Pulmonary Vein Isolation Using Ablation Index Improves Outcome in Patients with Atrial Fibrillation
Jonas Elmer Pedersen*, Kim Frost Lauritsen*, Jens Brock Johansen, Niels Christian Foldager Sandgaard, Jorgen Dalhoj, Stig Djurhuus, Jacob Pontoppidan
*These authors contributed equally to this work, and thus share the first authorship.Department of Cardiology
Odense University Hospital
Kløvervænget 47
DK-5000 Odense C, Denmark.
To evaluate the clinical outcome of pulmonary vein isolation (PVI) in radiofrequency ablation of atrial fibrillation (AF) comparing a strategy using Ablation Index (AI) and lesion contiguity with Contact Force (CF) only.
In a single-center retrospective design, we included 479 patients with AF (n=341 (71.2%) paroxysmal AF (PAF) and n=138 (28.8%) persistent AF (PeAF)) treated with first time radiofrequency ablation. In 2015, 210 patients underwent PVI based on a drag-and-ablate technique using CF only. In 2017, 269 patients underwent point-by-point PVI using AI and a maximum inter-lesion distance of 6 mm ensuring contiguity. Follow-up was performed after 12 months. Outcome was freedom from documented AF/atrial tachycardia (AT) after single procedure without use of anti-arrhythmic drugs at follow-up.
There was no significant difference in baseline characteristics between the groups. The median procedure time and mean ablation time were significantly longer in the AI-group compared to the CF-group (131.5[113;156] min vs. 120.0[97;140] min, P < 0.01) and (44.1±10.0 min vs. 37.1±13.3 min, P < 0.01), respectively. Freedom from documented AF/AT was significantly higher in the AI-group compared to the conventional CF –group (71.0% vs. 62.4%, P = 0.046). The improvement in clinical outcome in the AI group is mainly driven by the outcome in patients with PeAF (64.9% vs. 50.0%, P = 0.078) and not PAF.
An ablation strategy combining AI and lesion contiguity improves the clinical outcome after first time PVI in patients with AF compared to a strategy using CF only.
Key Words : Radiofrequency ablation, Pulmonary vein isolation, Persistent atrial fibrillation, Contact Force, Ablation Index.
Kim F. Lauritsen
Department of Cardiology
Odense University Hospital
Sdr. Boulevard 1,
DK-5000 Odense C, Denmark
Catheter ablation is a recognized treatment of symptomatic atrial fibrillation (AF). Various ablation strategies have been suggested, but pulmonary vein isolation (PVI) remains the cornerstone of radiofrequency ablation (RFA) in both paroxysmal AF (PAF) and persistent AF (PeAF).[1], [2] New catheter technologies providing real-time measurement of Contact Force (CF) sensing between the catheter tip and endocardium[3] and automated ablation lesion tagging technology based on catheter stability have been introduced to optimize the ablation lesions and thereby achieve durable isolation of the pulmonary veins (PV).[4], [5] Recently, the Ablation Index (AI) algorithm that incorporates CF, time and power has been developed as a marker of adequate ablation lesion.[2], [6]
AI accurately predicts lesion depth in animal models[7] and has shown improved clinical success rates when used in PVI ablation.[6], [8] A new concept using an automated ablation tagging module which combine AI values and a maximum distance of 6 mm between lesions to ensure contiguity has been investigated, and recent data show that this ablation strategy improves PVI durability and clinical outcome in patients with both PAF and PeAF.[9], [10]
The purpose of this study is to evaluate the efficacy and feasibility of this ablation concept in a population of PAF and PeAF patients compared to a conventional CF-guided PVI strategy.
Patients with symptomatic PAF or PeAF, who underwent first-time RFA during the year 2015 and 2017, were included. Patients with left ventricular ejection fraction (EF) <30%, age >80 years, previous major heart surgery (valve surgery, coronary artery bypass graft), congenital heart disease or additional lines during PVI procedure (except cavotricuspid isthmus ablation for typical flutter) were excluded. Patients with interrupted procedure due to complications were excluded in the analysis of outcomes. PAF and PeAF were defined according to guidelines.[1]
The study was approved by the Danish Data Protection Agency (J.nr. 2018-41-5401 Dok.nr. 468739) and performed according to the declaration of Helsinki.
