Psychogenic Pseudosyncope: Clinical Features, Diagnosis and Management
Alessandra Alciati1,2, Dana Shiffer3, Franca Dipaola1,3, Franca Barbic1,3, Raffaello Furlan1,3
1Department of Biomedical Sciences, Humanitas University, Rozzano, Milan, Italy.2Department of Clinical Neurosciences, Hermanas Hospitalarias, Villa S. Benedetto Menni Hospital, Albese con Cassano (CO), Italy.3Internal Medicine, Humanitas Clinical and Research Center-IRCCS, Rozzano, Milan, Italy.
Psychogenic Pseudosyncope (PPS) is the appearance of Transient Loss of Consciousness (TLOC) in which movements are absent, but there are no hemodynamic and electroencephalographic modifications as are induced by gravitational challenges which characterize syncope and true loss of consciousness.
For younger and adult populations, a detailed history is crucial for the diagnosis. Clinical clues that should raise the suspicion for PPS include prolonged duration of the LOC, eye closure during the episode, unusual triggers, no recognizable prodromes and the high frequency of attacks. The presence of an established diagnosis of syncope should not deter from the concomitant diagnosis of PPS. The gold standard for a proper diagnosis of PPS is the documentation by a tilt test of normal hemodynamic and electroencephalographic parameters, when recorded during an attack.
Treatment of PPS, based on the clear and empathetic communication of the diagnosis, can lead to an immediate reduction of attack frequency and lower the need to call on emergency services. Pharmacological treatment of associated psychiatric disorders and psychological interventions may be beneficial in patients with PPS. Cognitive-behavioural therapy holds the most reliable evidence of efficacy.
In the present review, we aimed to address PPS with historical aspects, main clinical features and diagnostic tests, current diagnostic classification, underlying neurobiological abnormalities, management and therapy.
Key Words : Syncope, Pseudosyncope, TLOC, Conversion Disorders, Cognitive Behavioural Therapy.
Correspondence to: Dr. Raffaello Furlan, M.D.Humanitas University, Humanitas Clinical and Research Center - IRCCSVia A. Manzoni, 56, 20089 Rozzano, Italy.
Psychogenic pseudosyncope (PPS) is an apparent loss of consciousness (LOC) in the absence of impaired cerebral perfusion or function. The prevalence of PPS in patients presenting for syncope evaluations has been reported from 0% to 12 %, with a mean rate of 4% 1,2. This range of frequency likely represents an underestimation as PPS may account for a significant proportion of the so-called ‘unexplained syncope’, i.e. syncope undiagnosed after an extensive evaluation. Notably, these episodes account for 20–30 % of cases observed in tertiary syncope clinics.
Figure 1. Representative example of multiparameters recording during a PPS attack in a young female undergoing a tilt test maneuver. The End graph (right panel) refers to the last 4 minutes of the PPS episode. Note absence of alterations in the hemodynamics, i.e. heart rate and blood pressure, and in the respiratory rate during the attack (right panel). In addition, during tilt, there was a proper increase of heart rate and TPR, blood pressure was unmodified and CO declined compared to the supine position, as expected. The only feature suggestive of the patient’s distress was the respiratory pattern. Indeed, this latter was more irregular (see ETCO2 trace) during PPS attack than in baseline supine position and during early tilt (left panel). Vertical dashed lines indicate the start (left) and the end (right) of the tilt maneuver.
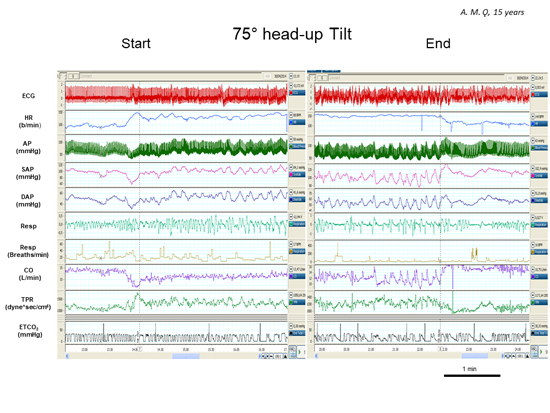
HR indicates heart rate; AP, arterial pressure; SAP, systolic arterial pressure; DAP, diastolic arterial pressure; Resp, respiratory activity; CO, cardiac output; TPR, total peripheral resistances; ETCO2 , end-tidal carbon dioxide partial pressure.
There is a historical evolution of the concept of PPS that seems to start from the Egyptians, in 1900 BCE. They described a condition suggestive of hysteria, which they attributed to spontaneous movement of the uterus within a female’s body. The Greek physician, Hippocrates, also believed that the illness was caused by the movement of the uterus (“hysteron”) and coined the term hysteria 3. Aulus Cornelius Celsus, a Roman medical writer (1st century BC) described a condition that “completely destroys the senses that on occasions the patient falls, as if in epilepsy”. Aulus Cornelius Celsus also stated “this case, however, differs in that the eyes are not turned, nor does froth issue forth, nor are there any convulsions: there is only a deep sleep” 4. In the middle ages, hysteria-related behaviour was framed as demonic possession and this view even culminated in the execution of 19 young “possessed” women in the village of Salem (Massachusetts) in 1692. This happened despite the new medical understandings and developments introduced from the beginning of the 16th century. In 1680, the English physician, Thomas Sydenham, recognized for the first time, that hysteria may simulate almost all forms of organic diseases.
