Second Generation Cryoballoon vs. Radiofrequency Ablation in Paroxysmal Atrial Fibrillation: Outcomes Beyond One-Year Follow-up
Galizia Brito V 1, Vecchio N1, Tomas L1, Jarma JJ 1, Mondragon I1, Burgos L1, Ordoñez S 1, Rivera S1, Albina G 1, Giniger A1, Scazzuso F 1
1Instituto Cardiovascular de Buenos Aires, Ciudad Autónoma de Buenos Aires, Argentina.
Pulmonary vein isolation (PVI) can be accomplished using radiofrequency (RF) or second generation cryoballoon (CB2). We aimed to compare the freedom from very late recurrence (VLR) defined as recurrence beyond one year in patients who were AF-free during the first post-procedural year after PVI using CB2 or RF.
Consecutive patients who underwent PVI by RF or CB2 ablation between August 2014 and December 2015 were included. The primary endpoint was the occurrence of VLR in follow-up after 12 months. Patients who experienced recurrence between the first 3 to 12 months after PVI and those who did not complete 15-month follow-up time were excluded.
139 patients were included: 68 underwent PVI by CB2 and 71 using RF. The global VLR rate was of 22.15%. The freedom from VLR beyond 12-month follow-up was of 84.5% (57 patients) for the CB2 group vs. 71% (50 patients) in the RF group (p=0.037). 15 patients underwent re-ablation (11 of the RF group and 4 of the CB2 group): all of the patients who had undergone PVI by RF in the index procedure were found to have vein reconnection, whereas none of the CB2 group had reconnected veins (3 cavotricuspid isthmus and 1 mitral isthmus).
In patients free of recurrence during the first post-procedural year after pulmonary vein isolation for the treatment of paroxysmal atrial fibrillation: second generation cryoballoon ablation showed a significantly lower very late recurrence rate compared to radiofrequency ablation.
The present study evaluates the freedom from very late recurrence (VLR), defined as recurrence in patients who were arrhythmia-free during the first post-procedural year after pulmonary vein isolation using second generation cryoballoon (CB2) or radiofrequency (RF). CB2 ablation had a lower VLR rate compared to RF.
Key Words : Atrial fibrillation, Catheter ablation, Cryoballoon, Pulmonary Vein Isolation, Recurrence.
Correspondence to: Victoria Galizia Brito. Postal address: Blanco Encalada 1543, Ciudad Autónoma de Buenos Aires, Argentina (1428).
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia found in clinical practice and a global healthcare problem with a prevalence of near 3% in adults aged 20 or older [1-2]. Incidence increases substantially with age and therefore prevalence is expected to rise as the population grows older as a result of increased life expectancy, especially in developed countries. Complications of AF include ischemic events such as cardioembolic stroke, heart failure, hospitalization, impaired quality of life and elevated mortality rates therefore entailing a large economic burden for healthcare systems [3].
Since Haïssaguerre et al. [4] reported that the main origin of AF trigger is located at the pulmonary veins (PV), targeting them is the main strategy for AF ablation [5-6]. PV isolation (PVI) by catheter ablation provides an effective and safe treatment option for drug-refractory symptomatic paroxysmal AF (PAF) and it is a class I, level A recommendation according to the last expert consensus statements [7-8] as it has proven to be superior to antiarrhythmic drugs (AAD) as a rhythm control strategy. Yet, AF may appear again after PVI despite improvement in technology.
PVI can be accomplished using any of two available techniques: a point-by-point mode application of radiofrequency (RF) energy or a single-step procedure with second generation cryoballoon (CB2) using circumferentially applied cryogenic energy. Currently, both methods are approved by the US food and drug administration for the treatment of PAF.
Several small studies have compared these two methods of ablation, showing heterogeneous results. The trial by Kuck et al. was the largest multicenter, randomized, controlled trial to assess the safety and efficacy of PVI in patients with drug-refractory symptomatic PAF using either CB2 or RF ablation: the mean follow-up time was 1.5 years, and the primary efficacy endpoint was a documented clinical failure (recurrence of AF, atrial flutter, atrial tachycardia, use of AAD or repeat ablation), following a 90-day blanking period after ablation. CB2 ablation proved to be as safe and non-inferior with respect to efficacy compared to RF ablation [9].
