Defining Cardiac Dysautonomia – Different Types, Overlap Syndromes; Case-based Presentations
Suzanne Feigofsky1, Artur Fedorowski2,3
1Iowa Heart Center, Carroll, Iowa 51401, US.2Department of Clinical Sciences, Lund University, 214 28 Malmö, Sweden.3Department of Cardiology, Skåne University Hospital, 214 28 Malmö, Sweden.
The cardiovascular branch of autonomic nervous system (ANS) is responsible for the regulation of heart rate, blood pressure, and maintaining homeostasis during physiological stress such as exercise and standing upright. ANS constantly controls the rate and force of heart contractions and the vascular tone with the aim to maintain the sufficient tissue perfusion with oxygenated blood and secure venous return to the heart. Dysautonomias, result of ANS malfunction, are often found in patients with cardiovascular symptoms. Apart from the most prevalent one, arterial hypertension, the cardiovascular dysautonomic continuum encompasses other important although less known conditions: postural orthostatic tachycardia syndrome, inappropriate sinus tachycardia, orthostatic hypotension and reflex syncope. Moreover, heart diseases may evoke autonomic imbalance by themselves; cardiac pump failure is usually associated with sympathetic hyperactivity, neuroendocrine vasopressor activation, higher heart rate, reduced heart rate variability and baroreflex hyposensitivity, all of which are predictors of adverse outcomes.
Cardiologists and electrophysiologist frequently see patients for the evaluation and management of unexplained syncope, orthostatic intolerance, heart rhythm abnormalities and symptoms of palpitations. Recognizing the presence of cardiac dysautonomia is an important skill which is necessary for the appropriate evaluation and treatment of these patients. Clinical presentations may overlap, and the importance of a thorough history cannot be over-emphasized. In this review we will present a cases of a patients with cardiac dysautonomia which is illustrative of a typical patient experience, followed by a review of the autonomic nervous system and discussion of prevalence, clinical presentation, and pathophysiology of common cardiac dysautonomias.
Key Words : Atrial fibrillation, Autonomic nervous system, Autonomic dysfunction, Risk factors, Obesity.
Correspondence to: Suzanne Feigofsky, MD Clinical Cardiac Electrophysiologist Iowa Heart Center 405 S Clark St, Suite 205 Carroll Iowa, 51401
Autonomic control of cardiovascular system
The autonomic nervous system (ANS) consists of the sympathetic, parasympathetic and enteric components 1. The cardiovascular branch of ANS is responsible for regulation of heart rate, and blood pressure, and to maintain homeostasis during physiological stresses including exercise and standing upright 2. The two crucial elements controlled by ANS are the heart (“the pump”) and the vessels (“the pipelines”), smoothly interacting in normal conditions Figure 1. The appropriate level of systemic blood pressure is governed by the peripheral baroreceptor system and cardiovascular center in the medulla oblongata 3. The ANS constantly regulates heart rate, cardiac contractility and arterial and venous vascular tone to maintain sufficient tissue perfusion with oxygenated blood and to secure venous return. The cerebral circulation is especially protected by the high-pressure baroreceptor reflex mediated through carotid sinus sensors 3,4 and the self-adapting system of cerebral autoregulation 5.
Figure 1. Global blood pressure control and spectrum of cardiovascular dysautonomias. Two major types of cardiovascular autonomic dysfunction, cardiac i.e. “pump” dysfunction and vascular i.e. “pipeline” dysfunction may overlap, combine and convert to each other over the life span. For instance, patients with arterial hypertension may have orthostatic and postprandial hypotension, and reduced heart variability. Younger individuals may suffer from both POTS and VVS. Apart from global autonomic circulatory disorders, vascular bed may be affected in special areas, such as cerebral (e.g. migraine), coronary (e.g. spasm angina), and peripheral zones (e g. Raynaud’s disease) as well as in the venous capacitance vessels (abnormal venous pooling). The etiologies of cardiovascular dysautonomias are heterogenous and span over neurodegenerative diseases (e.g. Parkinson’s disease), chronic conditions such as diabetes and renal failure, chronic inflammatory states such autoimmune diseases (e.g. Sjögren’s disease), cardiovascular diseases (e.g. essential hypertension, a cardiovascular dysautonomia per se, and heart failure) and processes associated with aging. Genetic predisposing factors and rare genetic diseases may also contribute. (Figure adapted from Ricci et al 2015 (27)). Red colour – hyperadrenergic conditions/ sympathetic dominance. Blue colour – hypoadrenergic conditions/ parasympathetic dominance. POTS, postural orthostatic tachycardia syndrome; VVS, vasovagal syncope; CSS, carotid sinus syndrome.