In the 19th century, the French neurologist, Jean-Martin Charcot, theorized that the functional motor symptoms were due to a “dynamic lesion”, adversely impacting motor pathways and studied the effectiveness of hypnosis on hysteria. Sigmund Freud, the founder of psychoanalysis, coined the term “conversion hysteria” highlighting the emergence of physical symptoms as an attempt to resolve or to communicate unconscious and unbearable psychic conflicts, often of sexual origin (psychic conflicts “converted” into physical symptoms).
The French psychologist Pierre Janet, Freud’s contemporary, theorized an important role for dissociation, framed as a “retraction of the field of personal consciousness”, in the psychological underpinnings of conversion disorder 5. By the late 20th century, various and often contradictory concepts of dissociation were suggested. Currently, dissociation is used to describe a wide range of phenomena in which behaviour, thoughts and emotions may become separated one from another.
In the following part of the present review, main clinical features and diagnostic tests, current diagnostic classification, underlying neurobiological abnormalities, management and therapy of PPS will be addressed.
Clinical Features and Diagnostic Tests
Transient LOC (TLOC) is the core presentation of PPS, but it is shared by two common clinical entities, vasovagal syncope (VVS) and epileptic seizures (ES). Despite clear pathophysiological differences of the various causes of TLOC (VVS is caused by transient global cerebral hypoperfusion whereas ES is related to abnormal paroxysmal neuronal electrical discharges), the similarity of clinical presentation leads to a misdiagnosis rate as high as 30% 6.
A detailed history is central for the diagnosis of PPS and its differentiation from VVS which is the most frequent cause of syncope in the absence of cardiovascular disease. Due to its transient nature, TLOC is rarely witnessed by medically trained individuals, but an eyewitness account is often crucial for a correct diagnosis 7.
For this reason, several studies have sought to identify the clinical features that can distinguish PPS from VVS. An analysis of 800 tilttable tests (TTT) indicated that the median duration of apparent TLOC was longer in PPS (44 seconds) than in VVS (20 seconds); the eyes were closed during the event in almost all PPS (97%) but in only 7% of VVS 8. Jerking movements occurred more frequently in VVS while a sudden head drop, as the tilt table moved down, was more common in PPS. A retrospective evaluation of patients referred to a syncope unit revealed that those with PPS had a high frequency of attacks (53±35 attacks) during the preceding year, whereas patients with VVS had a median number of syncopal events preceding the observation of 3–6 episodes per year 1.
Saal et al 9 demonstrated that more than half of the patients with the final diagnosis of PPS also experienced true syncopal episodes. The patients with a combination of tilt-induced VVS and PPS, compared with patients with pure VVS, had greater attack frequency, apparent LOC lasting more than one minute, ictal eye closure, atypical triggers (exercise, or supine position in the absence of predisposing factors such as venepuncture or pain) and the absence of prodrome 10. Those with VVS had symptoms and physical signs including pallor, sweating, nausea, asthenia and dizziness, most likely related to autonomic imbalance preceding LOC.
The 2018 European Society of Cardiology Guidelines for the diagnosis and management of syncope 7 reported other clinical features highly suggestive of PPS, in particular, the sleep-like body position with closed eyes, resistance to eye-opening, eyelid flickering, eyeball movements, lack of response to speech or touch, swallowing, and intact muscle tone. In a pediatric population, prolonged syncope duration, presence of upright posture and short QT dispersion, an index of ventricular repolarization, were independent predictors of PPS 11,12. In a prospective study of a young patient population (ages 10-21), ≥20 lifetime fainting spells, ≥2 fainting spells in a single day, self-reported loss of consciousness ≥2 minutes, and tearfulness associated fainting were predictors associated with PPS 13. In contrast, two or more typical prodromal symptoms such as lightheadedness, dizziness, blurred vision, nausea, and sweating predicted VVS. In a study on a similar population, Heyer and colleagues 14 suggested that symptom descriptions helped distinguish patients with PPS from those with true syncope. In particular, an account of sleepiness or imminent sleep with fainting should raise suspicion for PPS.
The 2017 American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society (ACC/AHA/HRS) Guideline for the Evaluation and Management of Patients with Syncope stated that TTT is reasonable to establish a diagnosis of PPS 15.
Among ES, the atonic seizure, characterized by a sudden loss or diminution of muscle tone without an apparent preceding myoclonic or tonic event, can be a challenging clinical presentation to be properly differentiated from VVS and PPS. An electroencephalogram (EEG) is commonly used to diagnose ES, by displaying abnormal paroxysmal activities correlated with clinical symptoms. However, because in up to 6.6% of cases ‘‘epileptiform’’ activities may be observed in healthy subjects 16, the patients with unexplained TLOC and ‘‘epileptiform’’ activities on an EEG outside the critical episode can be wrongly considered as been affected by epilepsy, and therefore incorrectly treated with antiepileptic drugs.