Recurrence during the 3-month blanking period is not considered actual recurrence and therefore must be excluded when presenting ablation results [7-8]. Late recurrence is the occurrence of atrial arrhythmia beyond the blanking period and very late recurrence (VLR) refers to arrhythmia recurrence happening for the first time beyond one-year follow-up after PVI.
Although the two techniques have points in favor and against, it is still unclear if one of them is superior to the other in terms of efficacy beyond one year follow-up, specially regarding VLR.
We aimed to compare the freedom from VLR in patients free of AF during the first year after PVI using CB2 or RF.
Every patient participating in our study signed informed consent for the procedure and for the use of their medical records, information about procedure and follow-up. The present study was approved by the Institutional Committee on Human Research in our institution.
Study Design AND Patient Population
We designed an observational, retrospective, single-center study based on data from a retrospective database. Consecutive patients with drug-refractory (one or more AAD class Ic or III or beta blockers) symptomatic PAF (defined as at least one episode of AF that terminates spontaneously or with intervention within seven days) who underwent PVI by RF or CB2 ablation between August 2014 and December 2015 were included. All patients were under effective oral anticoagulant treatment either with vitamin K antagonists (warfarin or acenocumarol) or direct anticoagulants (dabigatran, rivaroxaban or apixaban) continuously for 3 weeks or underwent transesophageal echocardiography to exclude the presence of thrombi in the left atria (LA). Before the index procedure, all patients underwent transthoracic echocardiography to determine ejection fraction and LA area, as well as multislice computed tomography (or cardiac magnetic resonance imaging if there was any contraindication for tomography) to evaluate the anatomic characteristics and number of PV.
Our primary endpoint was the occurrence of VLR in follow-up after 12 months. Patients who experienced late recurrence between the first 3 to 12 months (not considering recurrence during blanking period) after PVI and those who did not complete 15-month follow-up time were excluded, as well as patients who had previously undergone AF ablation and those with significant valvular disease.
All procedures were performed under general anesthesia. Transesophageal echocardiography was performed only if the level of anticoagulation were inconsistent to exclude LA thrombi or to guide difficult transseptal punctures. The aim of the procedures was to achieve PVI. Anticoagulation was achieved by I.V. heparin infusion targeting TCA ≥ 350 ms throughout the procedure.
RF ablation: 12-lead electrocardiogram and intracardiac bipolar electrograms were recorded using electronic calibrators (EP-WorkMate 4.2 System, St. Jude Medical, Inc.) at a screen speed of 50 to 200 mm/s and were filtered at band-pass settings of 50 to 500 Hz. A non-fluoroscopic mapping navigation system was used in all cases (Ensite® Velocity® cardiac mapping system, St.Jude Medical Inc.). After both femoral veins were punctured, a decapolar catheter was placed in the coronary sinus. Under radioscopic guidance in the 40° left anterior oblique projection, two transseptal punctures were performed with Brockenbrough needles; then, two long preshaped introducers SL1 and SL2 (St. Jude Medical Inc.) were positioned. A circular duodecapolar Optima Plus® catheter and an irrigated-tip ablation catheter Therapy-Cool® (St. Jude Medical Inc.) were advanced through the introducers to deliver RF. The anatomical reconstruction was performed using the circular Optima Plus® mapping catheter which is capable of simultaneous recording from multiple points. The ablation catheter was used to identify the ostia and the antrum of the PV. The electric activity of each PV was obtained using the circular catheter. PVI started in the left superior PV and continued in the left inferior PV. The same sequence was used for the right PVs. RF energy was delivered at the anterior and posterior aspect of each PV with a power output of 40 W and of 35 W, respectively. The lesions were applied to the antrum but not the ostia of the PV. The electrograms recorded by the ablation catheter before and after applying the ablation lesion were analyzed in each patient. The target was a reduction of the potential amplitude by 75% and the elimination or dissociation between atrial and PV activity. Once the isolation was completed, the presence of persistent block in each PV was evaluated. If necessary, ablation was repeated to consolidate the line of bidirectional block. We used all the methods available to discriminate local or remote electrical activity. After PVI, other AF ablation techniques were used at the discretion of the treating physician: ablation lines at the cavotricuspid isthmus, venae cavae or coronary sinus, complex fractionated atrial electrograms mapping, and areas with intermediate voltage values (between 0.1 and 0.8 mV) in the LA (substrate).