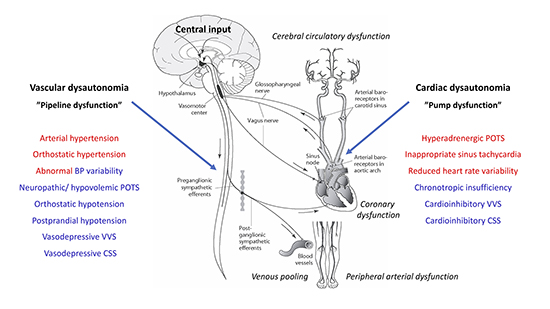
Malfunction of cardiovascular hemostasis regulated by the ANS is known as cardiac dysautonomia1. This process may be paroxysmal or chronic. Failure of either the sympathetic or parasympathetic system can result in unopposed action of the other. The most common cardiac manifestations of dysautonomia Figure 1 include reflex syncope, inappropriate sinus tachycardia, and syndromes of orthostatic intolerance: orthostatic hypotension (OH) and postural orthostatic tachycardia syndrome (POTS)1,6. In some chronic conditions, such as neurodegenerative diseases, diabetes, kidney failure, and autoimmune diseases, an umbrella term of cardiovascular autonomic neuropathy (CAN) is used 7. Cardiovascular autonomic neuropathy may typically manifest as OH, reduced heart rate variability, inadequate (resting) sinus tachycardia, and exercise intolerance 8. Similar autonomic abnormalities can be frequently found in heart failure, characterized by increased sympathetic drive, neuroendocrine vasopressor activation, higher heart rate, reduced heart rate variability and baroreflex hyposensitivity, all of which are predictors of adverse outcomes.9 Importantly, cardiovascular dysautonomia may also affect local circulation, leading to cerebral, coronary and peripheral circulatory dysfunction Figure 1. Furthermore, even arterial hypertension and chronotropic insufficiency can be seen as a part of cardiovascular dysautonomic continuum, although their etiologies are disputable10. Different types of cardiovascular dysautonomia may coexist; patients with orthostatic hypotension may have chronotropic insufficiency, whereas patients with POTS may have inappropriate sinus tachycardia (IST) and recurrent vasovagal syncope. Moreover, dysautonomias may coexist with primary cardiac diseases. For instance, OH may accompany heart failure, atrial fibrillation and ischemic heart disease, complicating the appropriate management of these conditions and worsening prognosis.11,12
Cardiologists are not always aware of cardiac dysautonomia as their main focus is usually on primary cardiac disorders such as heart failure, coronary artery disease, arrhythmias and hypertension, the latter not seen as a dysautonomia but rather as a potent cardiovascular risk factor. However, being alert to the possibility of cardiac dysautonomia is very important, especially when dealing with cardiovascular patients presenting with unusual symptoms and apparently normal vital parameters and ECG. In this article, we define the most common forms of cardiac dysautonomia and outline the adequate diagnostic methods for detection of cardiac dysautonomia in clinical practice
Different types of cardiac dysautonomia
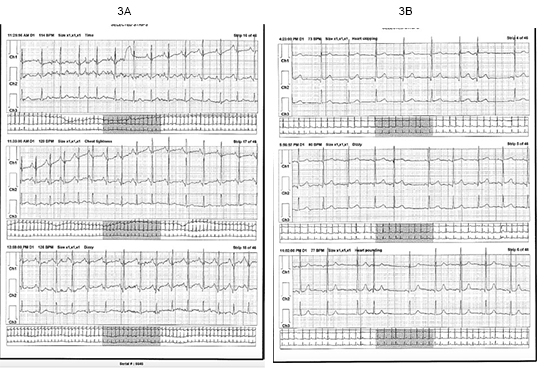
A 25-year-old Caucasian female presented for a “second opinion” regarding palpitations, dizziness, and near syncope. Symptoms began at age 12 with onset of menarche. She reports a long history of digestive issues, abdominal and pelvic pain thought due to irritable bowel syndrome. Beginning at age 20, she developed night sweats. Frequent rashes and redness of her skin involving the axillae, face and chest and urticaria of her legs have been a problem. Figure 2. Flecainide and a beta-blocker were tried for palpitations thought due to premature atrial contractions (PACs) and, for this underwent an ablation but symptoms continued. External monitoring correlated symptoms with sinus tachycardia and continued PACs Figure 3a 3b. Atenolol and metoprolol resulted in hypotension and fatigue. Midodrine and metoprolol, midodrine and propranolol, and digoxin and diltiazem drug combinations were ineffective. Ivabradine provided some benefit but ocular migraines limited its use.
Figure 2. Pictures of a typical rash involving the axillae and lower extremities of a woman with Postural Orthostatic Tachycardia Syndrome.
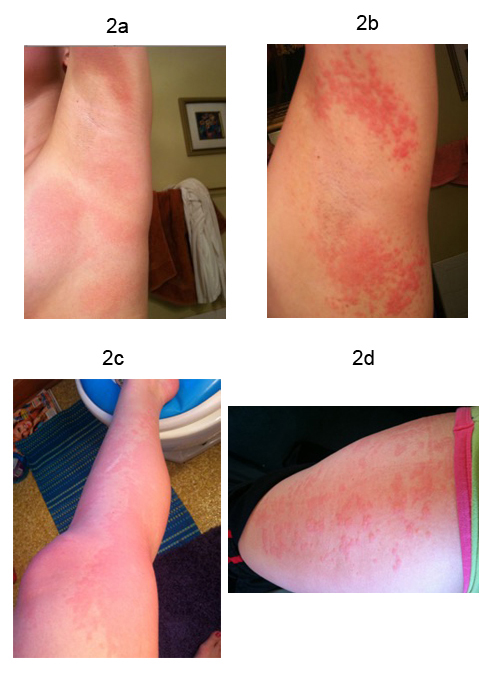
Figure 3. Symptomatic events from a Holter monitor in a patient with Postural Orthostatic Tachycardia Syndrome.
Due to continued symptoms and documented PAC’s, she underwent another electrophysiology study that was negative except for an exaggerated heart rate response to isoproterenol. Autonomic testing was normal. Tilt table testing was normal; however, the patient was wearing compression garments during the study. Figure 4. It was recommended that she increase hydration, salt and wear compression garments.
Figure 4. Results (in table format) of autonomic testing performed at a tertiary institution on a patient with Postural Orthostatic Tachycardia Syndrome.

At the time of referral, despite drinking 3 liters of water and 6 salt tablets daily, and Pilates 5 days/week, orthostatic intolerance made it impossible for her to continue working as a nurse or even grocery shopping. After 3-5 minutes of upright posture, scotomata, diaphoresis and dizziness occurred which improved with squatting or laying down. Symptoms are worse in the morning and after eating a large meal. She has self-documented standing heart rates as high as 180 bpm within 3 minutes of standing with a finger pulse oximeter (video supplement). As a result of heat intolerance and night sweats, she has begun sleeping with ice packs.
