Safety and Efficacy of Cryoballoon Ablation for the Treatment of Atrial Fibrillation in Diabetic Patients
ABDIN Amr14*, HEEGER Christian-H.12*, YALIN Kivanc13, SANTORO Francesco5, Natale Daniele BRUNETTI5, Thomas FINK1, Spyridon LIOSIS1, BRUEGGEMANN Ben1, KEELANI Ahmad1, Huong lan PHAN1, SANO Makoto1, Vanessa SCIACCA1, LYAN Evgeny1, Dong AN1, Roza MEYER-SARAEI12, OUYANG Feifan1, KUCK Karl-Heinz1, EITEL Charlotte1, Julia VOGLER1, Roland Richard TILZ12
1University Heart Center Luebeck, Medical Clinic II (Department of Cardiology, Angiology and Intensive Care Medicine), University Hospital Schleswig-Holstein, Luebeck, Germany.2German Center for Cardiovascular Research (DZHK), Partner Site Hamburg/Kiel/Luebeck, Luebeck, Germany.3Istanbul University-Cerrahpasa, Cerrahpasa Faculty of Medicine, Department of Cardiology, Istanbul, Turkey.4Current affiliation: University Hospital RWTH Aachen, Department of Cardiology, Angiology and Critical Care (Medical Clinic 1), Aachen, Germany.5Department of Medical and Surgical Sciences, University of Foggia, Foggia, Italy;.*AA and CHH contributed equally to this manuscript.
Cryoballoon based catheter ablation (CB-CA) is an established therapy for treatment of symptomatic atrial fibrillation (AF). However, data about AF ablation using the CB in the diabetic population is sparse. The aim of this single center retrospective study is to evaluate the safety and efficacy of CB ablation in patients with diabetes mellitus (DM) as compared to patients without DM.
Between July 2015 and December 2017, 397 consecutive patients underwent CB-CA for AF. Forty-eight consecutive patients with DM (DM group, study group) were compared with propensity score-matched patients without DM (n=48, control group). All patients underwent pulmonary vein isolation (PVI) using the second-generation CB (CB2). The mean age in the DM group was 66.9±9.5 years and 69.5±8.8 in the non-DM group (p=0.18). During a follow-up of 12.7±5.1 months, single procedure success rate for the DM and the non-DM group was 68.7% and 70.8%, respectively (p=0.82). The most common complication was transient phrenic nerve palsy (4 DM group vs. 0 non-DM group, p=0.04). No severe complication such as procedure related deaths, atrio-esophageal fistula or cerebrovascular embolic events occurred.
Our data strengthen the value of CB2 based ablation for the treatment of AF as an effective and safe procedure in DM patients, with similar success rates when compared with a non-DM population.
Key Words : Atrial fibrillation, Cryoballoon, Catheter ablation, Diabetes mellitus.
Prof. Dr. med. univ. Roland Richard TILZ, MD, FHRS, FESC, FEHRA
University Heart Center Luebeck,
Department of Cardiology, Angiology and Intensive Care Medicine,
University Hospital Schleswig-Holstein,
Ratzeburger Allee 160, 23538 Luebeck, Germany
The prevalence of atrial fibrillation (AF) is markedly increased in patients with diabetes mellitus (DM) [1],[2]. This risk of AF occurrence is increased due to macro- and microvascular dysfunction, diabetic autonomic neuropathy and systemic inflammation leading consequently to atrial remodeling and fibrosis [1].
Catheter ablation (CA) of symptomatic patients with AF by targeting the pulmonary veins (PV) is widely used as recommended by current guidelines [3]-[5]. Single shot techniques such as the cryoballoon (CB) technology have been developed to facilitate the ablation procedure and to improve outcome [6]-[8]. In the last years, CB based PV isolation (PVI) is increasingly performed [7],[8]. CB-CA has been proven to be non-inferior to the current gold standard of manual point by point mapping and radiofrequency (RF) CA for AF with respect to success and complication rates [6],[9]. Additionally, CB-CA has recently been shown to be an effective strategy for paroxysmal as well as persistent AF [4], [6]-[8].
