Does Left Atrial Appendage Closure Reduce Mortality? A Vital Status Analysis of the Randomized PROTECT AF and PREVAIL Clinical Trials
William Whang1, David R. Holmes2, Marc A. Miller1, Marie-Noelle Langan1, Subbarao Choudry1, Aamir Sofi1, Jacob S. Koruth1, Srinivas R. Dukkipati1, Vivek Y. Reddy1
1
Icahn School of Medicine at Mount Sinai, New York, NY, USA.2
Mayo Clinic, Rochester, MN, USA.
Compared with warfarin, left atrial appendage closure (LAAC) reduced mortality in an analysis of the PROTECTAF and PREVAIL trials. However, these data were limited by patient drop-out.We sought to estimatethe mortality benefit with LAACusing updated vital status information.In PROTECT AF and PREVAIL, 227 of 1114 randomized subjects failed to complete 5-year follow-up. Centers were manually queried for updated vital status on 76 subjects (33%).During median follow-up 5.0 years (interquartile range 3.8, 5.1), 112 of 732 LAAC subjects (15.3%) and 79 of 382 controls (20.7%) died. The hazard ratio for all-cause mortality with LAAC compared with warfarin was 0.70 (95% CI 0.53-0.94, p=0.017).Subgroup analyses suggested that subjects <75 years and those with higher CHA2DS2-VASc score, history of transient ischemic attack or stroke, and permanent AF derived particular benefit, although interaction terms were not significant.The number needed to treat(NNT) with LAAC to prevent one death over 5 years was 16 (95% CI 10-82). Despite competing mortality risks in this elderly cohort, updated vital status data from PROTECT AF and PREVAIL revealed that LAAC was associated with 30% improved survival compared with warfarin, with an NNT of 16.
Key Words : Left Atrial Appendage Closure, Atrial Fibrillation, Anticoagulation.
Correspondence to: William Whang,
One Gustave L. Levy Place, Box 1030
New York, NY
Percutaneous left atrial appendage closure (LAAC)has been studied for stroke prevention among patients with atrial fibrillation (AF)in two randomized trials, The Watchman Left Atrial Appendage Closure Technology for Embolic Protection in Patients With Atrial Fibrillation (PROTECTAF)[1]and Prospective Randomized Evaluation of the Watchman LAA Closure Device In Patients With Atrial Fibrillation Versus Long Term Warfarin Therapy (PREVAIL)[2].The initial patient-level meta-analysis of these trials demonstrated that compared with warfarin, LAAC conferred lower hemorrhagic and cardiovascular mortality without a statistically significant difference in all-cause mortality[3]. A repeat analysis using 5 year follow-up data additionallydemonstrated lower all-cause mortality in the LAAC arm (hazard ratio 0.73; 95% CI: 0.54 to 0.98, p=0.035)[4].
Even though overall follow-up in the combined trial cohorts was extensive, a limitation of the analysis was the lack of complete 5 year follow-up – the goal in each trial. For instance, in PROTECT AF, 22.5% of subjects in the warfarin arm withdrew consent during the study[1].In this analysis we sought to evaluate all-cause mortality in the LAAC and warfarin cohorts of the two randomized trials with the maximum amount of available follow-up through a supplemental collection of vital status information.
The PROTECT AF trial cohort enrolled 707 patients with non-valvular AF at 59 sites in the USA and Europe between February 2005 and June 2008[1].Patients aged 18 years or older with paroxysmal, persistent, or permanent AF and CHADS2 risk scores ≥1 were eligible.Exclusions were based on the following: contraindications to warfarin, comorbidities other than AF requiring anticoagulation, left atrial appendage thrombus, patent foramen ovale with atrial septal aneurysm and right-to-left shunt, mobile aortic atheroma, and symptomatic carotid artery disease. In PREVAIL 407 patients with non-valvular AF were enrolled at 50 sites in the United States between November 2010 and July 2012[2].Eligibility criteria included CHADS2 score ≥2 or a CHADS2 score ≥1 with one of the following higher risk characteristics; female ≥75 years of age, baseline ejection fraction ≥30% but <35%, 65 to 74 years of age with either diabetes or coronary artery disease, and ≥65 years of age with heart failure. Exclusion criteria were similar to the PROTECT AF trial, except patients in whom clopidogrel therapy was indicated were excluded because of the potential confounding influence of this drug on efficacy outcome. Both PROTECT AF and PREVAIL were approved by the Institutional Committee on Human Research at the authors' institutions.