To overcome this risk, ACC/AHA/HRS 2017 guidelines 15 suggested that the additional simultaneous monitoring of EEG to hemodynamic parameters recording during a TTT can make possible the differentiation among PPS, VVS and ES whenever a diagnosis cannot be established after a thorough initial evaluation. In this context, PPS attacks occurs mostly within 2 minutes after TTT and are associated with no decrease in blood pressure (BP) or significant changes in heart rate (HR) Table 1. Usually, BP and HR increase few minutes before PPS, reaching peak values during the attack 7. This pattern differs remarkably from that of VVS, where at least BP or HR decrease; more often, both decline before syncope Table 1. Regardless of the cause for syncope (i.e. vasovagal, cardiac or hypotensive), the EEG may indicate characteristic features and stereotyped changes, which appear to reflect the cerebral hypoperfusion. EEG modifications include the slowing of background rhythms followed by high-amplitude delta activity, mainly in the anterior leads recordings. The documentation of a patient’s unresponsiveness along with the lack of abnormally slow electrical activity and a normal alpha rhythm of the brain, suggests a psychogenic nature of the episode 17.
Table 1. Differences between VVS and PPS as far as objective parameters, clinical features and yearly number of episodes are concerned.
|
|
Objective Parameters
(during TTT)
|
TLOC Clinical Features
|
Average number of attacks per year
|
|
|
Hemodynamic parameters
|
EEG
|
Eyes closure
|
Duration TLOC
|
|
|
|
Blood pressure
|
Heart rate
|
Alpha rhythm
|
|
|
|
|
VVS
|
Decreased
|
Decreased
|
Slowing
|
7%
|
<1minute
|
Low (4±2 per year)
|
|
PPS
|
Increased or normal
|
Slightly increased or normal
|
Normal
|
97%
|
>1minute
|
High (53±35 per year)
|
PPS, psychogenic pseudosyncope; TLOC, transient loss of consciousness; TTT, Tilt-Table Testing; VVS, vasovagal syncope
In this context, Ninni and colleagues 18 showed that a combined TTT and video EEG recording in 101 patients with unexplained atypical TLOC, with syncope and seizure characteristics, enabled a diagnosis in 68 cases (67%). VVS was diagnosed in 59 and PPS in 9 patients. Of note, most of these patients had remained undiagnosed after the first-line investigation. The diagnostic yield of a combined TTT/video EEG approach could be considered high in patients previously undiagnosed in accordance with data reported by Laroche et al 19 in a similar population.
Because several syncope units lack prompt and easy access to an EEG and most of the time simply rely on demonstration of the absence of hypotension during the attack for the diagnosis of PPS, a near-infrared spectroscopy (NIRS) was recently proposed as a simple, non-invasive tool for continuous monitoring of cerebral perfusion during TTT, in the evaluation of suspected PPS. Claffey and colleagues 20 showed that cerebral perfusion, detected by NIRS, was unchanged despite the presence of patients’ subjective symptoms at the time of a PPS episode that occurred on a TTT. The latter was associated with a concomitant normal increase of BP and HR.
Current Diagnostic Classification
The World Health Organization diagnostic system International Classification of Diseases (ICD)-10 placed PPS under the category of dissociative (conversion) disorders in which the term “dissociative” implies compartmentalization or detachment of neurological functioning from the normal awareness. The ICD-11 eliminated the term “conversion” from the grouping title and coined the definition of dissociative neurological symptom disorder. This is presented as a single disorder with twelve subtypes based on the predominant neurological symptom, but none of these subtypes explicitly refers to the specific clinical picture of PPS 21.
However, the fifth and latest edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) 22 included PPS in the Conversion Disorder (CD) (Functional Neurological Symptom Disorder, FND) (CD/FND) diagnosis, under the category “Somatic symptoms and related disorders”. According to the DSM- 5 diagnostic criteria, CD/ FND is characterized by presence of one or more symptoms of altered voluntary or sensory function with clinical findings providing evidence of incompatibility between the symptom and recognized neurological or medical conditions. The symptom or deficit must not be explained by another medical or mental disorder and it must cause clinically significant distress or impairment in social and occupational activities.
Conversely, with an evidence for faking, the diagnoses should be factitious disorders or malingering. Compared to the previous DSM (DSM IV) criteria, the DSM-5 diagnosis of CD/FND added the criterion of physical diagnostic features, removed criteria requiring an association with psychological stressors and the exclusion of malingering or factitious disorder (because the absence of faking may not be reliably discerned). These changes, which moved this disorder away from being considered a diagnosis of exclusion, have made DSM-5 CD/FND diagnosis criteria appropriate for research studies due to the potential for greater inter-rater reliability and compatibility with specialty-specific diagnoses. Besides PPS, examples of CD/ FND include paralysis, functional movement disorders (FMD), blindness, non-dermatomal sensory deficits and psychogenic nonepileptic seizures (PNES).
PNES is a paroxysmal alteration of sensory and/or motor function that resembles epileptic seizure but does not show corresponding abnormalities in brain electrical activity. It is the same neurobehavioural condition as PPS, but motor activity is far more evident in PNES as suggested by Heyer et al 23, who compared clinical features between tilt-induced PPS and EEG-confirmed PNES in a cohort of young patients. PNES episodes are briefer than PPS events (median: 45 versus 201.5 seconds, respectively), had less negative motor signs, such as, head drop and body limpness (20% versus 85% , respectively), while the positive motor signs of convulsion occurred more often with PNES (90% versus 30%). Behavioral arrest and eye closure did not differ between PPS and PNES. These studies support the concept that the clinical features of PPS may resemble those of VVS whereas the features of PNES appear more like epileptic seizures. Thus, PPS and PNES likely represent a unique psychiatric disorder and differ from each other primarily in terms of clinical features and referral patterns.