The necessity of treating DM patients with AF is noticeably growing, as the prevalence of DM population increases [10]-[12]. DM has been associated with worse outcome after cardiac surgery and interventions [2],[10]. The abnormal glucose metabolism in DM patients, has been shown to affect the atrial substrate properties with an intra-atrial conduction delay and a low voltage area formation, resulting in higher AF recurrence rate after RF based CA [13]. Despite the fact that several non-randomized clinical studies have addressed the issue of CA in DM and have shown favorable rates of success [11],[12], little is known about the results of CB-CA in this population.
In this study, we aimed to assess the safety and efficacy of AF ablation using the second-generation CB (CB2, Arctic Front Advance, Medtronic, Inc. Minneapolis, MN, USA) in patients with DM compared to a non-DM group of patients and to analyze predictors of arrhythmia recurrence.
Between July 2015 and December 2017, 397 patients with symptomatic paroxysmal or persistent AF and an indication for AF ablation according to current guidelines, who were scheduled for PVI using the CB2, were enrolled in this retrospective single center study. Patients with left atrial (LA) thrombus, uncontrolled thyroid dysfunction, contraindication to anticoagulation, pregnancy, previous AF-ablation, severe valvular disease and a LA size >60 mm were excluded. Severity of symptoms was recorded according to European Heart Rhythm Association (EHRA) score. Informed consent was taken from each patient before the procedure. The study was in compliance with the principals outlined in the Declaration of Helsinki and approved by the local Ethics Committee (No. 17-298). Patients were divided into a study group with diagnosed type 1 or type 2 DM (DM group) and a control group (non-DM group).
Transesophageal echocardiography was performed in all patients prior to the procedure. Apart from echocardiography, no additional pre-procedural imaging was performed. In patients on vitamin K antagonists, anticoagulation was continued throughout the procedure aiming at an INR of 2-3. In patients treated with novel oral anticoagulants (NOACs), the drug was discontinued up to 24 hours prior to the procedure and and re-initiated 6 h post-ablation at half the regular dose, and at full dose the following day.
All procedures were performed under deep sedation using midazolam, fentanyl, and propofol [14]. One 10-pole diagnostic catheter (Webster® CS Uni-Directional, Biosense Webster, Inc., CA, USA) was introduced via the right femoral vein and positioned within the coronary sinus. A single transseptal puncture was performed via the right femoral vein under fluoroscopic guidance, using a modified Brockenbrough technique and an 8.5 French transseptal sheath (SL1, St Jude Medical, Inc., St Paul, MN, USA or PREFACE Biosense Webster). Heparin was administered after transseptal puncture to maintain an activated clotting time of ≥ 300 seconds. The transseptal sheath was then exchanged over a guidewire with a 12 French steerable sheath (Flexcath Advance, Medtronic). In order to identify all PV ostia, selective PV angiography was performed. In all patients, an esophageal temperature probe (Sensitherm, St Jude Medical, Inc. or CIRCA S-CATHTM) was inserted and positioned according to the individual CB position to facilitate esophageal temperature monitoring during energy delivery. All ablation procedures were conducted with the 28 mm CB2. The 28 mm CB2 was advanced into the LA via the 12 French steerable sheath and a spiral mapping catheter (20 mm diameter; Achieve, Medtronic) was advanced into the target PV to record electrical activity. The CB2 was inflated proximal to the PV ostium and gently pushed against the PV ostium to facilitate complete antral sealing. Contrast medium injected through the central lumen of the CB2 was used to verify complete occlusion of the PV ostium. Each freeze cycle duration lasted 180 seconds. If the time to PVI was longer than 60 seconds or no real time PV isolation recording could be obtained, 240 seconds freeze cycle and one more 180 seconds bonus freeze cycle were applied as per discretion of the operator. In patients demonstrating AF at the time of the procedure, electrical cardioversion was performed after the final freeze cycle and PVI was re-confirmed in sinus rhythm (SR). During energy delivery along the right PVs, continuous phrenic nerve pacing at maximum output and pulse width (12 mA, 2.9 ms) at a cycle length of 1000 ms was performed, using a diagnostic catheter positioned in the superior vena cava. Phrenic nerve capture was monitored by intermittent fluoroscopy and by tactile feedback of diaphragmatic contraction by the operator’s hand positioned on the patient’s abdomen. In addition, the continuous motor action potential (CMAP) was monitored. Refrigerant delivery was stopped immediately if weakening or loss of diaphragmatic movement, or the reduction of CMAP amplitude was noted. If phrenic nerve palsy (PNP) occurred, no additional freeze cycle was applied along the septal PVs. Cavotricuspid isthmus ablation (CTI), using an open irrigated radiofrequency catheter (Celsius ThermoCool or ThermoCool -SF, Biosense Webster), was solely performed in patients with documented or induced common type atrial flutter during the index procedure. PV abnormality was defined as left common ostium and/or a right middle PV.