Patients in both trials were randomly assigned to the intervention or control groups in a 2:1 ratio. After implantation, patients were treated with warfarin for 45 days to allow endothelialization of the device, followed by clopidogrel (75 mg daily) plus aspirin (81–325 mg daily) until completion of the 6-month follow-up visit, and aspirin alone thereafter. The control group was assigned chronic warfarin therapy with target international normalized ratio (INR) 2.5, range 2.0–3.0.
For the present analysis, centers that participated in each study were queried and updated vital status was requested from study coordinators at each site. In a patient-level pooled analysis, we used chi-square tests for two or more categories to compare demographic factors, cardiovascular risk factors, and pattern of atrial fibrillation between the group of subjects for whom vital status was updated and the group for which updated vital status was not available. We estimated actuarial mortality curves for the warfarin and LAAC arms using the updated vital status information and estimated proportional hazards models to obtain the hazard ratio associated with LAAC, overall and in subgroups defined by stroke risk factors.We also tested for differential effects of LAAC by subgroup through multiplicative interaction terms comprised of treatment assignment and subgroup category.These analyses were performed with SPSS version 22 (IBM Corp).
We calculated the number needed to treat (NNT) to prevent one death using the survival probabilities in the LAAC and control groups as described by Altman and Andersen:[5]
NTT = 1/{[Sc(t)]h – Sc(t)}
where ‘Sc(t)’ is the control group survival probability and ‘h’ is the hazard ratio associated with treatment.
We then calculated the NNT with other established therapies for AF, heart failure, and coronary heart disease, and compared these with the NNT for LAAC.
In PROTECT AF and PREVAIL, among the 1114 randomized subjects 227 (20.4%)failed to complete the requisite 5-year follow-up as of June 2017 – 136 of 732 (18.6%) and 91 of 382 (23.8%) in the device and warfarin groups, respectively. In particular, withdrawal of consent occurred among 24 of 732total subjects in the device arm (3.3%) and 57 of 382 in the warfarin arm (14.9%). The vital status query resulted in revised data for 76 (33%) of the 227 subjects with missing information -- 50 of the 732 subjects in the device arm (6.8%)and 26 of the 382 subjects in the warfarin arm (6.8%). Subjects for whom updated vital status was and was not obtained were not significantly different with respect to demographic factors, cardiovascular risk factors, and pattern of atrial fibrillation [Table 1].
Table 1. Comparison of subjects with and without updated vital status.
|
Updated vital status N=76 |
No updated vital status N=1038 |
P value |
| Age, yrs |
73.4 |
69.9 |
0.60 |
| CHADS2 score |
2.2 ± 1.1 |
2.3 ± 1.1 |
0.19 |
| CHF |
19.7 |
26.0 |
0.28 |
| Hypertension |
90.8 |
90.4 |
1.00 |
| Diabetes |
22.4 |
28.9 |
0.24 |
| Prior stroke/TIA |
19.7 |
22.8 |
0.67 |
| AF pattern |
| Paroxysmal |
43.4 |
45.1 |
0.19 |
| Persistent |
17.1 |
24.9 |
|
| Permanent |
38.2 |
27.7 |
|
Values are mean ± SD or %. CHADS2 = congestive heart failure, hypertension, 75 years of age or older, diabetes mellitus, and previous stroke or transient ischemic attack; CHF = congestive heart failure; TIA = transient ischemic attack; AF = atrial fibrillation. P values refer to
The mean age and CHA2DS2-VASc scores of the combined cohort were 72.9±8.5 years and 3.7±1.4, respectively. During a median follow-upof 5.0 years (interquartile range 3.8, 5.1), 112 of 732 subjects in the LAAC arm (15.3%) and 79 of 382 in the warfarin arm (20.7%) died. LAAC subjects experienced a lower risk of all-cause mortality [Figure 1]; p=0.016, log rank test). In a proportional hazards model, the hazard ratio for all-cause mortality with LAAC compared with warfarin was 0.70 (95% CI 0.53-0.94, p=0.017). Analyses by subgroup suggested that subjects <75 years and those withhigher CHA2DS2-VASc score, history of transient ischemic attack or stroke, and permanent AF derived particular mortality benefit from LAAC, although multiplicative interaction terms were not statistically significant [Figure 2]. Also,trends in all of the subgroups suggested some degree of mortality reduction.