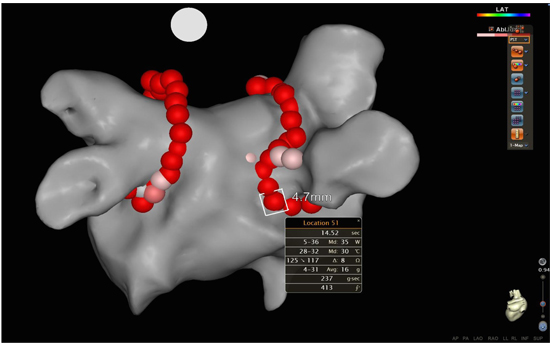
All procedures were performed with uninterrupted anticoagulant therapy (Warfarin or New Oral Anticoagulants (NOAC)). Before the procedure a CT scan of the left atrium was done. Pre-procedural transesophageal echocardiography was performed to exclude thrombus formation in the left atrium. The procedure was performed under minimal sedation using a combination of Midazolam and Fentanyl. After transseptal puncture, Heparine was administered with a target activated clotting time of 300-350 s. Electroanatomical mapping was performed with a multipolar mapping catheter (LASSO®, Biosense Webster Inc.). In 2015 (CF-group), PVI was performed with a CF catheter (SmartTouch™, Biosense Webster Inc.) and a drag-and-ablate technique using the CARTO® 3 system (version 6). During ablation, each ablation point was deployed using the VISITAG™ module (settings: stability 3 mm for 5 s., CF 25 % > 5 grams). In 2017 (AI-group), we performed point-by-point PVI using the SmartTouch™ catheter and AI (VISITAG™ setting: stability 3 mm for 5 s., CF 25% > 5 grams, an AI of 550 (anterior/superior) and 400 (posterior/inferior)) and lesion contiguity with a maximum inter-lesion distance of 6 mm [Figure 1]. The AI formula was described in the PRAISE study as a weighted formula , with constants replaced by letters [10]
Figure 1. illustrates a 3-dimensional electroanatomical map of the left atrium and adjacent pulmonary veins in a posterior-anterior projection. The red dots correspond to each ablation point obtained during pulmonary vein isolation using the VISITAG™ Module and Ablation Index target settings. The inter-lesion distance between the points is also measured.
Formula Formula.
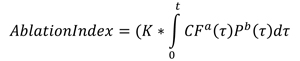
where CF = contact force, P = power and d = application duration.
Cavo-tricuspid isthmus (CTI) ablation was performed if patients had documented typical atrial flutter before or during procedure. The endpoint of procedure was bi-directional block in the PVs confirmed by the LASSO® catheter. Patients with PeAF underwent cardioversion during the procedure.
All patients were seen in the outpatient clinic after a 3 months blanking period and at 12 months of follow-up. The clinical visits included ECG, recording of symptoms and adjustments in medications. If no symptoms were presented and ECG showed sinus rhythm at the 3-month follow-up, all antiarrhythmic drugs (AAD) were discontinued. Patients underwent 7 days holter-monitoring to confirm any atrial arrhythmias if AF recurrence was suspected and the ECG was normal. In case of documented symptomatic AF recurrence, patients were scheduled for re-ablation. Patients with palpitations but no documented AF/AT continued AAD treatment. Patients with any AF symptoms between the clinical follow-up were advised to seek medical attention to confirm AF by ECG.
The primary outcome was freedom from documented AF or atrial tachycardia (AT) lasting more than 30 seconds verified on ECG at any time between 3- and 12 months without re-ablation or use of AAD, except betablockers.[1] Secondary outcome was the rate of re-ablation.
Continuous variables are presented as mean ±SD if they were normally distributed or median and [25th;75th] percentiles if the data are not normally distributed. For comparison we used Students t-test for normally distributed data. For comparison of non-normally distributed data we used Mann Whitney U-test or logistic transformation. Categorical data are presented as frequency and percentage and compared with chi-square test.
Logistic and multivariate logistic regression analyses were used to identify predictors of recurrence. Results are presented as relative risk ratio (95% confidence interval).
A P-value of < 0.05 was considered statistical significant.
All analysis was performed in Stata/IC (Version 15.1, StataCorp LLC, College Drive, USA)
In total, 479 patients who underwent first time PVI procedure for symptomatic AF were included (n=341 (71.2%) PAF and n=138 (28.8%) PeAF). There were no differences in baseline characteristics between the CF-group: (n=210 (43.8%)) and the AI-group: (n=269 (56.2%)) (see [Table 1]).