Postprocedural management
Following ablation, all patients underwent transthoracic echocardiography to rule out a pericardial effusion. All patients were treated with proton-pump inhibitors twice daily after the procedure until discharge and once daily for 6 weeks thereafter. Anticoagulation was continued for at least 3 months and thereafter based on the individual CHA2DS2-VASC score. To prevent early recurrence, antiarrhythmic drugs (ADD) were administered throughout a 3-month blanking period (BP). Our institutional approach strongly recommends the administration of AAD and discontinuation after BP. Due to patient preference or referring physician preference AAD was continued. Follow-up was performed either by the outpatient clinic or the referring cardiologist at 3, 6, and 12 months after the index procedure, as well as in case of symptoms suggestive of arrhythmia recurrence and included a ≥24 h-Holter recording and interrogations of implanted devices, if present. Symptoms suggestive of recurrent atrial tachyarrhythmia (ATA) prompted additional outpatient clinic visits.
Repeat ablation was offered to patients with symptomatic ATA recurrence after the BP, or symptomatic drug-refractory recurrent ATA within the BP that could not be managed without intervention. Repeat procedure was performed using a 3D Navigation system (CARTO-3, Biosense Webster). PVs were assessed for reconnection and re-isolation of the PVs was performed in case of recovered conduction. In case of AT, electro-anatomical mapping and entrainment mapping were performed to verify the mechanism and to guide the following RF ablation.
The primary endpoint of this study was any episode of documented ATA recurrence lasting longer than 30 seconds after a 3-month BP or triggering a repeat ablation within the blanking period. Secondary endpoints were complications related to the procedure, such as pericardial tamponade, PNP, cerebrovascular events, and groin complications.
Continuous data are presented as mean ± standard deviation, skewed continuous parameters were expressed as median (interquartile range defined as Q1–Q3). Overall, 48 patients with DM were matched to 48 patients without DM. Propensity score matching was on a logistic regression model including age, sex, type of AF, hypertension, LVEF, LA size and prior transient ischemic attack (TIA)/stroke. Categorical data were summarized as frequencies and percentages and were compared using χ2 test. Comparisons between baseline characteristics were performed by independent Student’s t-test, Mann– Whitney rank-sum, Fisher exact, or χ2 tests where appropriate. To analyze the association between baseline and procedural parameters on AF recurrence, binary logistic regression analysis was used. Parameters that were found to be univariately associated with the outcome and those that show a slight association with the outcome with P<0.20 were included in the multivariable analysis. Kaplan Meyer and cox regression analysis were performed to describe ATA free survival. Statistical analyses were performed using SPSS statistical software (version 22.0; SPSS Inc., Chicago, IL, USA). A 2-tailed P<0.05 was considered statistically significant.