CB2 ablation: After cutaneous antisepsis and by modified Seldinger's technique, puncture of the femoral veins was performed. A decapolar catheter was introduced into the coronary sinus to record the electrical activity of the AV groove. Transseptal puncture with Brockenbrough needle was performed using fluoroscopic guidance. Following that, a metal guidewire was placed in the LA and a Flexcath introducer (Arctic Front) was advanced. The CB2 system (Arctic Front Advance® 28mm) was then advanced, along with the circular mapping catheter (Achieve Mapping Catheter®) with which electrograms were obtained inside each of the PV. Each PV was then catheterized with the balloon catheter, adjusting it to the antrum until a good objective occlusion was achieved with retention of 50% contrast inside the PV and absence of drainage to the atrial cavity. Once this was verified, freezing was started keeping the pressure of the balloon on the PV until reaching 90 seconds; after this time, while the balloon remained completely adhered to the PV antrum, freezing was continued for a total time of 240 seconds. Once ablation of the left PV was done, the decapolar catheter was positioned in the superior vena cava for continuous phrenic stimulation (1000 ms) in order to monitor its integrity during CB2 ablation of the right PVs, especially the superior right PV.
Each PV was then catheterized with the balloon catheter, adjusting it to the antrum until achieving a adequate occlusion with retention of 50% contrast inside the PV and absence of drainage to the atrial cavity. Once this was verified, freezing was started keeping the pressure of the balloon on the PV until reaching 90 seconds; After this time, while the balloon remained fully adhered to the PV antrum, freezing was continued for a total time of 240 seconds. Once isolation of left PV was achieved (both superior and inferior), the decapolar catheter was positioned in the superior vena cava for continuous phrenic stimulation (1000 ms) in order to monitor its integrity during CB2 ablation of the right PV.
In every procedure, esophageal temperature monitoring was used, avoiding temperatures below 20ºC. Immediate success was defined as the isolation of the PV with bidirectional block.
Every patient was systematically followed up with a 24-hour holter monitoring per month, two, three, six and twelve months after the PVI. And every 6 months thereafter. Symptomatic patients underwent extra 24-hour holter monitoring or 12-lead EKG in between the scheduled ones in an effort to document AF.
Patients continued under AAD treatment after PVI according to the criteria of each treating cardiologist.
We defined VLR as any documented episode of supraventricular atrial tachyarrhythmia (AF, atrial flutter or atrial tachycardia) at follow-up ≥ 12 months, both symptomatic and asymptomatic, lasting 30 seconds or more, observed in 24-hour Holter monitoring, 12-lead EKG or monitoring. All tracings were analyzed by a cardiac electrophysiologist.
All continuous variables were presented as mean (± standard deviation) or median (interquartile range) according to their distribution and categorical variables as percentage. Continuous variables with normal or asymmetrical distributions were compared using Student's t-test or Mann-Whitney U test respectively. Categorical variables were compared using Chi-square test or Fisher's exact test, as appropriate.
Kaplan-Meier plots were constructed to perform the survival analysis, and freedom from VLR between the CB2 and RF groups was compared by log-rank test. Patients who experienced recurrence between the first 3 to 12 months (not considering recurrence during blanking period) after PVI and those who did not complete 15-month follow-up time were excluded.
All statistical analysis was performed using SPSS software, version .24 (IBM Corp., NY, USA).
