study, a re-PVI only strategy was highly successful with long term AF freedom achieved in 22 out of the 25 patients undergoing a repeat ablation.
Our findings highlight therefore the importance of PV triggers during index and repeat procedures as predictor of good outcomes. Unfortunately, spontaneous PV triggering without provocative measures is a rather uncommon finding, and its exact prevalence is not well defined. Valles et al [26] reported spontaneous triggers in 23% of their patients while De Greef et al reported a prevalence of 17%[25]. In the present study, we observed this finding in 15,6% and 16,9% of patients during initial and repeat ablation respectively.
Nowadays, there is no doubt that if a reconnected vein is found, it must be ablated to gain durable and complete disconnection of the PV[27]. However, in our study, a significant number of patients kept having AF recurrences despite re-isolation of reconnected veins. Besides other non-PV causes, the finding that PV reconnection is also frequently observed in patients without clinical AF recurrence [2,28], shows that reconnected PVs can also be a bystander phenomenon.Jiang et al [28], analysed PV reconnections in 32 patients free from AF recurrences at 1-year follow-up. The authors found that 29 of 32 patients (90.6%) presented with at least 1 PV reconnected, and in 10 patients (31.2%) the 4 veins were isolated.
The question therefore remains if the patients who did well after re-PVI only did so due to the achievement of durability of PVI or due to other aspects of the procedure such as, for example, autonomic modulation.
Our study has some limitations. First, even though we have a large sample size and our follow–up period is long, it is still a single centre, single arm, retrospective, case control study and is exposed to the biases related to this type of studies. However, the potential selection bias of the criteria determining the choice to do a re-PVI only procedure was minimized given this approach constitutes our standard approach for a first repeat ablation.
Second, contact force sensing ablation catheters, which have shown better isolation rates than standard catheters, were not used in this study. Third, adenosine testing was not performed to search for dormant conduction. Fourth, we do not have information concerning voltage of the LA and consequently the extension of LA scars. Fifth, our follow-up did not include scheduled Holter monitoring, and therefore, asymptomatic episodes of AF may have been missed with a potential overestimation of our success rate. Finally, the lack of knowledge of the status of the PVs after ablation in patients without a clinical recurrence is an important limitation of our study; to overcome this, a new invasive assessment of the PVs would have been necessary, thereby exposing asymptomatic patients to potential complications.
Carlo de Asmundis receive compensation for teaching purposes and proctoring from AF solutions, Medtronic, member steering committee ETNA-AF Europe Daiichi Sankyo Europe and research grants on behalf of the centre from Biotronik, Medtronic, St Jude Medical Abbot, Livanova, Boston Scientific. Pedro Brugada receives fees from Biotronik, Medtronic. Gian Battista Chierchia receives compensation for teaching purposes and proctoring from AF solutions, Medtronic. Other authors: No conflict of interest to declare.
A repeat ablation solely consisting of re-isolation of reconnected PVs has a five-year success rate of 63%, going up to 73% in paroxysmal AF patients. A re-PVI only strategy is particularly efficacious if a PV trigger is observed during first or repeat ablation. Patients with non-PAF and patients with an early AF recurrence during the first 3 months after first PVI tend to respond less well to a re-PVI strategy.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.