Quantifying Risk Factors for Atrial Fibrillation: Retrospective Review of a Large Electronic Patient Database
Jaclyn Rivington1, Patrick Twohig2
1Department of Cardiology, University of Nebraska Medical Center, Omaha, NE, USA.2Department of Gastroenterology & Hepatology, University of Nebraska Medical Center, Omaha, NE, USA.
Despite the numerous comorbidities associated with atrial fibrillation (AF), the relative risk has been varying and not well-documented.
To quantify the risk of diseases associated with AF.
Population-based retrospective analysis in IBM Explorys (1999-2019), an electronic database with over 63 million patients in the United States. Odds ratios were calculated between AF and other diseases. AF patients were also stratified by age, gender, and race to assess trends of AF in different demographic groups.
1,812,620 patients had AF in the database. Congestive heart failure had the highest association with AF (OR 42.95). Cardiomyopathy, coronary artery disease, hypertension, and myocardial infarction all had odds greater than 15. Anemia of chronic disease and chronic kidney disease had odds greater than 18, the highest for chronic inflammatory conditions. Other conditions commonly associated with AF were found to have odds less than 8, including hyperthyroidism, alcohol use, and sleep apnea. Helicobacter pylori infection had the lowest odds at 1.98.
Epidemiologic information could be integrated with current clinical algorithms to more rapidly identify patients at risk of AF.
Key Words : Atrial fibrillation, Congestive heart failure, Inflammation, Risk factors, Epidemiology.
Jaclyn Rivington MD
Department of Cardiology, University of Nebraska Medical Center
682265 Nebraska Medical Center, Omaha, NE, USA , 68198
Atrial Fibrillation (AF) is the most common cardiac dysrhythmia 1,2. It occurs when a premature atrial complex (PAC) triggers an ectopic focus of electrical activity rapidly firing from the pulmonary veins, coronary sinus, ligament of Marshall or other atrial tissue 1,3-5. Over time, this can create microfibrosis that perpetuates refractory rhythms 6,7. Refractory cardiac rhythms are dangerous as they increase the risk for life threatening complications such as heart failure, thromboembolic events, hospitalization, and death.
AF affects approximately 6.1 million people in the United States (US) 1,2,8. The risk of AF increases with age, so it is estimated that 12.1 million people in the US will have AF by 2030 3,4,8-10. Patients typically present with heart palpitations, irregular heart rate, light-headedness, extreme fatigue, dyspnea, and chest pain 8. Treatment includes anticoagulation to reduce the risk of embolic events like stroke, along with medications and/or surgical interventions like radiofrequency ablation to treat ectopic foci that contribute to cardiac symptoms 8. Risk scores have been modelled, genetic links have been investigated, but due to the complexity of the disease, a full understanding is still unfolding 1,3. Comorbidities that have been commonly associated with an increased the risk of AF include hypertension, valvular heart disease, cardiomyopathy, coronary artery disease and sleep apnea, or stressors such as surgery, pulmonary embolism, alcohol use and hyperthyroidism 1,4. Previous literature has also investigated an association with Helicobacter pylori (Hp) infection 7,11,12.
Although multiple risk factors for AF have been identified, the relative risk of these factors has not been well-documented. By quantifying the relative risk of comorbid conditions associated with AF, clinicians may have greater awareness of risk factor modification and screening for patients with AF so that treatment can be initiated, and morbidity may be reduced.
Our study quantifies the risk of commonly associated conditions and chronic inflammatory states with AF.
Study Design and Database:
We performed a population-based retrospective analysis in the Explorys database (1999-2019), a large, nationwide, commercial, electronic database created by IBM Corporation, Watson Health (Somers, New York, United States of America 13. Explorys pools de-identified clinical data from electronic medical records, laboratories, practice management, and claims systems, and then matches this data using Unified Medical Language System ontologies to create unique patient records from 26 health care networks and 300 hospital systems across the United States 14. Data such as diagnoses, procedures, and medications are standardized according to common classification systems such as the International Classification of Diseases (ICD), Systemized Nomenclature of Medicine–Clinical Terms (SNOMED-CT), Logical Observation Identifiers Names and Codes (LOINC), and RxNorm 15-17. Explorys is a password-protected web application that uses a gateway server which is accessible at participating healthcare institutions to allow individuals to search and analyze the aggregated and de-identified patient data (https://popex.explorys.com) 13. All Explorys data meets the standards of the Health Insurance Portability and Accountability Act (HIPPA) and Health Information Technology for Economic and Clinical Health (HITECH) Act. As a result, ethical review by our Institutional Review Board (IRB) was not needed because there is no identifiable information associated with any of the patient data.