Figure 1. All-cause actuarial mortality curves for warfarin and LAAC groups.
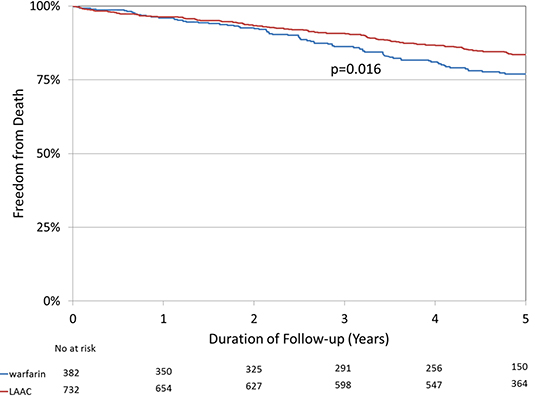
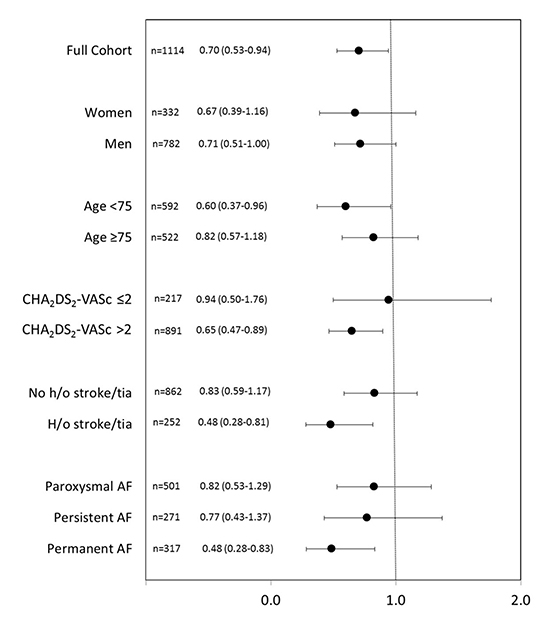
Figure 2. Hazard ratios and error bars delineating 95% confidence intervals from proportional hazards models by subgroups. Multiplicative interaction terms for each subgroup category were not statistically significant (p>0.05).
In an effort to understand the clinical relevance of thismortality benefit, we calculated the NNT value for LAAC to prevent one death over the course of five years. As shown in [Figure 3], the NNT was16 (95% CI 10-82). For comparison, the NNTs were also calculated for other cardiovascular therapies felt to confer mortality benefits, grouped by the time period studied for each treatment. This includes: i) the stroke prevention therapies of warfarin therapy as compared to aspirin (NNT = 164; 95% CI 49-Harm) and non-warfarin oral anticoagulant (NOAC) therapy as compared to warfarin (NNT =132; 95% CI 88-256), ii) device interventions such as left ventricular assist devices, implantable defibrillators for primary prevention, cardiac resynchronization therapy for wide QRS heart failure, and transcatheter aortic valve implantation, and iii) non-procedural medical interventions such as beta-blocker therapy in heart failure, dual antiplatelet therapy post-myocardial infarction, and statin therapy for primary prevention [Figure 3].
Figure 3. Number needed to treat (95% CI) to prevent one death for different interventions grouped according to the duration of treatment [8-17].Stroke prevention therapies are in bold.