Neurobiology of Conversion /Functional Neurological Symptoms Disorders
The reframing of CD/FND as a disorder that can be diagnosed by physical signs and the recent availability of brain imaging techniques, enabled the exploration of the neurobiology underpinning CD/ FND. The neuroimaging data reported in this section, were derived from studies on PNES and other CDs/FNDs, with the understanding that these disorders, likewise PPS, are different clinical expressions of the same psychopathological disorder.
Spence and colleagues 24 used positron emission tomography to test the central question whether patients with CD/FND are feigning their symptoms. They compared patients, diagnosed with CD/FND arm weakness, with two reference groups. One was instructed to mimic the patients’ deficit, the other to move naturally, without any restriction. If symptoms were feigned, similar patterns of brain activation would be expected between the patients and the group that was instructed to mimic arm weakness. Instead, distinctive activation in the left dorsolateral prefrontal cortex (DLPFC), a brain region specifically activated by the internal generation (i.e. ‘choice’) of action, was observed between groups, suggesting that patients with conversion weakness were not simply faking their symptoms.
Subsequent neuroimaging studies in patients with CD/FND have consistently identified a hyperactive amygdala to emotional stimuli and an increased coupling to supplementary motor area (SMA) 25,26,27. SMA is implicated in the subjective urge to move and the intention to move. The finding of a hyperactive amygdala and its heightened connectivity with motor circuitry at brain functional magnetic resonance imaging, may represent one of the underlying mechanisms by which strong emotions may directly influence motor control.
The right temporo-parietal junction (TPJ) is a further brain area in which neuroimaging studies have shown altered activity and functional connectivity in PNES 28 and other motor FND populations 29. A characteristic feature of CD/FND is the impairment of self-agency, i.e. the subjective experience of causing one’s actions and predicting the motor outcome. Indeed, patients frequently report a lack of voluntary control over their abnormal movements or behaviours 30. It has to be pointed out that right TPJ plays a critical role in the selfagency, acting as a detector of discrepancies between motor intentions and motor consequences 31. Thus, it has been hypothesized that the hypo-activation and diminished right TPJ-sensorimotor functional connectivity found in patients with CD/FND, could be one of the neurobiological mechanisms underpinning the impaired sense of self-agency, typical of CD/FND 32-33.
It is now accepted that brain activity is organized into functional networks of regions showing synchronous activity over time. These “brain networks” are characterized by brain regions (nodes) and connections (edges) linking them. Aberrant brain networks may arise from damaged neural nodes or edges 34. Structural and functional neuroimaging studies showed the involvement of salience network (SN) in the pathophysiology of CD/ FND. Core nodes of SN are in the anterior cingulate cortex, middle cingulate cortex, bilateral anterior insula, and in specific regions of the dorsolateral prefrontal cortex. In addition to these cortical nodes, the SN also includes nodes in the amygdala, hypothalamus, ventral striatum, thalamus, and dopaminergic brainstem nuclei 35.
SN is involved in detecting internal and extrapersonal "salient stimuli", namely those drawing attention for being unexpected or novel, as well as interoceptive representations of the physiological state of the body and in facilitating rapid access to the motor system to appropriately guide behaviour 36. In CD/FND, structural and functional alterations in the SN network were revealed by neuroimaging studies, and seem to underlie the lack of integration of affective, cognitive and viscero-somatic information, contributing to a network-mediated “functional unawareness” in patients with CD/ FND 37-39. Thus, emerging CD/FND neurobiology indicates that this condition may reflect a multi-network disorder rather than a focal brain abnormality.
Management of Psychogenic Pseudosyncope
Management of PPS includes three different stages: 1) diagnosis; 2) communication of the diagnosis; and 3) treatment.
The diagnosis of psychogenic pseudosyncope
An early diagnosis and a brief symptom duration are linked to CD/FND better outcome 40 although, the average time to diagnosis is often quite long, i.e., more than 7 years in PNES 41. Besides the strict diagnostic process of PPS described above, a formal psychiatric assessment should be provided with the purpose to rule out similar psychiatric disorders like panic attacks, and to recognize and treat possible psychiatric comorbidities. In PPS patients, data on the prevalence of associated psychiatric disorders are lacking. However, because the rates of psychiatric disorders in in individuals with unexplained syncope, which may roughly correspond to patients with PPS, ranged from 24 to 39% 42,43, we infer it to be greater in patients affected by PPS than in the general population 44. Anxiety, somatization, major depression, and panic attacks were recognized as the more frequent psychiatric diseases related to unexplained syncope 45. A prospective controlled study by Koukham and colleagues 46 revealed a psychiatric disorder, mainly anxiety and panic attacks, in 65% of patients presenting with unexplained syncope , a rate significantly higher than that observed in a control group of patients referred for arrhythmia.