A total of 102 patients underwent PVI using CB2 and 95 using RF in our center in the period analized. 3 patients lost follow-up and 139 patients that met the inclusion critaria were included: 68 underwent PVI by CB2 ablation and 71 using RF [Figure 1]. The mean (±SD) follow-up time was of 19 (±3) months. There were no differences in baseline characteristics between groups [Table 1]. The global VLR rate was of 22.15%. The freedom from VLR beyond 12-month follow-up was of 84.5% (57 patients) for the CB2 group vs. 71% (50 patients) in the RF group (p=0.037) [Figure 2]. There were no differences in the occurrence of early recurrence (ER) during blanking period between the two groups: 27% (3 patients) in CB2 vs 42% (9 patients) in RF (p=0.38). Finally, 15 patients underwent re-ablation (11 of the RF group and 4 of the CB2 group). In this second procedure, all of the patients who had underwent PVI by RF in the index procedure were found to have PV reconnection, whereas none of the CB2 group had reconnected PV (3 cavotricuspid and 1 mitral isthmus).
Figure 1. Patient selection
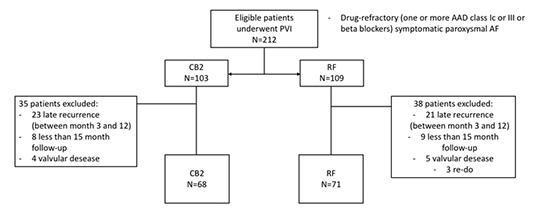
PVI: pulmonary vein isolation. AAD: antiarrythmic drug. AF: atrial fibrillation. CB2: second generation cryoballoon. RF: radiofrequency.
Table 1. Baseline characteristics
|
CB2 (68p) |
RF (71p) |
p |
| Age, yr (mean, ±SD) |
57.1 (±6.3) |
63.8 (±8.2) |
0.1 |
| Male (%) |
79.1 |
75.5 |
0.32 |
| Hypertension (%) |
42.4 |
56 |
0.25 |
| Diabetes mellitus (%) |
2.9 |
5.3 |
0.3 |
| Stroke or TIA (%) |
5.2 |
2.8 |
0.27 |
| EF (mean) |
62.3 (±6.9) |
62.1 (±7) |
0.74 |
| LA area (cm2) (mean ±SD) |
22.8 (±5.4) |
23.7 (±5.6) |
0.54 |
| ER during BP (%) |
27 |
42 |
0.38 |
| CHADS VASC 2 score |
1 |
1 |
0.8 |
CB2: second generation cryoballoon. RF: radiofrequency. TIA: Transient ischemic attack. EF: ejection fraction. LA: left atrium. ER: early recurrence. BP: blanking period.
Figure 2. Freedom from very late recurrence in follow-up in patients free from atrial fibrillation 12 months post-PVI by Kaplan-Meier survival with log rank test.
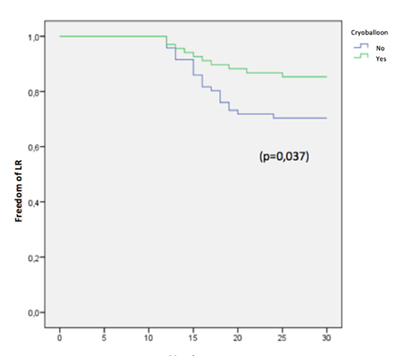
In patients free of recurrence during the first post-procedural year after pulmonary vein isolation for the treatment of paroxysmal atrial fibrillation: second generation cryoballoon ablation showed a significantly lower very late recurrence rate compared to radiofrequency ablation.
Even though there is limited data comparing RF to CB2: there are a series of pros and cons of each approach. Although it demands extensive training and is time-consuming, RF ablation is still the most widely used technique to achieve PVI [10], its benefits include the use of limited time of fluoroscopy. In contrast, PVI using CB2 leads to longer fluoroscopy times. On the other hand, CB2 ablation is a simpler technique and has a short learning curve, and several additional benefits such as increased catheter stability due to tissue-catheter cryo-adhesion; it generates a uniform, more delimited lesion with low inflammation burden and less platelet and coagulation cascade activation. CB2 ablation is a shorter procedure compared to RF [9,11]. In an economic analysis of the FIRE AND ICE trial by Chun et al., CB2 ablation was even associated with a reduction in resource use and costs [12]. PV can have anatomical variants and this can determine a difficulty for CB2 ablation, but regardless of anatomy, PVI by CB2 has shown similar results compared to RF in patients with unusual anatomic features [13].