A total of 397 patients with paroxysmal (180/397; 45.4%) and persistent (217/397; 55.6%) AF [30 (7.5%) patients had concomitant documented typical atrial flutter] undergoing PVI using the 28mm CB2 were included in the analysis; 48/397 (12%) patients had DM. The control group was selected by propensity score matching from patients without DM. It was based on a logistic regression model including age, sex, type of AF, hypertension, LVEF, LA size, and prior TIA/stroke. Baseline characteristics of the study population are summarized in [Table 1]. Patients in the DM group had a higher CHA2DS2-VASC score (3.8± 1.5 vs 3.0±1.5 p=0.01) and a higher prevalence of previously known myocardial infarction (MI) (29.1% vs 12.5%, p=0.04). Additionally, there was a higher trend of persistent AF in DM group (72.9% vs 60.4 %, p=0.13). All targeted veins were isolated. Procedural characteristics are presented in [Table 2]. Anatomical PV abnormalities were observed in 6 patients (12.5%) in the DM group and 14 (29.1%) patients in the non-DM group (p=0.03). Total procedural time in the DM and non-DM groups were 127.5±38.3 vs 122.5±33.3 minutes (p=0.50).
Table 1. Baseline characteristics of study patients.
|
Non-DM |
DM |
P value |
| Number of patients |
48 |
48 |
|
| Age (years) |
69.5 ± 8.8 |
66.9 ± 9.5 |
0.18 |
| Male gender, n |
27 (56.2%) |
26 (54.1%) |
0.83 |
| Height (cm) |
173.2±10.6 |
174.8±10.1 |
0.42 |
| Weight (kg) |
85.9±16.7 |
93.7±15.7 |
0.04 |
| Hypertension, n |
42 (87.5%) |
44 (87.5%) |
0.50 |
| LA diameter (mm) |
41.1±8.2 |
41.3±9.7 |
0.93 |
| LVEF % |
51.2±8.1 |
48.6±8.6 |
0.12 |
| CHA2DS2VASc score |
3.0±1.5 |
3.8±1.5 |
0.01 |
| EHRA score |
2.5±0.6 |
2.5±0.6 |
0.82 |
| Previous Stroke/TIA,n |
4 (8.3%) |
5 (10.4%) |
0.54 |
| Previous MI, n |
6 (12.5%) |
14 (29.1%) |
0.04 |
| Cardiomyopathy, n |
9 (18.7%) |
11 (22.9%) |
0.31 |
| Non-ischeamic |
5 (10.4%) |
7 (14.5%) |
0.53 |
| Ischeamic |
4 (8.3%) |
4 (8.3%) |
1.00 |
| Mean duration of AF (months) |
16.8±21.9 |
20.8±23.9 |
0.40 |
| Follow up period (months) |
11.8±4.7 |
13.6±5.55 |
0.87 |
| Persistent AF, n |
29 (60.4%) |
35 (72.9%) |
0.13 |
LA: left atrium, LVEF: left ventricular ejection fraction, MI: myocardial infarction, PCI: percutaneous coronary intervention, CABG: coronary bypass grafting operation, AF: atrial fibrillation. EHRA: European heart rhythm association. TIA: transient ischemic attack. CTI: cavotricuspid isthmus.
Table 2. Procedural characteristics of study patients.