In a recent study, the self-reporting psychometric questionnaire symptoms checklist-90-revised was used to screen for psychiatric symptoms in 43 patients with single or recurrent VVS or unexplained syncope and 124 healthy controls 47. Comparison between patients and controls revealed that somatization scores were significantly greater in patients than in controls. Moreover, average scores for depression, anxiety, and somatization were significantly greater in individuals experiencing six attacks or more, thus supporting the hypothesis of an association between the recurrence of syncope and the greater deterioration of patients’ psychiatric symptom profile. Previous investigations have clearly shown that psychiatric symptoms at baseline predicted higher rates of unexplained syncope and VVS recurrence during follow-up, with a clear positive effect of psychiatric interventions on syncope outcomes and response to conventional therapy 46,48,49.
In addition to psychiatric diagnoses, a careful evaluation of adverse life events should be performed, based on the evidence that psychological interventions, such as the cognitive behaviour therapy (CBT) with a trauma focus (CBT-T), as well as eye movement desensitization and reprocessing techniques (EMDR), resulted in a remarkable reduction of symptoms and improvement of the qualityof-life in individuals who had to cope with traumatic life events 50,51. Notably, patients with CD/FND have increased rate of general trauma history, with a relationship between magnitude of trauma experience and the severity of symptoms. A recent systematic review and meta-analysis reported that stressful life events and maltreatment occurring in childhood or adulthood were more common in patients with CD/FND than in healthy controls 52. A recent observational longitudinal study 53 revealed that childhood sexual abuse is associated with significantly worse treatment outcome in CD/FND.
Communicating the diagnosis
Data from studies on PNES populations suggested that patients may benefit from being informed clearly and empathically of the diagnosis. Unfortunately, the long-term outcome of PPS is still far from being elucidated. Currently, there is only one retrospective cohort study of 35 patients with PPS referred to a tertiary centre for syncope that revealed a reduction in the number of attacks, with onethird of patients who were attack-free at the follow-up of >4 years 9. Importantly, conveying the diagnosis to the patient resulted in an immediate decrease in the number of attacks within one month and a shift from somatic to mental health care. However, the quality-oflife was still poor for both attack-free patients and those who were still symptomatic, suggesting that the underlying psychopathology negatively impacts the quality-of-life, more than the mere presence of PPS attacks. These findings are in line with the results of longitudinal epidemiological studies showing that CD/FND symptoms persisted or recurred in 39%–70% of cases and were associated with a poor quality-of-life 54,55.
The positive effects of a clear explanation are strongly in keeping with extensive data obtained from a PNES population. In newly presenting, video EEG-confirmed PNES patients, half were seizurefree at 3 months after the presentation of PNES diagnosis and, for most of them, PNES ceased immediately thereafter suggesting a specific therapeutic effect of the diagnosis communication itself. However, diagnosis communication seemed to have a greater shortterm impact on healthcare utilization than on seizure clinical control40. The decrease in health care utilization was consistent with a reduction of PNES-related use of emergency services up to 69% and of diagnostic test costs by 76% in the presence of unmodified rate of attacks.
Studies detailing a supposed optimal communication strategy for diagnosis delivering, tended to agree with the need to present PPS/PNES as a common and recognizable condition, independent of the patient’s self-consciousness and control, frequently related to upsetting emotions most of which the patient might be completely unaware. However, it is important to emphasize that most patients hardly accept the diagnosis of CD/FND, as they are afraid that the lack of physical causes may be perceived as a sign of malingering. Some diagnostic labels, in particular those containing the prefix “pseudo” may represent an additional obstacle to the diagnosis acceptance 56,57.
Treatment of psychogenic pseudosyncope
Psychotherapy is currently viewed as the treatment of choice for PPS/PNES. Cognitive behavioral therapy (CBT) is the psychological intervention supported by the most solid evidence. CBT combines cognitive therapy with behaviour therapy by identifying faulty or maladaptive patterns of thinking, abnormal emotional response or behaviours, and substituting them with assumed desirable patterns. CBT includes education about functional neurological disorders and the stress response, trains patients in stress management techniques, and helps them to recognize and change unhelpful thought patterns that reinforce their symptoms.
The importance of CBT in the treatment of CD/FND derives mainly from studies in patients with PNES with no systematic studies in PPS populations. The CBT approach to the treatment of PNES is based on a “fear avoidance” model. PNES, as well as PPS, are viewed as dissociative responses to cognitive, emotional, physiological or environmental cues that patients tend to associate with previously intolerable or fearful experiences. Dissociative responses are maintained by the avoidance of conditions that can trigger the attacks. This model of PNES maintenance supports the use of a series of standard CBT interventions, including graded exposure to avoided situations, emotion-regulation strategies, and problem-solving techniques. The support of potential efficacy of CBT in PNES has come from small uncontrolled studies and two randomized controlled trials which have shown that structured CBT significantly reduced attack frequency as well as the level of depression and anxiety symptoms compared to standard medical care 58,59.
A Cochrane review concluded that there was poor evidence supporting the use of a specific treatment, including CBT, as therapeutic option for PPS/PNES 60. It is worth noting that several studies have shown that CD/FND is associated with neurocognitive impairments in several domains, particularly attention, working memory, verbal and visual memory, visuospatial functioning, and information processing speed 61-63. It has been hypothesized that the cognitive impairment can interfere with the possibility that patients with CD/FND profit from CBT effectively, due to the potential negative impact of altered attention, memory and information processing speed on the learning processes required by a successful CBT treatment.