Orthostatic vital signs at the time of her initial consultation Table 1:
She was sent for supine and upright norepinephrine levels, a 24- hour urine for catecholamines, and she was referred to an allergist for possible Mast Cell Activation syndrome (MCAS). Laboratory testing was normal, with the exception of an upright norepinephrine level of 1121pg/ml. She was diagnosed with chronic urticaria by an allergy and immunology specialist.
Table 1. Active standing test
| Time and position |
Blood Pressure |
Heart Rate |
Symptoms |
| Supine after 10min |
122/70 |
91 |
None |
| Standing 1 min |
124/86 |
103 |
None |
| Standing 2 min |
130/90 |
120 |
Dizziness, palpitations |
| Standing 3 min |
130/90 |
127 |
Palpitations, near syncope |
The patient was started on H1 and H2 blockers, Montelukast, Aspirin 81 mg daily, Cromolyn, and low dose clonidine, and began evaluating her diet for “triggers”. She was weaned off of ivabradine after being on this regimen for a few months. After 6 months, she returned to work and was able to go hiking in the mountains of Colorado. She continues to wear compression garments, aggressively hydrate and supplement with sodium tablets. Due to the elevated upright norepinephrine level, the patient was diagnosed as hyperadrenergic POTS. It was felt that, the chronic urticaria and subsequent histamine release exacerbated her syndrome. She continues to have postural tachycardia, albeit with less extreme elevations of heart rate, improved symptoms and quality of life.
Postural Orthostatic Tachycardia Syndrome and Inappropriate Sinus Tachycardia
Postural orthostatic tachycardia syndrome (POTS) and inappropriate sinus tachycardia (IST) are common forms of cardiac dysautonomia that may overlap in clinical presentation. Both predominantly affect young women and can significantly affect quality of life and functional status13,14. Distinguishing between the two in the clinical practice can be challenging and requires astute attention to detail, with particular focus on the correlation of symptoms with upright posture.
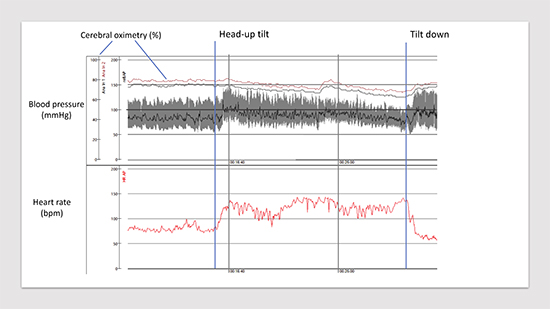
As its name implies, POTS is not a disease entity, but a syndrome. The syndrome consists of orthostatic intolerance of at least 6 months duration, which is defined by an increase in heart rate of 30 bpm (>40bpm if <19yo) within 10 minutes of active standing or passive tilt testing, and without a significant drop of systolic blood pressure (< 20mmHg) 15,16Figure 5. Upright heart rates generally exceed 120 bpm, especially in younger individuals. Symptoms frequently associated with upright posture can include dizziness, palpitations, fatigue, headache, nausea, pre-syncope, tunneling of vision, and “brain fog” 14,17,18. Although patients may report continued symptoms when sitting or supine, in order to make a diagnosis of POTS, there has to be a clear exacerbation of symptoms and an increase in heart rate when assuming an upright posture. Syncope is less common in patients with POTS, but POTS patients may also have coexistent neurally-mediated i.e. vasovagal syncope19. Those most affected are women between the ages of 15-35, with a prevalence of 0.2%. Recent data from the Mayo Clinic 20 supports this with 89% of diagnosed patients being female with an average age of 23.4-years-old.
Figure 5. Head-up tilt test performed on 15-year-old-girl with suspected POTS. Please, note a steep increase in heart rate after tilt-up (+60 bpm) associated with subjective symptoms of orthostatic intolerance, headache, dizziness and fatigue. Cerebral oximetry (normal values: 60-80%) measured through near-infrared spectroscopy demonstrates pronounced variation and fall tendency. Test is terminated after 15 min due to pronounced intolerance symptoms.
The underlying mechanism of POTS appears to be heterogeneous and may include catecholamine excess due to either abnormal biosynthesis and release or reduced clearance21, peripheral autonomic denervation22, and autoimmunity against adrenergic and other cardiovascular receptors14,23-25. Accordingly, several POTS subtypes have been proposed. These include: hyperadrenergic, neuropathic, and hypovolemic forms.26 Importantly, identification of specific POTS subtypes usually requires additional tests such as catecholamine measurement, skin biopsy, blood volume assessment, and immunological testing, which is not generally available outside very specialized diagnostic centers. Associations with hypermobility syndromes and mast cell disorders are also noted14,15,26. Patients with mast cell disorders will often experience flushing, abdominal discomfort, have symptoms triggered by stress or exertion, or have a history of urticaria, dermatographia, or rash. During an acute episode, these patients will have an elevated urine methylhistamine. It is important to note that this level will be normal between episodes27.
Hyperadrenergic POTS patients will have an elevated standing plasma norepinephrine level of >600 pg/ml and account for approximately 30% of all POTS patients28,29. During head up tilt testing, these patients typically have greater increases in systolic blood pressure and heart rate and clinically report more symptoms of headache, tremulousness and dizziness than other subtypes of POTS 30. Treatment focuses on reducing activation of the sympathetic nervous system. Most commonly beta blockers are utilized, however sympatholytic agents such as clonidine and methyldopa may also be effective treatment strategies31.