|
Non-DM |
DM |
P value |
| Number of patients |
48 |
48 |
|
| Additional CTI during the procedure, n |
2 (4.1%) |
2 (4.1%) |
1.00 |
| PV abnormality, n |
14 (29.1%) |
6 (12.5%) |
0.03 |
| LCPV |
8 (16.6%) |
3 (6.2%) |
0.10 |
| RMPV |
6 (12.5%) |
3 (6.2%) |
0.62 |
| Total procedure time (min) |
122.5±33.3 |
127.5±38.3 |
0.50 |
| Fluoroscopy time (min) |
24.1±10.7 |
27.8±10.8 |
0.61 |
| Freezes in LSPV (times) |
1.2±0.4 |
1.3±0.7 |
0.23 |
| Freezes in LIPV (times) |
1.3±0.5 |
1.2±0.4 |
0.20 |
| Freezes in RSPV (times) |
1.3±0.5 |
1.3±0.5 |
0.85 |
| Freezes in RIPV (times) |
1.6±0.7 |
1.3±0.5 |
0.06 |
| LSPV freeze duration (seconds) |
251.8±95.6 |
264.7±126.7 |
0.57 |
| LIPV freeze duration (seconds) |
266.7±121.2 |
252.5±93.9 |
0.47 |
| RSPV freeze duration (seconds) |
250.9±138.9 |
257.5±143.5 |
0.82 |
| RIPV freeze duration (seconds) |
317.2±157.1 |
282.2±131.2 |
0.23 |
| Minimum temperature in LSPV (°C) |
48.6±6.2 |
48.6±6.6 |
0.98 |
| Minimum temperature in LIPV(°C) |
46.9±6.5 |
46.8±7.3 |
0.84 |
| Minimum temperature in RSPV (°C) |
48.8±6.5 |
48.8±6.7 |
0.97 |
| Minimum temperature in RIPV (°C) |
47.6±6.2 |
45.5±6.6 |
0.99 |
| Balloon temperature <-60 °C (n) |
8 (14.11%) |
7 (12.2%) |
0.42 |
| Initial sinus rhythm, n |
316 (92.9%) |
52 (91.2%) |
0.4 |
| Cardioversion during procedure, n |
24 (50.0%) |
23 (47.9%) |
0.83 |
| Recurrence in blanking period, n |
9 (18.7%) |
8 (16.6%) |
0.78 |
| Late recurrence, n |
14 (29.1%) |
15 (31.2%) |
0.82 |
LIPV: left inferior pulmonary vein; LSPV: left superior pulmonary vein; RIPV: right inferior pulmonary vein; RMPV: right middle pulmonary vein; RSPV: right superior pulmonary vein.
Overall, during 12.7± 5.4 months of follow-up, single procedure success rate was 70.8 % in the DM group and 72.9% in the non-DM group (p=0.82) [Figure 1]. Early recurrence of ATA within the first 3 months after the index CB-CA occurred in 16.6% of patients in the DM group and 18.7% in the non-DM group (p=0.78). In multivariate analysis age, gender, hypertension, DM, PV abnormality, LA diameter, LVEF, cardioversion during procedure, early recurrence during the BP, Previous MI, cardiomyopathy of any type, documented atrial flutter, mean duration of AF and persistent AF, were included as these parameters had p<0.20 in univariate analysis [Table 3]. After multivariable Cox regression analysis, persistent AF (p=0.03) and early recurrence during the BP (p=0.001), but not DM, were found to be predictors of recurrence of ATA [Table 4]. AAD was prescribed to all patients after the index procedure.
Figure 1. Comparison of ATA free survival for DM vs non-DM patients
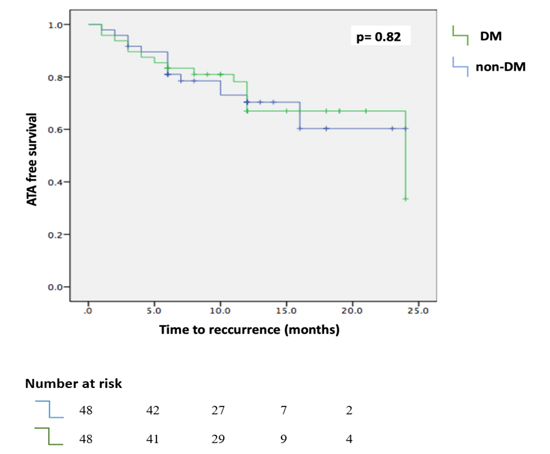
Table 3. Parameters included in univariate analysis
|
P value |
| Gender |
0.83 |
| Age |
0.79 |
| hypertension |
0.50 |
| PV abnormality |
0.03 |
| LA diameter |
0.32 |
| LVEF |
0.46 |
| Early recurrence during BP |
0.78 |
| Diabetes mellitus |
0.07 |
| Cardiomyopathy of any type |
0.31 |
| Persistent AF |
0.13 |
| Mean duration of AF |
0.26 |
| Documented atrial flutter |
0.56 |
| Previous MI |
0.04 |
| CV during procedure |
0.83 |
LA: left atrium, LVEF: left ventricular ejection fraction, MI: myocardial infarction, AF: atrial fibrillation, PV: pulmonary vein, CV: cardioversion, BP: blanking period.