Our primary aim was to quantify the association between AF and numerous disease states or comorbidities that have been linked with an increased risk of AF. Our secondary aim was to assess the prevalence of AF in different demographic sub-groups including age, gender, and race in the Explorys database. To assess age, AF patients were stratified into 10 age groups by years (18-65, 65+, 20-29, 30-39, 40-49, 50-59, 60-69, 70-79, 80-89, 90+).
Using the Explorys cohort definition feature, we obtained population-level counts of the number of patients with and without a diagnosis of AF. Subsequent searches were performed separately using AF and each comorbidity included in [Table 1]. Only patients over 18 years old were included. Explorys utilizes a patient matching algorithm to ensure that each individual patient is only captured once in their lifetime within the database 18. Explorys also can define index events in order to establish temporal relationships 19,20. This feature ensures that patients diagnosed with AF would have had one of the comorbidities listed in [Table 1] at the time of diagnosis of AF.
Table 1. Odds ratio (with 95% confidence intervals) of having different diseases among patients with atrial fibrillation in the 63 million patient Explorys Darabase. All p-values <0.0001.
| Comorbidity |
Odds Ratio |
Confidence Interval (95%) |
| Congestive Heart Failure |
42.95 |
(42.79, 43.10) |
| Cardiomyopathy |
29.73 |
(29.63, 29.89) |
| Coronary artery disease |
23.64 |
(23.56, 23.72) |
| Hypertension |
20.84 |
(20.76, 20.91) |
| Anemia of chronic disease |
19.47 |
(19.31, 19.64) |
| Chronic kidney disease |
18.77 |
(18.70, 18.84) |
| Myocardial infarction |
17.54 |
(1747, 17.61) |
| Chronic obstructive pulmonary disease |
13.62 |
(13.57, 13.66) |
| Pulmonary embolism |
11.87 |
(11.78, 11.96) |
| Obstructive sleep apnea |
7.47 |
(7.44, 7.50) |
| Rheumatoid arthritis |
5.87 |
(5.83, 5.92) |
| Hyperthyroidism |
5.78 |
(5.72, 5.83) |
| Inflammatory bowel disease |
2.88 |
(2.85, 2.92) |
| Alcohol |
2.84 |
(2.82, 2.86) |
| Helicobacter pylori infection |
1.98 |
(1.68, 2.34) |
Population-level counts obtained from Explorys were arranged in 4x4 tables in Microsoft Excel©. Odds ratios (with 95% confidence intervals (CI)) were then calculated to determine the relationship between AF along with each risk factor listed in [Table 1]. Corresponding forest plots of this data were generated based on type of comorbidity, as either non-inflammatory [Figure 1] or inflammatory states [Figure 2]. We assessed the prevalence of each comorbidity in atrial fibrillation (# patients with comorbidity / # patients with AF), listed as total count and percentages [Table 3]. Similarly, we assessed the prevalence of atrial fibrillation in each comorbidity (# patients with comorbidity and AF / # patients with comorbidity), listed as total count and percentages [Table 4]. The distribution of patients identified in the database by age [Table 2], gender, and race was also investigated and recorded as total counts and percentages.
Figure 1. Forest plots of odds ratios of comorbidities with Atrial Fibrillation.
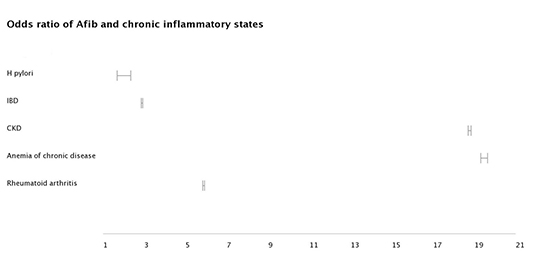
Figure 2. Forest plots of odds ratios of inflammatory conditions with Atrial Fibrillation.