Neuropathic POTS patients have signs of peripheral autonomic denervation and impaired sudomotor function with diminished sweating of the distal lower extremities 19,22. These patients have been shown to have lower heart rates, less anxiety and less impact on their quality-of-life than other POTS patients32. Testing for neuropathic POTS is not practical in most clinical settings. These patients may have excessive venous pooling, along with acrocyanosis while assuming upright posture. Use of compression garments to reduce venous pooling in the pelvis and lower extremities, exercise to strengthen the musculature of the lower extremities, and midodrine may prove to be beneficial.31
In hypovolemic POTS, as the name implies, patients are noted to have low plasma volumes.26 Dr. Raj and colleagues noted that, there was no compensatory increase in plasma renin activity despite the lower circulating volume. These patients were also noted to have low levels of aldosterone33. Treatment of these patients should focus on expanding blood/plasma volume and physical reconditioning31.
Cardiovascular deconditioning i.e. reduced exercise tolerance and diffuse muscle fatigue are common denominators among the various POTS subtypes26. Bedrest/physical inactivity will often exacerbate symptoms and can precede the onset of POTS. Physical inactivity results in cardiac atrophy and hypovolemia. Early physical activity in the recumbent/horizontal position is recommended as a treatment strategy, with a gradual increase in duration and intensity as patients become more fit34.
Inappropriate Sinus Tachycardia (IST) can be difficult to distinguish from POTS in the clinical setting, as it affects a similar demographic (primarily young women) and symptoms often overlap. A distinguishing feature is the lack of exacerbation of symptoms with upright posture. IST is defined as a resting heart rate >100 bpm and a mean heart rate of at least 90 bpm over a 24- hour period. 15Figure 6. Sinus tachycardia related to other conditions (anemia, thyroid abnormalities, hypovolemia, medications, etc.) should be excluded. A 12-lead ECG should be obtained during tachycardia and sinus rhythm to exclude other atrial tachycardias. The underlying mechanism is not clearly understood. A small study suggested a high intrinsic heart rate, depressed efferent cardiovagal reflex and betaadrenergic hypersensitivity35. Another small study, by Chiale et al, suggests anti- beta-1 receptor antibodies may play a role36.
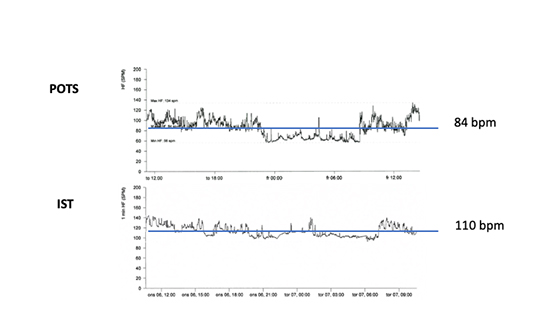
Figure 6. Comparison of two typical 24-hour Holter monitoring circadian curves in postural orthostatic tachycardia syndrome (POTS) and inappropriate sinus tachycardia (IST). Please, note higher average heart frequency and lack of typical high heart rate spikes, especially after awakening in IST compared with POTS.
69-year-old gentleman presented in 2016 after moving to the area. He has a history of severe native coronary artery disease status post 3-vessel bypass in 2013, ischemic cardiomyopathy with an EF of 30% post single chamber primary prevention ICD implanted several months after bypass surgery, Type-I diabetes with peripheral neuropathy, diabetic nephropathy, and dyslipidemia. He reports a history of “chronic hypotension” since his bypass treated with a combination of midodrine and fludrocortisone. He was diagnosed with Parkinson’s disease 12 months ago. He is unable to get out of bed until mid-afternoon due to weakness and dizziness. He denies syncope, but reports falling most days. Current medications: Carbidopa-Levodopa 25/100 1 tablet twice daily, Aspirin 81mg daily, Furosemide 60mg daily, Spironolactone 12.5mg daily, Midodrine 5mg po three times daily, Fludrocortisone 0.2mg in the morning, 0.1mg in the evening, Crestor 40mg daily, Gabapentin 300mg twice daily, Iron ER 325mg twice daily, Novolog 100 unit/ml: 8 units subcutaneously 3 times daily, Metoprolol succinate 12.5mg daily, Tamsulosin ER 0.4mg every other day. The patient was noted by staff to become weak and fall to the ground when being escorted from the waiting room. He was placed in a wheelchair. The first blood pressure obtained in a seated position was 68/42 with a pulse of 92bpm. Orthostatic vital signs were not performed due to extreme hypotension. On physical exam, the patient appeared ill and was pale. Cardiac exam did not reveal jugular venous distention, murmurs, rubs or gallops. Lunges were clear and there was no evidence of peripheral edema. An echocardiogram showed an EF of 33% with inferolateral akinesis and hypokinesis of the remaining walls. There were no valvular abnormalities and the pulmonary pressure was normal.
The patient was taken off of spironolactone, and furosemide was reduced to 40mg daily. Droxidopa was added to the medical regimen and titrated up to the maximum dose of 600mg three times daily. Active standing vital signs 1 week later was performed without symptoms.
|
|
Blood Pressure
|
Pulse
|
|
Lying
|
122/64
|
80
|
|
Sitting
|
124/60
|
80
|
|
3-minute stand:
|
112/48
|
80
|
Although orthostatic vital signs were not obtained on initial evaluation, Droxidopa was added for presumed neurogenic orthostatic hypotension in the setting of diabetic peripheral neuropathy and Parkinson’s disease.