Table 4. Cox regression analysis. Predictors of ATA recurrence after multivariate analysis.
|
β |
SE |
Wald |
df |
Significance level |
HR |
CI 0.95 HR |
| Recurrence in blanking period |
1.863 |
0.396 |
22.138 |
1 |
0.001 |
12.5 |
7.04 |
22.2 |
| Persistent AF |
1.171 |
0.543 |
4.662 |
1 |
0.03 |
2.45 |
1.41 |
4.27 |
SE: Standard error, df: degree freedom, HR: Hazard Ratio, AF: atrial fibrillation
Seven groin complications occurred (7 hematoma) in 4 DM patients and 3 in the non-DM group patients (p= 0.71). Transient PNP occurred in 4 patients in the DM group (4 vs 0, p=0.04). PNP had no effect on prolongation of hospital stay. All PNP cases resolved spontaneously during follow-up. In one case pericardial tamponade occurred, that was managed by pericardial puncture (one in the non-DM group, p=0.73), but hospital stay was not prolonged in this patient. There were no severe complications (procedure related deaths, atrio-esophageal fistula or cerebrovascular embolic events).
This retrospective analysis investigated the safety and efficacy of CB2-CA in DM patients with paroxysmal and persistent AF and assessed predictors of ATA recurrence. The current analysis demonstrates that CB2-CA is a feasible and safe procedure in DM patients with similar success and complication rates as compared to a non-DM population. Persistent AF and early recurrence during the BP were found as predictors of recurrence of ATA.
Diabetic patients with AF have a worse cardiovascular outcome than those without, regarding mortality and hospitalizations [15],[16]. Consequently, a rhythm control approach may be preferred in this group of patients to improve cardiovascular outcome [10],[17].
However, data investigating the effect of CB2-CA in DM patients were limited by non-randomized small studies and limited number of patients [2],[11],[17].
The long-term outcome of CA in patients with AF and DM was studied in a meta-analysis by Anselmino and colleagues [11]. In this meta-analysis 15 studies with 1464 patients were included. Mean follow-up was 27 months. The overall complication rate was between 1.5–5.0 % and overall long-term success rate of maintaining SR was 66 (58–73) %. Advanced age, higher body mass index, and higher basal glycated haemoglobin level were found to be predictors of recurrence of ATA. In our analysis, the complication and success rates were similar to previous publications without significant differences between the both groups [11], [18], [19].
Recently, Bogossian et al. evaluated the efficacy and safety of CA for AF and atrial flutter (AFlut) in patients with DM [2]. Between 2007 and 2010, 8175 patients who underwent CA for AFlut or AF were included in this multicenter prospective registry. Patients with DM (n=944) were compared to patients without DM (n=7231). After a follow-up period of 366 days, there was no significant difference between AF groups in terms of major adverse cardiac and cerebrovascular events. CA of AF was mostly done with RF CA. CB-CA was the most frequent alternative energy source (DM:17.1%, non-DM: 17.5%) [2].
The AF prevalence and the risk of AF recurrence are increased in patients with DM due to atrial remodeling and fibrosis [1], [10]. This correlation was also proven in our analysis where patients with DM presented more often with persistent AF and persistent AF was a predictor of recurrence of ATA. However, during our follow-up, single procedure success rate was similar between both groups. Similar ATA free survival in diabetic patients with more complex substrate can be attributed to additional ablative effects of CB-2. Besides ostial PV isolation, application of CB-2 results in large atrial lesion formation causing substrate modification, especially in the posterior LA wall. The spherical shape of CB-2 may cause a mismatch between the balloon and PV ostia during optimal contact causing additional atrial wall ablation that may eliminate components responsible for more diseased LA such as CFAEs, rotors, and vagal ganglia as well [20]. Recently the left atrial appendage was identified as a possible target for catheter ablation and radiofrequency and CB-2 based LAAI has been shown to improve clinical outcome. [21], [22], [23].