Table 1. Baseline Characteristics
|
Contact Force (n=210) |
Ablation Index (n=269) |
P-value |
| Age |
60.6±9.1 |
61.8±9.3 |
0.16 |
| BMI |
28.2(24.7;31.6) |
27.2(24.8;31.0) |
0.27 |
| Sex (Male) |
142(67.6%) |
191(71.0%) |
0.43 |
| Hypertension |
112(53.3%) |
131(48.7%) |
0.31 |
| Congestive heart failure |
14(6.7%) |
12(4.5%) |
0.29 |
| Diabetes |
18(8.6%) |
24(8.9%) |
0.89 |
| TCI |
17(8.1%) |
14(5.2%) |
0.20 |
| Vascular disease |
15(7.1%) |
29(10.8%) |
0.17 |
| CHA2DS2-VASc score |
1.6±1.3 |
1.6±1.4 |
0.97 |
| Type of AF |
|
|
|
| PAF |
146(69.5%) |
195(72.5%) |
|
| PeAF |
64(30.5%) |
74(28.8%) |
0.51 |
(BMI: “Body Mass Index”. TCI: “Transitory Cerebral Ischemia”. PAF: “Paroxysmal atrial fibrillation”. PeAF: “Persistent atrial fibrillation”. Congestive heart failure: “Defined as left ventricular ejection fraction (EF) less than 40% but higher than 30%”).
Procedural evaluation results
Successful PVI was achieved in 476 (99.4%) patients. Three procedures were interrupted before complete PVI isolation due to excessive pain during ablation. The total procedural- and ablation times were significantly longer in the AI-group compared to the CF-group (135.5[114;160] min vs. 120.0 [99;144.5] min, P ≤ 0.01) and (44.7±11.3 min vs. 37.1±13.4 min, P ≤ 0.01), respectively. The fluoroscopy time was shorter in the AI-group (11.4±7.1 min vs. 15.1±8.4 min, P ≤ 0.01) [Table 2]. Four procedural complications were registered (0.8%). In the CF-group, one patient had cardiac tamponade that required pericardial drainage and one patient experienced a minor stroke. In the AI-group, two patients had cardiac tamponade requiring drainage.
Table 2. Procedural Data
| Contact Force | Ablation Index | P-value |
|---|
| Xray time(min) | 15.1±8.4 | 11.4±7.1 | <0.01 |
| Xray dose (cGY/m^2) | 10(5;21) | 7(4;16) | <0.01 |
| Ablationtime(min) | 37.1±13.4 | 44.7±11.3 | <0.01 |
| Total proceduretime(min) | 120.0(99;144.5) | 135.5(114;160) | <0.01 |
| Procedural complications | 2(1.0%) | 2(0.7%) | 0.80 |
(AF: “Atrial Fibrillation”. PAF: “Paroxysmal atrial fibrillation”. PeAF: “Persistent atrial fibrillation”.).
In 94 patients, CTI ablation was performed. CTI was equally distributed between the AI-group and CF-group (21.9% vs. 16.8%, P = 0.157). Subgroup analysis of patients with PAF (24.6% vs. 19.3%, P = 0.246) and PeAF (14.9% vs. 10.9%, P = 0.495) showed no significant difference between the AI-group and CF-group.
Primary and secondary outcomes
The proportion of patients in sinus rhythm without any documentation of AF/AT after single PVI procedure was significantly higher in the AI-group compared to the conventional CF-group (71.0% vs. 62.4%, P = 0.046) [Figure 2]. Fewer patients underwent re-ablation in the AI-group compared to the CF-group although not statistically significant (16.8% vs. 23.3%, P = 0.071).
Figure 2. Freedom from documented recurrence of any atrial tachycardia or re-ablation or anti-arrhythmic drugs after a 3 months blanking period.
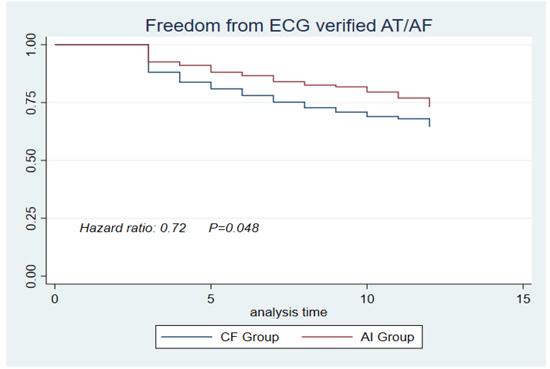
Subgroup analysis [Table 3] showed a trend toward improved outcome with AI especially in patients with PeAF compared to the CF-group (64.9% vs. 50.0%, P = 0.078). Significantly less patients with PeAF underwent re-ablation in the AI-group (18.9% vs. 34.4%, P = 0.039). In contrast, the re-ablation rate in patients with PAF was similar in the AI-group and CF-group (15.9% vs. 18.5%, P = 0.528).