Orthostatic hypotension (OH) is the most common form of cardiovascular dysautonomia in middle-aged and older people12. The prevalence of OH ranges from less than 5% in younger individuals aged <45 years to 20% and more in those aged >70 years 37,38. Moreover, OH can be frequently found in hypertension, heart failure, Parkinson’s disease, diabetes, renal failure, autoimmune diseases and cancer 39-42. In the center of OH pathophysiology are insufficient compensatory mechanisms acting after assuming a standing position, impaired peripheral vasoconstriction, inadequate chronotropic response and reduction in venous return. Fall in BP while standing may be due to structural or functional sympathetic denervation, or impaired baroreflex response due to malfunction of effector organs, the heart and vessels10. The former is usually denominated as neurogenic OH and accompanied by blunted chronotropic response on standing and history of neurodegenerative disease or other chronic conditions affecting ANS such as diabetes or kidney failure.43,44
Traditionally, OH Figure 7 is defined as sustained reduction of systolic BP (SBP) ≥ 20 mmHg and/or diastolic BP (DBP) ≥ 10 mmHg within 3 minutes of active standing test or during a headup tilt test (HUT) 45,46. In patients with supine hypertension, a reduction in systolic BP of at least 30 mmHg might be considered as the magnitude in BP fall is proportional to baseline values 45. The delayed form OH Figure 8 is due to a gradual impairment of adaptive mechanisms during orthostasis, resulting in a slow progressive drop in arterial pressure of the same magnitude as in classical form but occurring first after 3 minutes of standing45. Delayed OH has been associated with a milder impairment of sympathetic function, suggesting that this form may be a less severe or an early form of autonomic failure, along with age-related impairment of compensatory reflexes associated with a stiffer and more preloaddependent heart in older patients 47-50. In terms of syncope incidence, OH is the second most common etiology of syncope, occurring in approximately 10-15% of syncope presentations, and being strongly overrepresented in advanced age 50-52. Unfortunately, OH is often unrecognized or even misdiagnosed. As such pacemaker implants do not eliminate recurrent syncope in all patients with bradycardia even if adequate pacemaker function is present unless OH is recognized and addressed 53. Orthostatic hypotension should be always suspected when syncope or characteristic symptoms occur while standing, in a warm environment, after large meals (overlap with postprandial hypotension), while going to the toilet during night or after awakening and getting up in the morning52,54. The management of OH is multifaceted, beyond the scope of this article, and the interested reader is referred to specific monographies on the subject3,12,55.
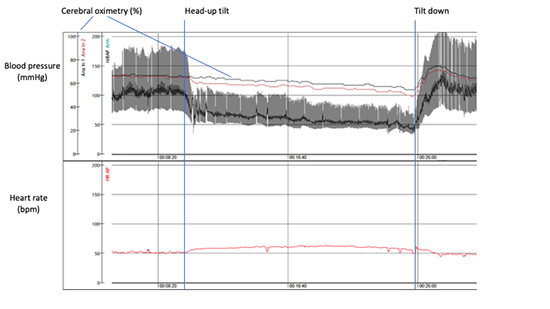
Figure 7. Classical orthostatic hypotension. Head-up tilt test performed on 82-year-old-man. Please, note a steep decrease in blood pressure after tilt-up ( -70 mmHg) associated with subjective symptoms of orthostatic intolerance, dizziness and fatigue. Cerebral oximetry (normal values: 60-80%) measured through near-infrared spectroscopy demonstrates progressive fall. Test is terminated after 15 min due to pronounced intolerance symptoms and presyncope.
Reflex syncope: vasovagal reflex and carotid sinus syndrome
Reflex syncope is the result of a paroxysmal parasympathetic activation and sympathetic withdrawal resulting in hypotension and bradycardia. The two most common forms of reflex syncope are vasovagal syncope and carotid sinus syndrome52. Vasovagal syncope (VVS) is the most frequent form of syncope among all age groups. Female patients experience more episodes of syncope over longer periods of their life than men56. The diagnosis of vasovagal syncope is often implied by the history. Vasovagal syncope often has: 1) a trigger (emotional distress, pain, fear, orthostatic stress) followed by 2) a prodrome of non-cardiovascular symptoms resulting from activation of the vagus nerve (nausea, sweating, diaphoresis, pallor, yawning, salivation) and 3) symptoms associated with cerebral hypoperfusion (dizziness/light-headedness, blurred vision, scotomata) before total loss of consciousness.57 In term of hemodynamic response, VVS may be cardioinhibitory, mixed or vasodilitatory. A mixed mechanism is most prevalent52.
Jardine and colleagues58 use tilt table data to describe the physiologic phases that occur prior to the reflex syncope response. The first phase is the “early stabilization” which occurs when assuming upright posture. There is a shift in central blood volume from the thorax to the lower extremities and the heart rate increases in an attempt to compensate for a fall in cardiac output. There is a simultaneous rise in diastolic blood pressure due to a baroreflexmediated constriction of the splanchnic arterioles and skeletal muscle (increased muscle sympathetic nerve activity MSNA.) The second phase is a period of “circulatory instability” characterized by decreased systolic blood pressure and cardiac output. Bursts of MSNA are noted to increase blood pressure variability. The third “terminal hypotension” phase occurs 30-60 seconds prior to syncope. In adults, the mean arterial pressure, blood pressure variability, and heart rate decrease despite maintaining SVR. In younger patients, the fall in cardiac output can be seen with concomitant vasodilation. The fourth phase is the “recovery” in which there is a rapid recovery of MAP with symptomatic improvement when returned to a supine position. Fu and Levine demonstrate similar findings and noted that MSNA withdrawal occurred late, after the onset of hypotension59. The duration of the phases of vasovagal syncope varies among individuals, as does the mechanism resulting in decreased cardiac output.
Reflex syncope is generally benign and does not lead to significant morbidity and mortality. Why some individuals are predisposed to vasovagal syncope remains unclear. Dr. Sheldon’s work suggests the possibility of a genetic predisposition related to serotonin signaling. More conclusive work is needed in this area60,61.
Carotid sinus hypersensitivity (CSH, afferent baroreceptor dysfunction) is another common form of reflex syncope, predominantly found in older individuals52. By nature, CSH is a chronic condition, which when associated with episodic syncopal attacks is termed “carotid sinus syndrome” with a substantial proportion of patients demonstrating cardioinhibitory response (ventricular pause>3 sec) and being suitable for pacemaker therapy.62 CSH may overlap with other chronic dysautonomic conditions such as OH, jointly contributing to circulatory collapse and syncope Figure 8.