Phrenic nerve palsy was found to be more common in DM patients. In all cases, PNP resolved spontaneously during follow-up and did not prolonged hospital stay. However, DM itself may cause neuropathy and phrenic nerves of diabetic patients may be more sensitive to cryoenergy compared to nondiabetic individuals [1],[2].
The results of our analysis as well as of the abovementioned studies suggest that in patients with DM, CB2-CA is a safe and effective treatment strategy for symptomatic AF. Moreover, DM patients with good functional status and controlled glycemic levels [19] should be carefully evaluated and highly considered to receive CB2-CA with a favorable success rate and similar complication rate in comparison to non-DM patients.
There are some limitations of this study that need to be acknowledged. This analysis is retrospective in nature with moderate number of patients and without preprocedural cardiac imaging. Moreover, our follow-up did not include routine continuous monitoring with implanted devices or 7-day Holter recording and therefore our success rate may be overestimated. Nevertheless, follow up included 24-hour Holter monitoring, and/or device interrogations (if present), at three, six and 12 months. The effect of DM type, duration of DM, and HbA1c levels on CB2-CA outcome has not been assessed. Finally, no systematical esophagoscopy was performed in this study. Consequently, no data about the incidence of esophageal injury is available.
Conflicts of interests
CHH received travel grants and research grants by Medtronic, Pfizer, Novartis, Claret Medical, SentreHeart, Biosense Webster, Boston Scientific and Cardiofocus. He received speaker's honoraria from Cardiofocus, Boston Scientific and Novartis. RRT received travel grants from St. Jude Medical, Topera, Biosense Webster, Daiichi Sankyo, SentreHeart and speaker’s honoraria from Biosense Webster, Biotronik, Pfizer, Topera, Bristol-Myers Squibb; Bayer, Sano Aventis and research grants by Cardiofocus. CE received travel grants and educational grants by Medtronic. KHK received travel grants and research grants from Biosense Webster, Stereotaxis, Prorhythm, Medtronic, Edwards, Cryocath, and is a consultant to St. Jude Medical, Biosense Webster, Prorhythm, and Stereotaxis. He received speaker's honoraria from Medtronic. All other authors have no relevant disclosures.
Our data strengthen the value of CB2-CA for the treatment of AF as an effective and safe procedure in DM patients, with similar success and complications rates compared to non-DM population.
- Huxley RR, Filion KB, Konety S et al. Meta- analysis of cohort and case-control studies of type 2 diabetes mellitus and risk of atrial fibrillation. Am J Cardiol. 2011;108:56–62.
- Bogossian H, Frommeyer G, Brachmann J et al. Catheter ablation of atrial fibrillation and atrial flutter in patients with diabetes mellitus: Who benefits and who does not? Data from the German ablation registry. Int J Cardiol. 2016;214:25-30.
- Tilz RR, Heeger CH, Wick A et al. Ten-Year Clinical Outcome After Circumferential Pulmonary Vein Isolation Utilizing the Hamburg Approach in Patients With Symptomatic Drug-Refractory Paroxysmal Atrial Fibrillation. Circ Arrhythm Electrophysiol. 2018;11:e005250.
- Bertaglia E, Senatore G, De Michieli L et al. Twelve-year follow-up of catheter ablation for atrial fibrillation: A prospective, multicenter, randomized study. Heart Rhythm. 2017; 14:486-492.
- Kirchhof P, Benussi S, Kotecha D et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Europace. 2016;18:1609–1678.
- Heeger CH, Bellmann B, Fink T et al. Efficacy and safety of cryoballoon ablation in the elderly: A multicenter study. Int J Cardiol. 2019; 278:108-113.
- Heeger CH, Wissner E, Knöll M et al. Three-Year Clinical Outcome After 2nd-Generation Cryoballoon-Based Pulmonary Vein Isolation for the Treatment of Paroxysmal and Persistent Atrial Fibrillation - A 2-Center Experience. Circ J. 2017 ;81:974-980.
- Abdin A, Yalin K, Lyan E et al. Safety and efficacy of cryoballoon ablation for the treatment of atrial fibrillation in elderly patients. Clin Res Cardiol. 2019; 108:167-174.