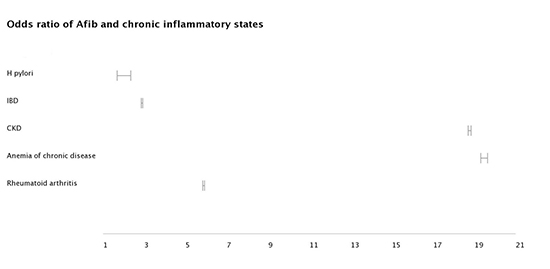
At the time of analysis there were 63,656,860 patients in the Explorys Database. Our search identified 1,812,620 (2.9%) adults (>18 years old) with AF. Multiple comorbidities were highly associated with AF ([Table 1]. Patients with congestive heart failure (CHF) had the highest risk of developing AF (OR 42.95, 95% CI 42.79, 43.10). Cardiomyopathy, coronary artery disease (CAD), hypertension, and myocardial infarction all had odds greater than 15 for AF. Anemia of chronic disease and chronic kidney disease (CKD) had odds greater than 18, the highest for chronic inflammatory conditions. Other conditions commonly associated with AF were found to have odds less than 8, including hyperthyroidism, alcohol use, and sleep apnea. Helicobacter pylori infection had the lowest odds of all comorbidities at 1.98 (95% CI 1.68, 2.34). Full results are listed in [Table 1].
Table 2. Relative incidence of atrial fibrillation by age.
| Age group (years) |
Total population size (count) |
Percentage of total patients (%) |
| 18-65 |
293,800 |
16% |
| 65+ |
1,470,640 |
81% |
| 20-29 |
5,770 |
<1% |
| 30-39 |
16,840 |
1% |
| 40-49 |
36,920 |
2% |
| 50-59 |
104,400 |
6% |
| 60-69 |
263,880 |
15% |
| 70-79 |
462,580 |
26% |
| 80-89 |
537,760 |
30% |
| 90+ |
364,630 |
20% |
Prevalence of comorbidities among AF patients
Hypertension was the most common comorbidity among AF patients (78.9% of AF population), followed by CAD (44.8%), CHF (36.7%), chronic obstructive pulmonary disease (COPD) (27.4%), and CKD (26.9%). With the exception of anemia of chronic disease, all of the comorbidities with an OR < 5 carried the lowest number of patients within the AF population. Patients with Hp had both the lowest OR with AF (OR 1.98, 95% CI 1.68, 2.34) as well as the smallest total population size in the database (150 patients). Full results are listed in [Table 3].
Table 3. Prevalence of different comorbidities in the atrial fibrillation population (1,812,620 patients).
| Comorbidity |
Total population size (count) |
Percentage of total patients (%) |
| Congestive Heart Failure |
664,740 |
36.7% |
| Cardiomyopathy |
274,680 |
15.2% |
| Coronary artery disease |
821,610 |
44.8% |
| Hypertension |
1,429,810 |
78.9% |
| Anemia of chronic disease |
85,380 |
4.7% |
| Chronic kidney disease |
488,460 |
26.9% |
| Myocardial infarction |
382,170 |
21.1% |
| Chronic obstructive pulmonary disease |
496,630 |
27.4% |
| Pulmonary embolism |
97,710 |
5.4% |
| Obstructive sleep apnea |
350,180 |
19.3% |
| Rheumatoid arthritis |
70,920 |
3.9% |
| Hyperthyroidism |
57,920 |
3.2% |
| Inflammatory bowel disease |
22,890 |
1.3% |
| Alcohol |
64,970 |
3.6% |
| Helicobacter pylori infection |
150 |
0.0083% |
Prevalence of AF among comorbidities
Patients with CHF have the highest prevalence of AF (44% of CHF patients also have AF), followed by cardiomyopathy (41.9%), anemia of chronic disease (34.6%), and myocardial infarction (28.9%). Patients with Hp had the lowest proportion of patients with AF (5.5%), followed by alcohol (7.3%) and inflammatory bowel disease (IBD) (7.5%). Full results are listed in [Table 4].