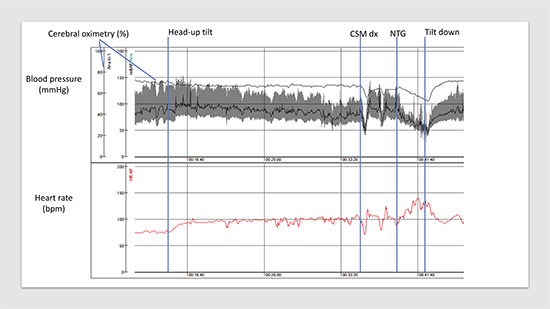
Figure 8. Delayed orthostatic hypotension, carotid sinus hypersensitivity and vasovagal reflex. Head-up tilt test performed on 65-yearold- man with unexplained syncope. Please, note a progressive decrease in blood pressure after tilt-up (- 30 mmHg ), positive carotid sinus massage right side with symptom reproduction (dizziness) and finally positive nitroglycerine provocation with syncope. Cerebral oximetry (normal values: 60-80%) measured through near-infrared spectroscopy demonstrates only a slight progressive fall. CSM dx, carotid sinus massage right side; NTG, nitroglycerine 400 μg sublingually.
Other forms of cardiac dysautonomia
Dysautonomias are not only limited to very rare or less known syndromes and conditions such as POTS and IST. Indeed, all disorders of the autonomic control of global and local circulation may fit under the umbrella term of cardiovascular dysautonomia1,3,10. It should be remembered that, one of the most common, if not commonest, cardiovascular disorders, arterial hypertension, is a form of cardiovascular autonomic dysfunction of hyperadrenergic type with upregulation of neuroendocrine mechanisms responsible for blood pressure control 63Figure 1. Moreover, disturbances of natural circadian blood pressure variability such as a non-dipping pattern and nighttime hypertension are also dysautonomias and suggest impaired autonomic control of global circulation64. These conditions can be found in hypertension, orthostatic hypotension, diabetes, and kidney failure. Likewise, the reduced heart rate variability and chronotropic insufficiency are two opposite sides of the same problem, either a result of sympathetic upregulation and constant hyperadrenergic drive (“too fast”)63, or an impaired heart rate response to autonomic stimuli (“too slow”), falling within the broad category of autonomic “pump dysfunction” Figure 1. One should not forget that disorders of vascular bed control in specific organs and body zones are also an integral part of dysautonomic continuum. To that, category belong such disorders as migraine, coronary spasm, acrocyanosis and venous pooling Figure 1.
Diagnostic strategies for cardiovascular dysautonomias
There are multiple diagnostic modalities for cardiovascular dysautonomias, some of which are well-known from the standard diagnostic procedures in cardiology for detection of coronary disease, cardiac arrhythmias, and impairment of cardiac pump function. This is a summary of recommendations for different types of cardiac dysautonomias according to current syncope guidelines 46,52,54.
|
Modality
|
POTS
|
IST
|
OH
|
VVS
|
CSS
|
HTN
|
Chronotropic
insufficiency
|
|
HUT
|
X
|
|
X
|
X
|
X
|
|
|
|
CSM
|
|
|
|
|
X
|
|
|
|
24-h-ECG
|
X
|
X
|
|
|
|
|
X
|
|
24-h-ABPM
|
X
|
|
X
|
(X)
|
|
X
|
|
|
ILR
|
|
|
|
X
|
X
|
|
X
|
HUT, head-up tilt testing; CSM, carotid sinus massage; 24-h-ABPM, 24-hour-ambulatory blood pressure monitoring; ILR, implantable loop recorder; POTS, Postural Orthostatic Tachycardia Syndrome; IST, Inappropriate Sinus Tachycardia; OH, Orthostatic Hypotension; VVS, Vasovagal Syncope; CSS, Carotid Sinus Syndrome; HTN, hypertension.
Apart from the most prevalent arterial hypertension, conditions like postural orthostatic tachycardia syndrome, inappropriate sinus tachycardia, orthostatic hypotension, reflex syncope, chronotropic incompetence and cardiovascular circadian rhythm disorders should be known for cardiologists. Cardiac dysautonomias vary in prevalence during the life span and may overlap, thus posing a challenge for a clinician. It is therefore important to recognize their typical manifestations, critical diagnostic methods as well as prognostic and therapeutic consequences.
-
Novak P. Autonomic Disorders. The American journal of medicine. 2019;132(4):420-36.
- Gordan R, Gwathmey JK, Xie LH. Autonomic and endocrine control of cardiovascular function. World J Cardiol. 2015;7(4):204-14.
- Kaufmann H, Norcliffe-Kaufmann L, Palma JA. Baroreflex Dysfunction. N Engl J Med. 2020;382(2):163-78.
- Cooper VL, Hainsworth R. Carotid baroreceptor reflexes in humans during orthostatic stress. Exp Physiol. 2001;86(5):677-81.
- Novak V, Novak P, Spies JM, Low PA. Autoregulation of cerebral blood flow in orthostatic hypotension. Stroke; a journal of cerebral circulation. 1998;29(1):104-11.
- Fedorowski A, Melander O. Syndromes of orthostatic intolerance: a hidden danger. Journal of internal medicine. 2013;273(4):322-35.
- Spallone V, Ziegler D, Freeman R, Bernardi L, Frontoni S, Pop-Busui R, et al. Cardiovascular autonomic neuropathy in diabetes: clinical impact, assessment, diagnosis, and management. Diabetes Metab Res Rev. 2011;27(7):639-53.
- Vinik AI, Ziegler D. Diabetic cardiovascular autonomic neuropathy. Circulation. 2007;115(3):387-97.