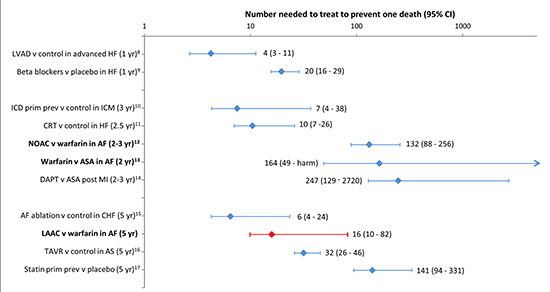
LVAD = left ventricular assist device; ICD = implantable cardioverter-defibrillator; CRT = cardiac resynchronization therapy; NOAC = non-warfarin oral anticoagulant; ASA = aspirin; DAPT = dual anti-platelet therapy; TAVR = transcatheter aortic valve replacement.
In this analysis of mortality using updated vital status data from PROTECTAF and PREVAIL, subjects assigned to LAAC experienced improved survival compared with subjects assigned to warfarin (HR 0.70, 95% CI 0.53-0.94; p=0.017). Our findings provide support to the previously published data using the maximum available follow-up for overall and subgroup analyses.
The ability to discontinue long-term anticoagulation after LAAC likely plays a role in reducing the mortality risk compared with warfarin.In the most recent meta-analysis of PROTECT AF and PREVAIL, a major reduction in the risk for hemorrhagic stroke was detected among subjects assigned to LAAC (HR 0.20; 95% CI 0.07-0.56, p=0.0022)[4].Hemorrhagic stroke is associated with poor prognosis, with one population-based study estimating 50% mortality at 30 days[6].The concept that reduced risk of hemorrhagic stroke was a driver of the mortality benefit from LAAC is consistent with findings from randomized trials that showed analogous benefits with non-vitamin K antagonist oral anticoagulants compared with warfarin[7].If the mechanism of mortality benefit from LAAC is related to discontinuation of anticoagulation and reduced bleeding risk, we would expect this to continue to accruewith longer follow-up. And indeed, this is what was observed. This is particularly striking given that not only would one expect competing risks for mortality in this elderly population (with a mean age of ~73), but the duration of follow-up was also quite long in duration (5 years).
To gain a better understanding of the clinical relevance to this mortality reduction, the NNTs for LAAC therapy as well as other cardiovascular therapies were determined. The NNT for LAAC versus control, 16 (95% CI 10-82), was notable in comparison to that calculated for NOAC therapy versus warfarin in AF, where the NNT was 132 (95% CI 88-256) over a period of two to three years. The NNT for left ventricular assist device therapy versus control in severe heart failure was particularly low, 4 (95% CI 3-11) over a time period of one year. Although direct comparisons of NNT need to account for factors such as differences in underlying risk and the period of time studied, our results nonetheless suggest that LAAC offers tangible clinically-relevant mortality benefits on a population basis.
Limitations of our analysis include the fact it was retrospective and not pre-specified during the clinical trials. Also, even with updated vital status information for 76 subjects, a substantial proportion (13.6%) still did not complete full 5-year follow-up. There is also the possibility of confounding if the acquisition of updated vital status was not random. However, cardiovascular risk factors were not significantly different among the groups of subjects with and without updated information; of course, selection bias remains a possibility. In addition, because both studies in this analysis had been previously closed out, we were unable to obtain more detailed information such as whether mortality was due to cardiac causes. Finally, because social security identifications were not recorded in these trial data forms, it was not possible to query the social security death index. Another limitation is thatour vital status update still left approximately 14% of randomized subjects with incomplete follow-up, although the additional information still strengthened the mortality reduction from LAAC that was recently estimated[4]. Another limitation of our study is the lack of data on other outcomes such as stroke or major bleeding.
In conclusion, in this analysis of PROTECT AF and PREVAIL with updated vital status data, among patients with AF who were at risk for stroke, LAAC conferred a 30% reduction in all-cause mortality (NNT = 16) compared with warfarin therapy during long-term follow-up. Future studies are necessary to determine whether this mortality benefit of LAAC over warfarin would also extend over NOACs.
Dr. Reddy has received grant support and has served as a consultant to Boston Scientific, manufacturer of the Watchman device. The remaining authors declare no conflicts of interest.