Episodes of recurrence are usually diagnosed within the first year after PVI. Regarding CB2 vs RF in one-year follow up time, there were no differences in late recurrence [9].
There is no published data on VLR in patients free of AF in one year follow-up after PVI using CB2 and only limited information about VLR in general. There are studies focused on patients who underwent PVI and experienced their very first recurrence after one year of being free of AF. For example, in the study by Bertaglia et al. patients who were recurrence-free at one year follow-up were seen to have a progressive increase in the risk of VLR (13% at 2 years, almost 22% at 3 years, 35% at 4 years, 47% at 5 years and over 54% after 6 years) [14] . In the study by Mainigi et al. [15] on patients undergoing PVI using RF, VLR was an uncommon event and re-connectivity of segments of the PV was demonstrated in 93% of isolated PV in the index procedure. In our study, although the number of patients was limited, 100% of the the patients who had undergone PVI by RF had reconnection of PV.
In the last years, several studies have aimed to determine scoring systems to predict the occurrence of recurrence in order to identify high risk patients [16-19]. Particularly, the MB-LATER (male gender, bundle branch block, LA ≥47mm, type of AF, ER) scoring system published by Mujovic et al. [17] aimed to identify predictors of VLR. However, none of these scoring systems included the technique used (CB2 or RF) as a variable.
Regarding the comparison between the two ablation techniques, long term outcomes of the FreezeAF published by Armin Luik et al. [20] provided data comparing the efficacy of CB2 ablation vs. RF showing that CB2 was non-inferior to RF. This was seen in our study, suggesting that those patients that underwent PVI using CB2 and had no recurrence during the first year, will not experience VLR due to PV reconnection at all. This can be explained by the fact that CB2 lesion is more stable and generate less local inflammation, therefore the isolation of the PV can be thought to be more effective than point-by-point application of RF.
Recent studies have demonstrated that technical aspects of lesions creation such as contiguity, stability and contact force are important for clinical results, therefore stating that cryoballoon ablation is more reproducible than RF [21-22]. Techniques such as pacing the ablation line [23] and adenosine testing after PVI [24] have also demonstrated utility to avoid reconnection after RF ablation. Overall, this translates in large between-center results after PVI using RF. These techniques, although useful are not a gold-standard of care in PVI and there is no evidence from large randomized trials supporting their use. In our study, none of this techniques were used, this may have translated in more favorable outcomes for the CB2 group.
Our study has an important clinical implication, further larger and randomized trials are necessary to confirm our findings but the fact that the patients who had undergone PVI using CB2 with VLR had no reconnected PV suggest these patients will not experience AF recurrence at all. The importance of our study relies on the fact that patients with no episodes of VLR could more likely be considered as “cured” by CB2 ablation, therefore long-term follow-up and rhythm monitoring could be optimized. This information can also determine the length of treatment, for example oral anticoagulation.
Although our research reached its aims, we are still aware there were a series of limitations in its design. First, our study was a retrospective analysis and therefore we could be in presence of a selection bias, randomized and prospective studies are necessary to confirm our findings. Second, patient data were collected retrospectively. Third, the AAD treatment after PVI was not compared between the two groups. Fourth, our center is a monovalent institute, with a great number of patients and our electrophysiologists have vast experience in ablation procedures, therefore, results may not apply to the general population in our country. Also, there was no predefined criteria for choosing CB2 or RF for PVI, following each physitian´s judgement. Last, irrigated tip catheters were used, it can be thought that the use of contact force-guided catheters would have resulted in more effective ablation lesions and therefore better results.
We can conclude that in patients free of atrial fibrillation recurrence during the first post-procedural year after pulmonary vein isolation for the treatment of paroxysmal atrial fibrillation: second generation cryoballoon ablation showed a lower very late recurrence rate compared to radiofrequency.