- Florea VG, Cohn JN. The autonomic nervous system and heart failure. Circ Res. 2014;114(11):1815-26.
- Goldstein DS, Robertson D, Esler M, Straus SE, Eisenhofer G. Dysautonomias: clinical disorders of the autonomic nervous system. Ann Intern Med. 2002;137(9):753-63.
- Ricci F, Fedorowski A, Radico F, Romanello M, Tatasciore A, Di Nicola M, et al. Cardiovascular morbidity and mortality related to orthostatic hypotension: a meta-analysis of prospective observational studies. Eur Heart J. 2015;36(25):1609-17.
- Ricci F, De Caterina R, Fedorowski A. Orthostatic Hypotension: Epidemiology, Prognosis, and Treatment. J Am Coll Cardiol. 2015;66(7):848-60.
- Olshansky B, Sullivan RM. Inappropriate sinus tachycardia. J Am Coll Cardiol. 2013;61(8):793-801.
- Fedorowski A. Postural orthostatic tachycardia syndrome: clinical presentation, aetiology and management. Journal of internal medicine. 2019;285(4):352-66.
- Sheldon RS, Grubb BP, 2nd, Olshansky B, Shen WK, Calkins H, Brignole M, et al. 2015 heart rhythm society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm. 2015;12(6):e41-63.
- Olshansky B, Cannom D, Fedorowski A, Stewart J, Gibbons C, Sutton R, et al. Postural Orthostatic Tachycardia Syndrome (POTS): A critical assessment. Progress in cardiovascular diseases. 2020.
- Zadourian A, Doherty TA, Swiatkiewicz I, Taub PR. Postural Orthostatic Tachycardia Syndrome: Prevalence, Pathophysiology, and Management. Drugs. 2018;78(10):983-94.
- Goodman BP. Evaluation of postural tachycardia syndrome (POTS). Auton Neurosci. 2018.
- Bryarly M, Phillips LT, Fu Q, Vernino S, Levine BD. Postural Orthostatic Tachycardia Syndrome: JACC Focus Seminar. J Am Coll Cardiol. 2019;73(10):1207-28.
- AbdelRazek M, Low P, Rocca W, Singer W. Epidemiology of Postural Tachycardia Syndrome. Neurology. 2019;92:S18.005.
- Shannon JR, Flattem NL, Jordan J, Jacob G, Black BK, Biaggioni I, et al. Orthostatic intolerance and tachycardia associated with norepinephrine-transporter deficiency. N Engl J Med. 2000;342(8):541-9.
- Benarroch EE. Postural tachycardia syndrome: a heterogeneous and multifactorial disorder. Mayo Clin Proc. 2012;87(12):1214-25.
- Fedorowski A, Li H, Yu X, Koelsch KA, Harris VM, Liles C, et al. Antiadrenergic autoimmunity in postural tachycardia syndrome. Europace. 2017;19(7):1211-9.
- Gunning WT, 3rd, Kvale H, Kramer PM, Karabin BL, Grubb BP. Postural Orthostatic Tachycardia Syndrome Is Associated With Elevated G-Protein Coupled Receptor Autoantibodies. Journal of the American Heart Association. 2019;8(18):e013602.
- Yu X, Li H, Murphy TA, Nuss Z, Liles J, Liles C, et al. Angiotensin II Type 1 Receptor Autoantibodies in Postural Tachycardia Syndrome. Journal of the American Heart Association. 2018;7(8).
- Raj SR, Guzman JC, Harvey P, Richer L, Schondorf R, Seifer C, et al. Canadian Cardiovascular Society Position Statement on Postural Orthostatic Tachycardia Syndrome (POTS) and Related Disorders of Chronic Orthostatic Intolerance. The Canadian journal of cardiology. 2020;36(3):357-72.
- Shibao C, Arzubiaga C, Roberts LJ, 2nd, Raj S, Black B, Harris P, et al. Hyperadrenergic postural tachycardia syndrome in mast cell activation disorders. Hypertension. 2005;45(3):385-90.
- Thieben MJ, Sandroni P, Sletten DM, Benrud-Larson LM, Fealey RD, Vernino S, et al. Postural orthostatic tachycardia syndrome: the Mayo clinic experience. Mayo Clin Proc. 2007;82(3):308-13.
- Low PA, Sandroni P, Joyner M, Shen WK. Postural tachycardia syndrome (POTS). J Cardiovasc Electrophysiol. 2009;20(3):352-8.
- Zhang Q, Chen X, Li J, Du J. Clinical features of hyperadrenergic postural tachycardia syndrome in children. Pediatr Int. 2014;56(6):813-6.
- Mar PL, Raj SR. Postural Orthostatic Tachycardia Syndrome: Mechanisms and New Therapies. Annual review of medicine. 2020;71:235-48.
- Gibbons CH, Bonyhay I, Benson A, Wang N, Freeman R. Structural and functional small fiber abnormalities in the neuropathic postural tachycardia syndrome. PLoS One. 2013;8(12):e84716.
- Raj SR, Biaggioni I, Yamhure PC, Black BK, Paranjape SY, Byrne DW, et al. Renin-aldosterone paradox and perturbed blood volume regulation underlying postural tachycardia syndrome. Circulation. 2005;111(13):1574-82.
- Fu Q, Levine BD. Exercise in the postural orthostatic tachycardia syndrome. Auton Neurosci. 2015;188:86-9.
- Morillo CA, Klein GJ, Thakur RK, Li H, Zardini M, Yee R. Mechanism of 'inappropriate' sinus tachycardia. Role of sympathovagal balance. Circulation. 1994;90(2):873-7.
- Chiale PA, Garro HA, Schmidberg J, Sanchez RA, Acunzo RS, Lago M, et al. Inappropriate sinus tachycardia may be related to an immunologic disorder involving cardiac beta andrenergic receptors. Heart Rhythm. 2006;3(10):1182-6.