Table 4. Prevalence of atrial fibrillation among different comorbidities.
| Comorbidity |
Population with AF (count) |
Total population size (count) |
Percentage of total patients (%) |
| Congestive Heart Failure |
664,740 |
1,511,710 |
44% |
| Cardiomyopathy |
274,680 |
654,790 |
41.9% |
| Coronary artery disease |
821,610 |
2,981,440 |
27.8% |
| Hypertension |
1,429,810 |
11,112,560 |
12.9% |
| Anemia of chronic disease |
85,380 |
246,880 |
34.6% |
| Chronic kidney disease |
488,460 |
1,716,960 |
26.1% |
| Myocardial infarction |
382,170 |
1,338,410 |
28.9% |
| Chronic obstructive pulmonary disease |
496,630 |
2,215,210 |
22.4% |
| Pulmonary embolism |
97,710 |
402,090 |
24.3% |
| Obstructive sleep apnea |
350,180 |
2,329,210 |
15% |
| Rheumatoid arthritis |
70,920 |
502,810 |
14.1% |
| Hyperthyroidism |
57,920 |
419,630 |
13.8% |
| Inflammatory bowel disease |
22,890 |
304,050 |
7.5% |
| Alcohol |
64,970 |
887,390 |
7.3% |
| Helicobacter pylori infection |
150 |
2,720 |
5.5% |
Eighty-one percent of AF were in adults older than 65 years, with 80-89 years being the most prevalent age group [Table 2]. Fifty-three percent of patients in our population were male. Eighty-two percent of AF patient were Caucasian, 8% were African American, and <2% were Hispanic or Asian.
Although multiple conditions have been associated with an increased risk of AF, our retrospective cohort electronic database study of over 1.8 million patients quantifies the odds of having AF with different comorbidities.
Non-inflammatory conditions
Previous have indicated that structural heart disease and chronic inflammation increase the risk of AF 1,3-4,21. Our population cohort analysis confirmed findings of prior studies, as CHF and cardiomyopathy were the two comorbidities with the highest odds of AF. The suspected mechanism for why these conditions increase the risk of AF is because patients with CHF and cardiomyopathy have significant cardiac remodeling, which leads to myocardial fibrosis, which promotes the development of ectopic electrical foci in the heart 22.
Hyperthyroidism promotes hyperdynamic circulation and increased workload on the heart 23. Individuals who have pre-existing heart disease experience further impairment of cardiac function under these circumstances, which can impair heart function leading to heart failure and AF 23. Additionally, hyperthyroidism can shorten the repolarization phase in atrial tissue, which can further promote the development of AF 24.
Chronic Obstructive Pulmonary Disease (COPD)
Reduced lung function has been shown to be an independent risk factor for AF 25. Although the exact mechanism how COPD increases the risk of AF is unclear, it is known that the most common source of ectopic beats that initiate AF are in the pulmonary veins 26. It is therefore possible that changes in chronic hypoxia, pulmonary hypertension, and cor pulmonale that are caused by COPD could then predispose to AF 25. Given the close link between reduced lung function and ischemic heart disease, it is possible that a common physiologic pathway exists for AF as well 27-31.
Increased consumption of alcohol, especially in the setting of binge drinking, has been shown to cause conduction delays and depressed cardiac performance, which leads to cardiac remodeling and greater susceptibility to AF 32.
Pulmonary embolism likely predisposes to AF in multiple ways, but a major mechanism is suspected to be secondary to increased right-heart pressure and volume overload 33. Additional processes like increased inflammation and hypercoagulability are also strongly suspected to contribute to the increased risk of AF in patients with PE 33.
