Bronchial Injury – Yet Another Collateral Damage of Cryoablation
Amit Rout1, Arjun Kanwal1, William Han 2, Yousuf Kanjwal3
1
Department of Medicine, Sinai Hospital of Baltimore, Baltimore, MD, USA
.2
Sinai Division of Pulmonary Medicine, Sinai Hospital of Baltimore, Baltimore, MD, USA
.3
Sinai Cardiovascular Faculty, Sinai Hospital of Baltimore, Baltimore, MD, USA
.
With the aging population, the burden of atrial fibrillation is increasing. Cryoablation is a novel technique for pulmonary vein isolation and is considered equally effective as radiofrequency ablation. Some of the known complications of cryoablation include phrenic nerve damage, esophageal injury, pulmonary vein stenosis, new onset atrial flutter, pericardial effusion, and stroke. We present a case of bronchial injury immediately after cryoablation for symptomatic paroxysmal atrial fibrillation. A 67-year-old woman underwent successful pulmonary vein isolation using cryoablation. Her post-operative period was complicated with cough and hemoptysis. During the procedure, she had an extra 3 minutes of freezing of the left inferior pulmonary vein. Her bronchoscopy examination showed blood and erythema in the left main bronchus. She was managed conservatively with cough suppressants and anticoagulation was stopped. Possible etiology of bronchial injury is likely cryoenergy transfer between the pulmonary veins and the bronchus due to their close anatomic proximity. With the increasing use of cryoablation for atrial fibrillation, more cases of bronchial injury will likely be reported in the future. Physicians including electrophysiologist and pulmonologists should be aware of this complication.
Key Words : Atrial Fibrillation, Bronchial Injury, Cryoablation, Cryoballoon Catheter Ablation, Hemoptysis.
Correspondence to: Yousuf Kanjwal, M.D. FACC, FHRS.
Director, Cardiac Electrophysiology Sinai Cardiovascular Faculty
LifeBridge Health Cardiovascular Institute Sinai Hospital of Baltimore
2435 W. Belvedere Ave. Suite 32
Baltimore, MD 21215
In 1998 Haissaguerre et al. observed that foci of ectopic beats originating from pulmonary veins are capable of triggering atrial fibrillation (AF), which led to the basis of Pulmonary Vein Isolation (PVI) technique. [1] PVI is associated with improvement in morbidity, and quality of life when compared with medical therapy in patients with AF. In the recently published Catheter Ablation vs ANtiarrhythmic Drug Therapy in Atrial Fibrillation (CABANA) trial investigators reported that compared to drug therapy, ablation leads to significant improvement in the quality of life at 12 months. [2] PVI can be achieved by balloon cryoablation or radiofrequency ablation. The “Fire and Ice” trial showed cryoablation was non-inferior to radiofrequency ablation. [3] Routine complications like stroke, pericardial effusion and new onset atrial flutter can arise from any type of ablation procedure but cryoablation is unique to cause possible injury to nearby structures like esophagus, phrenic nerve and bronchial injury. In the “STOP AF” trial, 163 patients were followed after cryoablation for AF and postoperative cough was reported in 17% of patients with no reports of bronchial injury or hemoptysis. [4] In the “Fire and Ice” trial, out of 374 patients in the cryoablation group, cough was reported in 3 (0.8%) and hemoptysis was reported in 1 (0.3%) patients. [3] We report a case of bronchial injury which presented as hemoptysis within 2 hours of cryoablation. In addition, we also reviewed the available literature regarding bronchial injury after cryoablation and its management.