Table 3. Clinical outcomes
| Primary outcomes |
Contact Force (n=210) |
Ablation Index (n=269) |
P-value |
| Success |
131(62.4%) |
191(71.0%) |
0.046 |
| Success by AF type: |
|
|
|
| PAF |
99(67.8%) |
143(73.3%) |
0.266 |
| PeAF |
32(50.0%) |
48(64.9%) |
0.078 |
|
|
|
|
| Freedom from ECG verified AT |
136(64.8%) |
195(72.5%) |
0.069 |
| Freedom from anti arrhythmic drugs at 12 months |
174(89.2%) |
241(92.6%) |
0.015 |
| Secondary outcome |
|
|
|
| No of re-ablations |
49(23.3%) |
45(16.7%) |
0.071 |
(AF: “Atrial Fibrillation”. PAF: “Paroxysmal atrial fibrillation”. PeAF: “Persistent atrial fibrillation”).
Logistic regression of prognostic factors for AF recurrence
In a multivariate analysis we tested for significant predictors for recurrence of AF. We tested gender, age, CHA2DS2-VASc, EHRA-score, AADs before procedure, type of AF, ablation time, X-ray dose, CTI ablation and procedure time. The only significant predictor of outcome was the type of AF (P = 0.006).
This study compared an ablation strategy combining AI and lesion contiguity to CF alone in AF patients scheduled for first-time PVI ablation. The main finding was a significant lower recurrence of AF/AT at 12 months follow-up in the AI-group. There was no difference regarding complication rate between the two ablation methods.
The primary goal in AF ablation is a durable PVI. The CF sensing catheters were introduced to optimize force feedback and produce more effective lesion sets to achieve the goal. A meta-analysis on CF-guided ablation found a success-rate of 80.1% in PAF patients and only 48.9% in PeAF patients.[11] In our subgroup analysis we found a success-rate in PAF and PeAF after CF-guided ablation of 67.8% and 50.0% respectively, indicating that our data are representative. In the STAR-AF2 trial, a similar outcome was found after PVI in patients with PeAF, and there were no clinical effect of additional linear ablation or ablation of complex fractionated electrograms.[12] The importance of a durable PVI has pushed the technology further in developing algorithms which incorporate various information such as CF, time and power into each lesion set during RFA. The TOCCASTAR and EFFICAS I/II studies have shown that CF technology combined with force-time integral improve the rate of isolated PV after 3 months of follow-up and long-term clinical outcome compared to non-CF ablations.[3], [5] The AI algorithm which incorporates CF, time and power into each lesion set was developed as a marker of ablation outcome. AI was evaluated in animal and human studies and has shown a rate above 90% of isolated PV in both PAF and PeAF patients at scheduled re-interventions 3 months after index ablation.[8], [10] The recent PRAISE trial reported 93% isolated PV at re-evaluation after 3 months in 40 patients with PeAF scheduled for PVI only.[10] Freedom from AT after 12 months of follow-up was 80%, which is higher than our results. This may be explained by a difference in our sedation protocol and PV isolation control maneuvers. We used minimal sedation, but patients undergoing AF ablation in general anaesthesia have a higher success rate.[13] Furthermore, we did not check for dormant reconduction with adenosine which in some studies have shown a better clinical outcome.[14] Nevertheless, our data supports that PVI only in patients with AF undergoing first time ablation is a reasonable strategy, and AI is effective, probably due to durable PVI. Furthermore, focus on lesion contiguity by checking the lesion sets around the PV for gaps above 6 mm may also explain the improvement in clinical outcome. Studies have shown that non-contiguity between point-by-point lesions along the ablation lines results in PV reconduction even with effective lesion sets.[3], [15] Ablation- and total procedural time were longer in the AI group. This indicates that operators use more time creating and placing the lesions which might lead to more sustainable lesions leading to higher success rates.[16] This finding is consistent with other studies.[8]
Our overall improvement in clinical outcome in the AI group is mainly driven by the improvement for patients with PeAF and not PAF. Previous studies have found that the PV often are reconducting even in patients without AF recurrences, and maybe a strategy using CF only is sufficient to achieve reasonable clinical freedom from AF/AT in PAF patients[17]. It is uncertain if the AI strategy in our population increased the PVI durability compared to the CF-group since we did not re-evaluate this during follow-up. Notably, the PRAISE trial found that only 1 of 8 patients with recurrent AF had PV re-conduction.[10] This emphasizes the need for other strategies to treat patients with recurrent AF after PVI such as targeting extrapulmonary triggers, low-voltage areas or rotors.