Multiple chronic inflammatory states have been linked to an increased risk of AF, including anemia of chronic disease, chronic kidney disease, inflammatory bowel disease, autoimmune conditions such as rheumatoid arthritis, and infection with Helicobacter pylori 21,34. Persistent inflammation, oxidant stress, apoptosis, and fibrosis of cardiomyocytes leads to remodeling which promotes the development of AF 35-40. The results of our study confirmed that these conditions do increase the odds of developing AF. In the Explorys database, anemia of chronic disease and chronic kidney disease were two inflammatory conditions with odds greater than 18, which was higher than other more commonly associated risk factors for AF such as hyperthyroidism, alcohol use, and sleep apnea.
Inflammatory bowel disease (IBD)
Patients with IBD have demonstrated prolonged atrial conduction times and other physiologic dysfunction that promote AF 37,38. Additionally, increases in serum inflammatory markers such as c-reactive protein, erythrocyte sedimentation rate, and interleukin-6 levels have been observed in patients with AF, which are commonly elevated in IBD patients 41-43.
Chronic kidney disease (CKD)
Patients with CKD have increased activation of the renin-angiotensin-aldosterone system, which modulates total body volume, electrolyte balance via modulation of ion channels, and activation of the sympathetic nervous system which all result in structural and electrical remodeling in the heart that can promote AF 44-49.
There are two suspected mechanisms by which infection with Hp predisposes to AF. First, Hp causes an increased production of autoantibodies to the H+/K+-ATP enzyme, which damages atrial cells and delays cardiac depolarization, which can trigger AF 50. Second, Hp can produce cytotoxic proteins that stimulate gastric epithelial cells to produce inflammatory markers such as interleukin-8, which promotes systemic inflammation that can predispose to AF 51.
Previous retrospective studies have suggested that there may be a link between AF and Hp 7,11,12. Although the odds of developing AF after having Hp was increased in our population (OR 1.98, 95% CI 1.68, 2.34), these odds are minimally increased and show the lowest association of all other comorbidities included in this study population.
Prevalence of AF and comorbidities
Prior studies have indicated that patients with an MI have AF at a prevalence of 6-10% 52. Our study found a significantly higher risk of AF in MI patients at 28.9%. Cardiomyopathy has been found to co-occur in up to 28% of AF patients 53. We found that 41% of patients with cardiomyopathy also had a diagnosis of AF. Hyperthyroidism has been reported in up to 8% of AF patients 23. We found that 13% of hyperthyroid patients had AF in our cohort.
The risk of AF has been shown to increase with age 10,54. The results of our analysis confirm this finding, as the highest prevalence of AF was found in individuals aged 80-89 years. One difference with the results of our analysis was that >50% of patients in our database were over the age of 75 years. Prior studies had approximately 45% of patients over the age of 75 years 10,54. Identifying those at risk for AF may allow for earlier intervention and reduction of morbidity and mortality, as patients may become less optimal candidates for treatment with increased age (cite).
Caucasians and males have also been shown to be more prevalent in prior studies evaluating the epidemiology of AF. Our results are consistent with this finding, but we had significantly more Caucasian and male patients with AF than prior studies, which listed 2.2% Caucasian and 1.5% African American patients 10, compared to 82% and 8% in our study, respectively. For gender, prior studies found 1.1% vs. 0.8% in favor of male 54, but we found 53% vs. 47%. The reasoning behind why these sub-populations are at greater risk of AF is not entirely understood and may be an area for future research.
The main strengths of our study include the large sample size of over 1.8 million patients with AF and the population-based study design which included more than 63 million patients from across the United States. We present data that highlights the relative association between AF and comorbidities that have been suggested to increase the risk of AF. We also provide a unique perspective that assesses the prevalence of comorbidities in AF patients, rather than just what comorbidities increase the risk of AF.
The main limitations of the Explorys database include its retrospective nature, and reliance on appropriate diagnostic codes being used by clinicians, as incorrect coding has been shown to occur up to 18% of the time 19. Further, the Explorys database is comprised of population level data, so individual patient data regarding a diagnosis and risk factors is not available.
Millions of patients are hospitalized in the US due to AF annually. Understanding the relative risk of predisposing conditions may help improve their management and therefore limit the burden that they pose to patients and the health care system. By quantifying risk factors for AF using a retrospective cohort of 1.8 million patients, we hope to more identify conditions that need high-priority control in reducing the morbidity of AF.