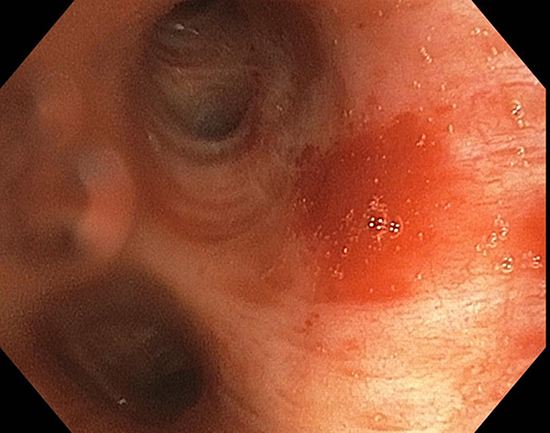
A 67-year-old woman was referred to electrophysiology clinic for symptomatic paroxysmal AF. She was switched from diltiazem to sotalol a few months ago; However, she continued to have breakthrough episodes. Her past medical history was significant for hypertension, diabetes mellitus, tobacco use, coronary artery disease, Takotsubo cardiomyopathy, transient ischemic attack, and renal vein thrombosis. Given failed medical management a decision was made for PVI using cryoablation technique. She was taking apixaban for anticoagulation which was stopped two days before the procedure. Her echocardiogram showed a normal left ventricle function and a normal size left atrium. Physical examination was benign and laboratory studies pre-procedure were within normal limits. EKG showed normal sinus rhythm on the day of the procedure. The patient underwent successful PVI using a 28 mm cryobaloon catheter (Arctic FrontTM Cardiac Cryoballoon, Medtronic, Inc., Minneapolis, Minnesota) and NavX™ cardiac mapping system (Abbott, Chicago, IL, USA). Peri-procedural heparin was used to maintain activated clotting time (ACT) between 350-400. All four pulmonary veins (PV) showed pulmonary vein potential. Cryoablation was performed using the freeze-thaw-freeze technique, 3 + 3 minutes, for each PV. Contrast was used to verify complete occlusion of pulmonary veins with the cryoballoon before each isolation. Left inferior PV (LIPV) had an extra 3 minutes of cryoablation for complete isolation. Complete isolation was achieved only after the third freeze. The lowest temperatures noted in the left superior PV (LSPV) were -50oC and -51oC in two freezes and in the LIPV temperatures were -38oC, -31oC, and -37oC in three freezes. All four PVs were ablated, and successful exit block was documented. The procedure was completed without any complication and heparin was reversed using protamine. The patient was extubated and transferred to the recovery room where she started complaining of a sore throat and cough. Two hours post procedure she had multiple episodes of hemoptysis. A chest CT showed left lower lobe infiltrate without any evidence of pulmonary embolism, aortic dissection or thrombosis [Figure 1]. The patient continued to have a small amount of hemoptysis. Apixaban was not restarted and aspirin was stopped. A diagnostic bronchoscopy showed a moderate amount of dark maroon blood in the left mainstem bronchus, which was completely occluding it. After the blood was suctioned away, an area of redness approximately 1cm was seen just proximal to the origin of the left upper lobe bronchus, and no active bleeding was noted [Figure 2]. A small amount of maroon colored secretion was present more distally as well as in the left lower lobe. The patient was hemodynamically stable and was managed with cough suppressants. Hemoptysis completely resolved two weeks after the initial procedure. Repeat chest CT after four weeks showed complete resolution of left lower lobe infiltrate. Aspirin was resumed once the hemoptysis stopped and anticoagulation was resumed after 4 weeks. At six months follow up the patient was asymptomatic and in sinus rhythm.
Figure 1. Illustration showing left lower lobe consolidation in Computed Topography of the chest with contrast.
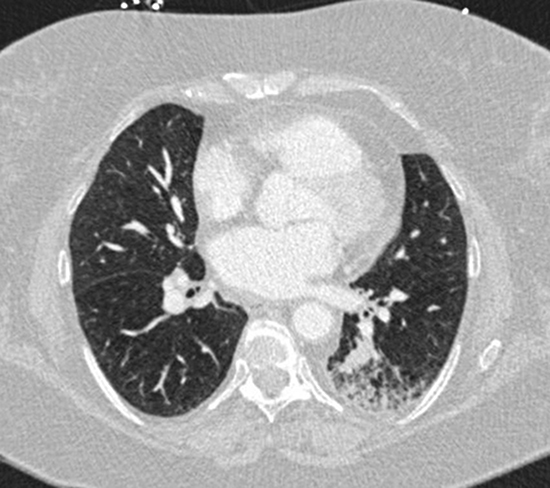
Figure 2. Bronchoscopic imaging showing erythema proximal to the origin of the left upper lobar bronchus.
Hemoptysis after cryoablation is a rare but can be a life-threatening complication, and so far very few cases have been reported [Table 1]. [6,11] The overall incidence is around 1.7 - 2.1% based on retrospective studies. [11,12] Reported cases of hemoptysis vary in their presentation from mild, self-limiting to massive life-threatening, causing respiratory compromise and requiring intubation. Even the time of onset of presentation ranges from hours to days to even months after cryoablation. Case reports of hemoptysis after radiofrequency (RF) ablation are also found in the literature. [13,14] Etiology of hemoptysis after RF ablation seems likely secondary to PV stenosis, but serious complications like atrio-bronchial fistula and communication between bronchus and pericardium have also been reported. [13,14] Overall incidence of PV stenosis after ablation is around 0.5-1.5 % and associated cases of hemoptysis are even less. [15,16] In a single center, prospective, observational study of 124 patients with PV stenosis mainly after RF ablation, the incidence of hemoptysis was reported in 27 % of patients. [17]
Table 1. Individual case reports of bronchial injury after cryoablation for atrial fibrillation.
