Catheter Ablation for Hospitalized Atrial Fibrillation Patients with Reduced Systolic Function: Analysis of Inpatient Mortality, Resource Utilization and Complications
Muhammad Bilal Munir1, Muhammad Zia Khan2, Pratik Agrawal2, Zain Ul Abideen Asad3, Moinuddin Syed2, Kinjan Patel2, BilYasir Abdul Ghaffarlal2, Muhammad U. Khan2, Safi U. Khan2, Sudarshan Balla2, Jonathan C. Hsu1
1Section of Electrophysiology, Division of Cardiology, University of California San Diego, La Jolla, CA, USA.2Division of Cardiovascular Medicine, West Virginia University Heart and Vascular Institute, Morgantown, WV, USA.3Division of Cardiology, University of Oklahoma School of Medicine, Oklahoma City, OK, USA.
Randomized trials have shown improvement in hard clinical end points when catheter ablation (CA) is employed as a management strategy for certain atrial fibrillation (AF) patients with heart failure and reduced ejection fraction (HFrEF). Limited data, however, exist in this realm outside the controlled clinical trial settings. We sought to determine real-world data on mortality and complications after utilization of CA in such patients.
Data were derived from National Inpatient Sample from January 2008 to August 2015. Patients were identified using the International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM) codes. Baseline characteristics and outcomes were compared among HFrEF and AF patients undergoing CA or not. Propensity matching was done to mitigate selection bias and balance confounding variables. Various CA related complications were assessed. Logistic regression was done to determine predictors of mortality in our study cohort. A total of 2,569,919 patients were analyzed and a total of 7773 patients underwent CA. Mortality was significantly better in CA group in both unmatched (1.2% vs. 4.9%, p < 0.01) and propensity matched cohorts (1.2% vs. 3.6%, p < 0.01). Overall complication rate was 10.2% in CA cohort and primarily driven by cardiac and neurological etiologies. In regression analysis, CA remained a strong predictor of reduced mortality (OR 0.301, 95% CI 0.184-0.494).
CA is associated with improved mortality in admitted AF patients with concomitant HFrEF. Overall complication rate after CA was modest at 10.2%. Consideration can be given to the utilization of this therapeutic modality in hospitalized AF patients with concomitant HFrEF.
Key Words : Atrial fibrillation, Heart failure with reduced ejection fraction, Catheter ablation, Outcomes, National sample.
Muhammad Bilal Munir, MD
Cardiac Electrophysiology Section, Division of Cardiology
University of California San Diego School of Medicine
9452 Medical Center Drive MC 7411
La Jolla, CA 932037
Atrial fibrillation (AF) is the most common sustained arrhythmia encountered in clinical practice 1. AF and congestive heart failure (CHF) frequently co-exist due to similar predisposing risk factors and ability of one to perpetuate the other 2,3. AF is associated with increased CHF hospitalizations, stroke and all-cause mortality 4,5,6. Various randomized trials conducted in last two decades have shown efficacy of catheter ablation (CA) for AF in HF and reduced ejection fraction (HFrEF) patients with respect to hard clinical end-points of mortality and CHF hospitalizations 7,8. With the result of these trials, it is expected that the volume of CA would continue to grow for management of AF and HFrEF patients. It is therefore imperative that data be sought from real world settings with respect to mortality and complications associated with CA in AF and HFrEF patients. Till to date, studies utilizing national databases for assessing aforementioned outcomes did not discriminate based on HF status of the patient 9,10,11,12. We, therefore, utilized National Inpatient Sample (NIS) to assess contemporary trends in mortality and complications associated with CA in AF and HFrEF patients.
We conducted analysis on National Inpatient Sample (NIS) from January 2008 to August 2015. NIS is part of Healthcare Resource and Utilization Project (HCUP) and made possible by a Federal-State-Industry partnership sponsored by the Agency for Healthcare Research and Quality (AHRQ). The NIS is derived from all States and utilized for computing national estimates of healthcare utilization, cost and outcome 13. NIS is compiled annually and the data can be used for analysis of disease trends over time. Institutional Review Board approval and informed consents were not required for this study given the de-identified nature of the NIS dataset and public availability.