There are several limitations to this study. Although the study reflects real life outcome in consecutive patients undergoing PVI procedure in a high volume center, the two interventions are compared in a retrospective design. We characterized the AF type according to the definition in guidelines, but some misclassification cannot be ruled out. We only used ECG to evaluate the patients at follow-up, and more frequent ECG monitoring could probably have documented episodes of asymptomatic AF/AT during follow-up. Furthermore, follow-up was limited to 12 months thus long-term success rates are not available. Patients did not follow a strict protocol of AAD withdrawal after the procedure. We considered the use of AAD at the 12 months clinical control as a recurrence of AF/AT, however theoretically a patient could have stopped AAD the day before 12 months clinical control.
This retrospective study shows that an ablation strategy combining AI and lesion contiguity improves the clinical outcome after first-time PVI in patients with paroxysmal or persistent AF.
- Calkins, H., et al., 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation: Executive summary. J Arrhythm, 2017. 33(5): p. 369-409.
- Phlips, T., et al., Improving procedural and one-year outcome after contact force-guided pulmonary vein isolation: the role of interlesion distance, ablation index, and contact force variability in the 'CLOSE'-protocol. EP Europace, 2018. 20: p. 419-427.
- Kautzner, J., et al., EFFICAS II: optimization of catheter contact force improves outcome of pulmonary vein isolation for paroxysmal atrial fibrillation. EP Europace, 2015. 17(8): p. 1229-1235.
- Marijon, E., et al., Real-time contact force sensing for pulmonary vein isolation in the setting of paroxysmal atrial fibrillation: procedural and 1-year results. journal of cardiovascular physiology, 2014. 2: p. 130-137.
- Natale, A., et al., Paroxysmal AF catheter ablation with a contact force sensing catheter: results of the prospective, multicenter SMART-AF trial. Journal of American College of Cardiology, 2014. 64(7): p. 657-659.
- Solimene, F., et al., Safety and efficacy of atrial fibrillation ablation guided by Ablation Index module. J Interv Card Electrophysiol, 2018. 44: p. 9-15.
- Nakagawa, H. and W.M. Jackman, The Role Of Contact Force In Atrial Fibrillation Ablation. Journal of atrial fibrillation, 2014. 7(1): p. 1027-1027.
- Hussein, A., et al., Prospective use of Ablation Index targets improves clinical outcomes following ablation for atrial fibrillation. J Cardiovasc Electrophysiol, 2017. 28(9): p. 1037-1047.
- Duytschaever, M., et al., P6227Reproducibility and acute efficacy of a standardized approach to isolate the pulmonary veins: results from multicenter VISTAX study. European Heart Journal, 2018. 39(suppl_1): p. ehy566.P6227-ehy566.P6227.
- Hussein, A., et al., Use of Ablation Index-Guided Ablation Results in High Rates of Durable Pulmonary Vein Isolation and Freedom From Arrhythmia in Persistent Atrial Fibrillation Patients. Circ Arrhythm Electrophysiol, 2018. 11(1941-3084 (Electronic)): p. e1006576.
- Kirchhof, P. and H. Calkins, Catheter ablation in patients with persistent atrial fibrillation. Eur Heart J, 2017. 38(1): p. 20-26.
- Verma, A., et al., Approaches to catheter ablation for persistent atrial fibrillation. New England Journal of Medicine, 2015. 372(1533-4406 (Electronic)): p. 1812-1822.
- Martin, C.A., et al., Improved outcome and cost effectiveness in ablation of persistent atrial fibrillation under general anaesthetic. EP Europace, 2018. 20(1532-2092 (Electronic)): p. 935-942.
- Macle, L., et al., Adenosine-guided pulmonary vein isolation for the treatment of paroxysmal atrial fibrillation: an international, multicentre, randomised superiority trial. (1474-547X (Electronic)).
- Park, C.I., et al., Mechanisms of pulmonary vein reconnection after radiofrequency ablation of atrial fibrillation: the deterministic role of contact force and interlesion distance. journal of cardiovascular electrophysiology, 2014(1540-8167 (Electronic)): p. 701-708.
- Gul;, E.E., et al., Contact-Force Guided Pulmonary Vein Isolation does not Improve Success Rate in Persistent Atrial Fibrillation Patients and Severe Left Atrial Enlargement: A 12-month Follow-Up Study. Journal of Atrial Fibrillation, 2018. 11(2): p. 2060.
- Kuck, K.H., et al., Impact of Complete Versus Incomplete Circumferential Lines Around the Pulmonary Veins During Catheter Ablation of Paroxysmal Atrial Fibrillation: Results From the Gap-Atrial Fibrillation-German Atrial Fibrillation Competence Network 1 Trial. Circ Arrhythm Electrophysiol, 2016. 9(1): p. e003337.