| Case |
Author,
Year
|
Patient
Age (y), Sex
|
Presentation |
Time after Procedure |
Site of Injury |
Temperature (°C) |
Duration of Cryo |
Special Note |
| 1 (6) |
Van Opstal,
2011
|
65, M |
Hemoptysis |
4 Days |
Division of left upper & lower bronchus |
LIPV: -64 |
NA |
Deep application in LIPV during procedure |
| 2 (7) |
Marti- Almor,
2014
|
55, M |
Cough & Hemoptysis |
24 Hours |
Lingular bronchus bifurcation |
LSPV: -70
LIPV: -73
RSPV: -56
RIPV: -48
|
2 x 300s |
NA |
| 3 (8) |
Desai,
2015
|
29, M |
Hemoptysis |
Post Procedure |
Left Mainstem Bronchus |
Left PV: -60 |
150s |
Rapid & sustained drop in temperature |
| 4 (9) |
Aksu,
2015
|
55, M |
Hemoptysis |
Intra-operatively |
NA |
LSPV: -50 |
NA |
Imaging suggestive of Pulmonary Hemorrhage |
| 5 (11) |
Jayaschandran,
2017
|
59, M |
Epistaxis, hemoptysis, Dyspnea & chest pressure |
1 Month |
Left Mainstem Bronchus |
NA |
NA |
Life threatening hemoptysis requiring intubation |
| 6 |
Rout, 2019 |
67, W |
Cough & Hemoptysis |
2 Hours |
Proximal to left upper lobar bronchus |
LSPV: -50 & -51
LIPV: -38, -31 & -37
|
LSPV: 2x 180s
LIPV: 180s, 50s &180s
|
Three freezes to LIPV |
Y: Years, M: Male, Temperature in degree Celsius, S: seconds, LSPV: Left Superior Pulmonary Vein, LIPV: Left Inferior Pulmonary Vein, RSPV: Right Superior Pulmonary Vein, RIPV: Right Inferior Pulmonary Vein.
Previous studies have discussed the possible etiologies of hemoptysis following cryoablation, though the exact cause is still not very clear. [11] Animal studies done by Aryana et al. and review of previous studies give us some insight. Animal models suggest collateral injury and prolonged duration with low temperature as the possible culprit.[18] Initial case reports suggested PV stenosis and pulmonary infarction might be the cause but this had been refuted by CT imaging and histopathologic studies. The left and right mainstem bronchus are in close proximity with PVs making them vulnerable for injury from cryoablation. [19] Kumar et al. reviewed retrospective data of 283 patients and noted hemoptysis in 6 patients. In their study, lower temperature and deeper positioning of cryoballoon during cryoablation are possible risk factors for bronchial injury. [12] In another human study, frequent ice formation was noted in left mainstem bronchus in patients undergoing cryoablation. [20] Verma et al. performed simultaneous bronchoscopy in 10 patients undergoing cryoablation and found ice formation in 7 out of 10 patients in the left main bronchus only during LSPV isolation. They found no significant differences in minimum balloon temperature in freezes with or without ice formation. In a recent study, patients undergoing ablation with second-generation cryoballoon were followed by post-procedure bronchoscopy next day. [21] Out of 11 patients only 1 patient showed bronchial injury. In that study, they measured the distance between LSPV and LMB and the mean distance was 6.4 ± 2.7 mm. Interestingly, the distance was 2.1 mm in the patient with the bronchial injury. They also reported intraoperative coughing during the thawing phase and proposed it as a predictor of bronchial injury. [21] In our patient, symptoms started within 2 hours of procedure. The possible contributing factor for bronchial injury may have been the extra 3 minutes of cryoablation to the LIPV, though the temperatures recorded were not very low especially when compared to the temperatures in LSPV isolation.
Currently, no specific guidelines exist for prevention of bronchial injury. Review of literature provides some information, though they are mainly based on single-center experience. [19] Duration of cryotherapy and temperature play a significant role in the extent of the bronchial injury. We suggest limiting these factors as much as possible. In recent years, the concept of “less is better” has been advocated for cryoablation to reduce collateral damage in the adjacent esophagus, phrenic nerve, and bronchus. With the advent of 3rd generation cryoballoon catheter, it is possible to decrease the duration of freeze without compromising the efficacy of the procedure as shown by Pott et al. [22] Future trials need to validate whether decrease in duration will lead to improvement in safety outcomes. While we have means of avoiding phrenic nerve injury by constant pacing to monitor diaphragmatic stimulation and we also have esophageal probe to monitor intraoperative temperatures, currently no such similar techniques or tools are available to avoid bronchial injury. Physicians need to be extra vigilant in cases where longer duration or extreme temperature are seen during the procedure. A A low threshold should be maintained for bronchoscopy and CT imaging with the first presentation of symptoms, this will help prevent misdiagnosis and early initiation of intensive level of care if required. Once diagnosed, management mainly consists of cough suppressants, airway monitoring and holding anticoagulation. Repeat bronchoscopy may be warranted if symptoms fail to improve or depending on the extent of the initial injury requiring follow up.
The authors have no conflicts to disclose.
Funding and Financial Disclosures
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
As cryoablation is getting more popular for AF ablation, it is likely that the number of cases of bronchial injury will increase. Due to close anatomic proximity of left and right bronchus with PVs, they are always at risk of injury. The mechanism of injury seems to be a direct collateral injury from the cryoablation. High level of suspicion for bronchial injury should be maintained in symptomatic patients. Diagnosis can be made by early imaging and bronchoscopy. Management is mainly conservative and close monitoring.