- Kuck K-H, Brugada J, Fürnkranz A et al. Cryoballoon or Radiofrequency Ablation for Paroxysmal Atrial Fibrillation. N Engl J Med 2016; 374:2235-2245.
- Matta M, Saglietto A, De Salvo P et al. Catheter ablation in atrial fibrillation: is there a mortality benefit in patients with diabetes and heart failure? Herz. 2019; 44: 218–222.
- Anselmino M, Matta M, D'ascenzo F et al. Catheter ablation of atrial fibrillation in patients with diabetes mellitus: a systematic review and meta-analysis. Europace. 2015; 17: 1518–1525.
- Metzner A, Reissmann B, Rausch P et al. One-year clinical outcome after pulmonary vein isolation using the second-generation 28-mm cryoballoon. Circ Arrhythm Electrophysiol. 2014; 7:288–292.
- Chao TF, Suenari K, Chang SL et al . Atrial substrate properties and outcome of catheter ablation in patients with paroxysmal atrial fibrillation associated with diabetes mellitus or impaired fasting glucose. Am J Cardiol. 2010; 106:1615-20.
- Du X, NinomiyaT, de Galan B et al. Risks of cardiovascular events and effects of routine blood pressure lowering among patients with type 2 diabetes and atrial fibrillation: results of the ADVANCE Study. Eur HeartJ. 2009; 30:1128–1135.
- Tilz RR, Chun KRJ, Deneke T et al. Positionspapier der Deutschen Gesellschaft für Kardiologie zur Kardioanalgosedierung. Kardiologe. 2017;11:369–382.
- Fumagalli S, Said SA, Laroche C et al. Management and prognosis of atrial fibrillation in diabetic patients: an EORP-AF General Pilot Registry Report. Eur Heart J Cardiovasc Pharmacother. 2018; 4:172–179.
- Forleo GB, Mantica M, De Luca L et al. Catheter ablation of atrial fibrillation in patients with diabetes mellitus type2: results from a randomized study comparing pulmonary vein isolation versus antiarrhythmic drug therapy. J Cardiovasc Electrophysiol 2009;20:22–8
- Freeman JV, Tabada GH, Reynolds K et al. Contemporary procedural complications, hospitalizations, and emergency visits after catheter ablation for atrial fibrillation. Am J Cardiol. 2018; 121:602–608.
- Hijioka N, Kamioka M, Matsumoto Y et al. Clinical impact of insulin resistance on pulmonary vein isolation outcome in patients with paroxysmal atrial fibrillation. J Cardiovasc Electrophysiol. 2019; doi.org/10.1111/jce.13827.
- Yalin K, Abdin A, Lyan E, Sawan N, Liosis S, Elsner C, Jobs A, Brüggemann B, Koester J, Eitel I, Eitel C, Tilz RR. Safety and efficacy of persistent atrial fibrillation ablation using the second-generation cryoballoon. Clin Res Cardiol. 2018 Jul;107(7):570-577
- Yorgun H, Canpolat U, Kocyigit D, Coteli C, Evranos B and Aytemir K. Left atrial appendage isolation in addition to pulmonary vein isolation in persistent atrial fibrillation: one-year clinical outcome after cryoballoon-based ablation. Europace. 2017;19:758-768.
- Tilz RR, Liosis S, Vogler J, Reil JC, Eitel C and Heeger CH. Left atrial appendage thrombus formation less than 24 hours after empirical cryoballoon-based left atrial appendage isolation: A serious warning. HeartRhythm Case Rep. 2019;5:124-127.
- Heeger CH, Rillig A, Geisler D, Wohlmuth P, Fink T, Mathew S, Tilz RR, Reissmann B, Lemes C, Maurer T, Santoro F, Inaba O, Sohns C, Huang Y, Alessandrini H, Dotz I, Schluter M, Metzner A, Kuck KH and Ouyang F. Left Atrial Appendage Isolation in Patients Not Responding to Pulmonary Vein Isolation. Circulation. 2019;139:712-715.