Regarding pharmacological treatments, a Cochrane systematic review and meta-analysis of 26 placebo-controlled studies investigated the pharmacological interventions for somatoform disorder 64. The results showed no evidence of a significant difference between tricyclic antidepressants and placebo and very low-quality evidence for new-generation antidepressants being effective in reducing the severity of medically unexplained physical symptoms in adults when compared with placebo. These conclusions further support the view that psychopharmacological intervention in PPS should be based on treatment of the identified psychiatric comorbidities, if any.
PPS is a disorder with a serious impact on the patient’s qualityof-life and a delay in diagnosis may adversely affect the outcome. Importantly, PPS is not a factitious disorder, a malingering where the patient is faking it. Although the biological mechanisms underpinning PPS are far from being elucidated, the progress of neuroimaging enables an initial understanding of the mechanisms underlying the detachment of neurological functioning from the patient’s awareness. The simultaneous monitoring of an EEG and hemodynamic parameters during TTT, may offer a diagnostic “goldstandard” with high levels of certainty. The diagnostic assessment of PPS should be completed with an evaluation and treatment of psychiatric comorbidity. There is some evidence that CBT is beneficial on CD/FND.
-
Walsh KE, Baneck T, Page RL, Brignole M, Hamdan MH. Psychogenic pseudosyncope: not always a diagnosis of exclusion. Pacing Clin Electrophysiol 2018; 41:480–486.
- Raj V, Rowe AA, Fleisch SB, Paranjape SY, Arain AM, Nicolson SE. Psychogenic pseudosyncope: diagnosis and management. Auton Neurosci 2014; 184:66–72.
- Cosmacini G. The long art: the history of medicine from antiquity to the present. 1997; Rome: Oxford University Press
- Penso G. Roman medicine. 3rd ed. Noceto 2002: Essebiemme.
- Pérez-Rincón H. Pierre Janet, Sigmund Freud and Charcot's psychological and psychiatric legacy. Front Neurol Neurosci 2011; 29: 115-24.
- Leach JP, Lauder R, Nicolson A, Smith DF. Epilepsy in the UK: misdiagnosis, mistreatment, and undertreatment? The Wrexham area epilepsy project. Seizure 2005;14:514–20.
- Brignole M, Moya A, de Lange FJ, Deharo JC, Elliott PM, Fanciulli A, Fedorowski A, Furlan R, Kenny RA, Martín A, Probst V, Reed MJ, Rice CP, Sutton R, Ungar A, van Dijk JG; ESC Scientific Document Group 2018 ESC Guidelines for the diagnosis and management of syncope. Eur Heart J 2018; 39:1883–1948
- Tannemaat MR, van Niekerk J, Reijntjes RH, Thijs RD, Sutton R, van Dijk JG. The semiology of tilt-induced psychogenic pseudosyncope. Neurology 2013;81:752–758.
- Saal DP, Overdijk MJ, Thijs RD, van Vliet IM, van Dijk JG. Long-term follow-up of psychogenic pseudosyncope. Neurology 2016;87:2214– 2219.
- Blad H, Lamberts RJ, van Dijk GJ, Thijs RD. Tilt-induced vasovagal syncope and psychogenic pseudosyncope: Overlapping clinical entities. Neurology 2015;85:2006–2010.
- Zhang Z, Jiang X, Han L, Chen S, Tao L, Tao C, Tian H, Du J. Differential Diagnostic Models Between Vasovagal Syncope and Psychogenic Pseudosyncope in Children. Front Neurol 2020;10:1392.
- Sucu M, Ozer O, Davutoglu V, Ercan S, Yuce M, Coskun FY. Relationship between neurocardiogenic syncope and ventricular repolarization. Pacing Clin Electrophysiol 2015; 38:625–629.
- Heyer GL, Harvey RA, Islam MP. Comparison of specific fainting characteristics between youth with tilt-induced psychogenic nonsyncopal collapse versus reflex syncope. Am J Cardiol 2017;119:1116e1120.
- Heyer GL Atypical Prodromal Symptoms Help to Distinguish Patients With Psychogenic Nonsyncopal Collapse Among Youth Referred for Fainting. Pediatr Neurol 2019;95:67-72.
- Shen WK, Sheldon RS, Benditt DG, Cohen MI, Forman DE, Goldberger ZD, Grubb BP, Hamdan MH, Krahn AD, Link MS, Olshansky B, Raj SR, Sandhu RK, Sorajja D, Sun BC, Yancy CW. 2017 ACC/AHA/HRS Guideline for the evaluation and management of patients with syncope: executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Circulation 2017; 136: e25–59.
- So EL. Interictal epileptiform discharges in persons without a history of : what do they mean?. J Clin Neurophysiol 2010;27:229–238.
- Benbadis SR, Chichkova R. Psychogenic pseudosyncope: an underestimated and provable diagnosis. Epilepsy Behav 2006; 9:106–10.
- Ninni S, Kouakam C, Szurhaj W, Baille G, Klug D, Lacroix D, Derambure P. Usefulness of head-up tilt test combined with video electroencephalogram to investigate recurrent unexplained atypical transient loss of consciousness. Arch Cardiovasc Dis 2019;112:82-94.