- Rose KM, Tyroler HA, Nardo CJ, Arnett DK, Light KC, Rosamond W, et al. Orthostatic hypotension and the incidence of coronary heart disease: the Atherosclerosis Risk in Communities study. Am J Hypertens. 2000;13(6 Pt 1):571-8.
- Rutan GH, Hermanson B, Bild DE, Kittner SJ, LaBaw F, Tell GS. Orthostatic hypotension in older adults. The Cardiovascular Health Study. CHS Collaborative Research Group. Hypertension. 1992;19(6 Pt 1):508-19.
- Low PA. Prevalence of orthostatic hypotension. Clin Auton Res. 2008;18 Suppl 1:8-13.
- Benvenuto LJ, Krakoff LR. Morbidity and mortality of orthostatic hypotension: implications for management of cardiovascular disease. Am J Hypertens. 2011;24(2):135-44.
- Goodman BP, Crepeau A, Dhawan PS, Khoury JA, Harris LA. Spectrum of Autonomic Nervous System Impairment in Sjogren Syndrome. Neurologist. 2017;22(4):127-30.
- Li J, Li Y, Xing S, Zhang J, Qiu B, Zeng J, et al. Orthostatic Hypotension and Albuminocytologic Dissociation as Primary Manifestations of the Paraneoplastic Syndrome. Eur Neurol. 2018;80(1-2):78-81.
- Goldstein DS, Sharabi Y. Neurogenic orthostatic hypotension: a pathophysiological approach. Circulation. 2009;119(1):139-46.
- Olshansky B, Muldowney J. Cardiovascular Safety Considerations in the Treatment of Neurogenic Orthostatic Hypotension. Am J Cardiol. 2020.
- . Freeman R, Wieling W, Axelrod FB, Benditt DG, Benarroch E, Biaggioni I, et al. Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Clin Auton Res. 2011;21(2):69-72.
- Brignole M, Moya A, de Lange FJ, Deharo JC, Elliott PM, Fanciulli A, et al. Practical Instructions for the 2018 ESC Guidelines for the diagnosis and management of syncope. Eur Heart J. 2018;39(21):e43-e80.
- Verheyden B, Gisolf J, Beckers F, Karemaker JM, Wesseling KH, Aubert AE, et al. Impact of age on the vasovagal response provoked by sublingual nitroglycerine in routine tilt testing. Clin Sci (Lond). 2007;113(7):329-37.
- Gibbons CH, Freeman R. Clinical implications of delayed orthostatic hypotension: A 10-year follow-up study. Neurology. 2015;85(16):1362-7.
- Fedorowski A, van Wijnen VK, Wieling W. Delayed orthostatic hypotension and vasovagal syncope: a diagnostic dilemma. Clin Auton Res. 2017;27(4):289-91.
- Torabi P, Ricci F, Hamrefors V, Sutton R, Fedorowski A. Classical and Delayed Orthostatic Hypotension in Patients With Unexplained Syncope and Severe Orthostatic Intolerance. Front Cardiovasc Med. 2020;7:21.
- Sutton R. Clinical classification of syncope. Prog Cardiovasc Dis. 2013;55(4):339-44.
- Brignole M, Moya A, de Lange FJ, Deharo JC, Elliott PM, Fanciulli A, et al. 2018 ESC Guidelines for the diagnosis and management of syncope. Eur Heart J. 2018;39(21):1883-948.
- Yasa E, Ricci F, Holm H, Persson T, Melander O, Sutton R, et al. Cardiovascular Autonomic Dysfunction Is the Most Common Cause of Syncope in Paced Patients. Front Cardiovasc Med. 2019;6(154):154.
- Shen WK, Sheldon RS, Benditt DG, Cohen MI, Forman DE, Goldberger ZD, et al. 2017 ACC/AHA/HRS Guideline for the Evaluation and Management of Patients With Syncope: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2017;70(5):620-63.
- Eschlbock S, Wenning G, Fanciulli A. Evidence-based treatment of neurogenic orthostatic hypotension and related symptoms. J Neural Transm (Vienna). 2017;124(12):1567-605.
- Kenny RA, Bhangu J, King-Kallimanis BL. Epidemiology of syncope/collapse in younger and older Western patient populations. Progress in cardiovascular diseases. 2013;55(4):357-63.
- Alboni P. The different clinical presentations of vasovagal syncope. Heart. 2015;101(9):674-8.
- Jardine DL, Wieling W, Brignole M, Lenders JWM, Sutton R, Stewart J. The pathophysiology of the vasovagal response. Heart Rhythm. 2018;15(6):921-9.
- Fu Q, Verheyden B, Wieling W, Levine BD. Cardiac output and sympathetic vasoconstrictor responses during upright tilt to presyncope in healthy humans. J Physiol. 2012;590(8):1839-48.
- Sheldon R, Rose MS, Ritchie D, Martens K, Maxey C, Jagers J, et al. Genetic Association Study in Multigenerational Kindreds With Vasovagal Syncope. Circ Arrhythm Electrophysiol. 2019;12(1):e006884.
- Sheldon RS, Sandhu RK. The Search for the Genes of Vasovagal Syncope. Front Cardiovasc Med. 2019;6:175.
- Brignole M, Ammirati F, Arabia F, Quartieri F, Tomaino M, Ungar A, et al. Assessment of a standardized algorithm for cardiac pacing in older patients affected by severe unpredictable reflex syncopes. Eur Heart J. 2015;36(24):1529-35.
- Carthy ER. Autonomic dysfunction in essential hypertension: A systematic review. Ann Med Surg (Lond). 2014;3(1):2-7.
- Mancia G. Short- and long-term blood pressure variability: present and future. Hypertension. 2012;60(2):512-7.