- LaRoche S, Taylor D, Walter P. Tilt table testing with videoEEG monitoring in the evaluation of patients with unexplained loss of consciousness. Clin EEG Neurosci 2011;42:202-205.
- Claffey P, Pérez-Denia L, Rivasi G, Finucane C, Kenny RA. Near-infrared spectroscopy in evaluating psychogenic pseudosyncope-a novel diagnostic approach.QJM 2020 ;113:239-244.
- Reed GM, First MB, Kogan CS, Hyman SE, Gureje O, Gaebel W, Maj M, Stein DJ, Maercker A, Tyrer P, Claudino A, Garralda E, Salvador-Carulla L, Ray R, Saunders JB, Dua T, Poznyak V, Medina-Mora ME, Pike KM, Ayuso-Mateos JL, Kanba S, Keeley JW, Khoury B, Krasnov VN, Kulygina M, Lovell AM, de Jesus Mari J, Maruta T, Matsumoto C, Rebello TJ, Roberts MC, Robles R, Sharan P, Zhao M, Jablensky A, Udomratn P, Rahimi-Movaghar A, Rydelius PA, Bährer-Kohler S, Watts AD, Saxena S. Innovations and changes in the ICD-11 classification of mental, behavioural and neurodevelopmental disorders. World Psychiatry 2019;18 :3-19.
- American Psychiatric Association: Diagnostic and Statistical Manual of Mental disorders, 5th ed (DSM-5). Washington, DC, American Psychiatric Association, 2013.
- Heyer GL, Albert DV, Weber A, Gedela S, Vidaurre J. Comparison of semiologies between tilt-induced psychogenic nonsyncopal collapse and psychogenic nonepileptic seizures. Epilepsy Behav 2016;62:171e175.
- Spence SA, Crimlisk HL, Cope H, Ron MA, Grasby PM. Discrete neurophysiological correlates in prefrontal cortex during hysterical and feigned disorder of movement. Lancet 2000; 355:1243–1244.
- Aybek S, Nicholson TR, O'Daly O, Zelaya F, Kanaan RA, David AS. Emotion-motion interactions in conversion disorder: an FMRI study. PLoS One 2015; 10:e0123273
- Voon V, Brezing C, Gallea C, Ameli R, Roelofs K, LaFrance WC Jr, Hallett M. Emotional stimuli and motor conversion disorder. Brain 2010; 133: 1526–1536.
- Hassa T, Sebastian A, Liepert J, Weiller C, Schmidt R, Tüscher O. Symptom-specific amygdala hyperactivity modulates motor control network in conversion disorder. NeuroImage: Clinical 2017; 15: 143–150.
- McSweeney M, Reuber M, Levita L. Neuroimaging studies in patients with psychogenic nonepileptic seizures: A systematic meta-review. Neuroimage Clin 2017; 16:210–221.
- Voon V, Cavanna AE, Coburn K, Sampson S, Reeve A, LaFrance WC Jr. Functional Neuroanatomy and Neurophysiology of Functional Neurological Disorders (Conversion Disorder). J Neuropsychiatry Clin Neurosci 2016; 28:168–190.
- Kranick SM, Hallett M. Neurology of volition. Exp Brain Res 2013; 229:313–327.
- Decety J, Lamm C. The role of the right temporoparietal junction in social interaction: how low-level computational processes contribute to meta cognition. Neuroscientist 2007;13:580–593.
- Farrer C, Frey SH, Van Horn JD, Tunik E, Turk D, Inati S, et al. The angular gyrus computes action awareness representations. Cereb Cortex 2008; 18:254–261.
- Desmurget M, Sirigu A. A parietal-premotor network for movement intention and motor awareness. Trends Cogn Sci 2009; 13:411–419.
- Passingham RE, Stephan KE, Kötter R. The anatomical basis of functional localization in the cortex. Nature Rev Neurosci 2002, 3.8: 606-616.
- Seeley WW, Menon V, Schatzberg AF, Keller J, Glover GH, Kenna H, Reiss AL, Greicius MD Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci 2007; 27:2349-2356.
- Menon V, Uddin LQ. Saliency, switching, attention and control: a network model of insula function Brain Struct Funct 2010; 214: 655–667.
- Voon V, Brezing C, Gallea C, Hallett M. Aberrant supplementary motor complex and limbic activity during motor preparation in motor conversion disorder. Mov Disord 2011; 26:2396– 2403.
- Czarnecki K, Jones DT, Burnett MS, Mullan B, Matsumoto JY. SPECT perfusion patterns distinguish psychogenic from essential tremor. Parkinsonism Relat Disord 2011; 17:328–332.
- Perez DL, Dworetzky BA, Dickerson BC, Leungc L, Cohnc R, Basletc G, Silbersweiget DA. An integrative neurocircuit perspective on psychogenic nonepileptic seizures and functional movement disorders: neural functional unawareness. Clin EEG Neurosci 2015; 46: 4–15.
- McKenzie P, Oto M, Russell A, Pelosi A, Duncan R. Early outcomes and predictors in 260 patients with psychogenic nonepileptic attacks. Neurology 2010; 74:64–69.
- Reuber M. The etiology of psychogenic non-epileptic seizures: toward a biopsychosocial model. Neurol Clin 2009;27:909-924.
- Kapoor WN, Fortunato M,Hanusa BH, Schulberg HC. Psychiatric illnesses in patients with syncope. Am J Med 1995; 99: 505–512.
- Koening D, Linzer M, Pontinen M, Divine GW. Syncope in young adults: evidence for a combined medical and psychiatric approach. J Int Med 1992; 232:169–176.
- Linzer M, Felder A,Hackel A, Perry AJ, Varia I, Melville ML, Krishnan KR. Psychiatric syncope: a new look at an old disease. Psychosomatics 1990; 31: 181–188.
- Linzer M,Varia I, Pontinen M, Divine GW, Grubb BP, Estes NA 3rd. () Medically unexplained syncope: relationship to psychiatric illness. Am J Med 1992; 92:18s–25s.
- Kouakam C, Lacroix D, Klug D, Baux P, Marquie C, Kacet S. Prevalence and prognostic significance of psychiatric disorders in patients evaluated for recurrent unexplained syncope. Am J Cardiol. 2002;89: 530–535.
- Alhuzaimi A, Aljohar A, Alhadi AN, Aljenedil A, Hersi AS. Psychiatric traits in patients with vasovagal and unexplained syncope. Int J Gen Med. 2018;11:99-104.
- D'Antono B, Dupuis G, St-Jean K, Lévesque K, Nadeau R, Guerra P, Thibault B, Kus T. Prospective evaluation of psychological distress and psychiatric morbidity in recurrent vasovagal and unexplained syncope. J Psychosom Res. 2009;67:213–222.
- Ventura R, Maas R, Ruppel R, Stuhr U, Schuchert A, Meinertz T, Nienaber CA. Psychiatric conditions in patients with recurrent unexplained syncope. Europace. 2001;3(4):311–316.
- Newton JL, Kenny RA, Baker CR. Cognitive behavioural therapy as a potential treatment for vasovagal/neurocardiogenic syncope – a pilot study. Europace. 2003;5(3):299–301.
- Lewis C, Roberts NP, Andrew M, Starling E, Bisson JI. Psychological therapies for post-traumatic stress disorder in adults: systematic review and meta-analysis. Eur J Psychotraumatol. 2020 ;11:1729633.
- Ludwig L, Pasman JA, Nicholson T, Aybek S, David AS, Tuck S, Kanaan RA, Roelofs K, Carson A, Stone J. Stressful life events and maltreatment in conversion (functional neurological) disorder: systematic review and meta-analysis of case-control studies. Lancet Psychiatry 2018;5:307-320.
- Van der Feltz-Cornelis CM, Allen SF, Van Eck van der Sluijs JF. Childhood sexual abuse predicts treatment outcome in conversion disorder/functional neurological disorder. An observational longitudinal study. Brain Behav 2020;10: e01558.
- Martlew JPJ, Marson A G . Psychological and behavioural treatments for adults with non-epileptic attack disorder. Cochrane Database Syst Rev 2014 ;11(2): CD006370.
- Régny P, Cathébras P. Conversion disorder in an internal medicine department: A series of 37 cases. Encephale French 2016 ; 42: 150–155.
- Plug L, Sharrack B, Reuber M. Seizure, fit or attack? The use of diagnostic labels by patients with epileptic or non-epileptic seizures. Appl Ling 2009; 31: 94–114.
- Dworetzky B. Perceptions in PNES: A Bidirectional Problem. Epilepsy Curr 2019;19:31-32.
- Goldstein LH, Chalder T, Chigwedere C, Khondoker MR, Moriarty J, Toone BK, Mellers JD. Cognitive-behavioral therapy for psychogenic nonepileptic seizures: a pilot RCT. Neurology 2010; 74:1986–1994.
- LaFrance WC Jr, Baird GL, Barry JJ, Blum AS, Frank Webb A, Keitner GI, Machan JT, Miller I, Szaflarski JP; NES Treatment Trial (NEST-T) Consortium. Multicenter pilot treatment trial for psychogenic nonepileptic seizures: a randomized clinical trial. JAMA Psychiatry 2014; 71: 997–1005.
- Martlew J, Pulman J, Marson AG. Psychological and behavioural treatments for adults with non-epileptic attack disorder. Cochrane Database Syst Rev 2014; 2: CD006370.
- Brown LB., Nicholson TR, Aybek S, Kanaan RA, David AS. Neuropsychological function and memory suppression in conversion disorder. J Neuropsychol 2014; 8:171–185.
- Kozlowska K, Palmer DM, Brown KJ, Scher S, Chudleigh C, Davies F, Williams LM. Conversion disorder in children and adolescents: A disorder of cognitive control. J Neuropsychol 2015; 9: 87–108.
- de Vroege L, Koppenol I, Kop WJ, Riem MME, van der Feltz-Cornelis CM. Neurocognitive functioning in patients with conversion disorder/functional neurological disorder. J Neuropsychol 2020 Mar 29. [Epub ahead of print]
- Kleinstäuber M, Witthö¬ M, Steanowski A, van Marwijk H, Hiller W, Lambert MJ. Pharmacological interventions for somatoform disorders in adults. Cochrane Database Syst Rev 2014;11: CD010628.