We analyzed NIS data from January 2008 to August 2015 using the International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM) codes. Patient under 18 years of age were excluded. Inclusion criteria included patients with HFrEF and AF. Cases with concurrent diagnostic codes for atrial flutter, supraventricular tachycardia, ventricular tachycardia, other premature beats, cardiac dysrhythmia, Wolf-Parkinson-White syndrome, atrioventricular nodal tachycardia and open surgical ablation were excluded. Age was divided into three groups, <65, 65-74 and ≥75. CHAD2VASC2sub> score was calculated. Complications associated with ablation were subsequently assessed. Please see [Figure 1] for flow sheet of patient selection.
Figure 1. Flow sheet of patient selection
Baseline characteristics of patients undergoing ablation versus not along with hospital outcomes were derived. Length of stay and mean cost of stay (inflation adjusted) were subsequently calculated.
For missing values imputation, multiple iterations of Markov Chain Monte Carlo (MCMC) method were used. To account for potential confounding factors and selection bias, a propensity score-matching model was developed using logistic regression to derive two matched groups for comparative outcomes analysis. Given larger non-ablation group and to minimize case loss, a nearest neighbor 1:2 variable ratio, parallel, balanced propensity-matching model was made using a caliper width of 0.2. Descriptive statistics were presented as frequencies with percentages for categorical variables and as means with standard deviations for continuous variables. Baseline characteristics were compared using a Pearson𝜒2 test and Fisher's exact test for categorical variables and independent samples t-test for continuous variables.
Logistic regression was performed to estimate odds ratios (ORs) with 95% confidence intervals (CIs) to determine predictors for mortality in our cohort. Initially, binomial logistic regression model was used to identify variables from demographic data Table 1] that were significantly associated with patient mortality (P value ˂ 0.10). These variables were then subsequently utilized in a multiple logistic regression model to identify predictors of mortality. A type I error rate of <0.05 was considered statistically significant. All statistical analyses were performed using statistical package for social science (SPSS) version 26 (IBM Corp) and R 3.5 for propensity matching. All analyses were done on a weighted sample.
Table 1. Baseline characteristics of the study population stratified on the basis of AF ablation versus not
| Variable no. (%) |
No Ablation (n=2562145) |
Ablation (n=7773) |
All patients with Afib† and HFrEF‡ (n=2569919) |
P value |
| Age (mean [SD]) years |
76.2(11.6) |
69.6(12) |
76.2(11.6) |
<0.01 |
| Age <65 |
415112(16.5%) |
2504(33.1%) |
417616(16.50%) |
<0.01 |
| 65-74 |
521723(20.7%) |
2088(27.6%) |
523811(20.70%) |
<0.01 |
| ≥75 |
1584144(62.8%) |
2980(39.4%) |
1587124(62.80%) |
<0.01 |
| Female |
1068414(41.70%) |
2635(33.90%) |
1069086(41.60%) |
<0.01 |
| CHAD2VASC2sub> score (Median, IQR) |
4(2) |
3(2) |
4(2) |
|
| Race |
|
|
|
|
| Caucasian |
1896153(80.2%) |
5801(83.7%) |
1901954(80.20%) |
<0.01 |
| African American |
254526(10.8%) |
597(8.6%) |
255123(10.80%) |
|
| Hispanics |
120305(5.1%) |
319(4.6%) |
120624(5.10%) |
|
| Asian or Pacific Islander |
33809(1.4%) |
45(0.6%) |
33854(1.40%) |
|
| Native American |
10747(0.5%) |
31(0.4%) |
10778(0.50%) |
|
| AHRQ§ Medical comorbidity |
|
|
|
|
| Alcohol abuse |
72809(2.8%) |
211(2.7%) |
73020(2.80%) |
0.49 |
| Anemia |
34472(1.3%) |
54(0.7%) |
34526(1.30%) |
<0.01 |
| Chronic pulmonary disease |
858847(33.5%) |
2098(27.0%) |
860945(33.50%) |
<0.01 |
| Coagulopathy |
208605(8.1%) |
492(6.3%) |
209097(8.10%) |
<0.01 |
| Diabetes |
218960(8.5%) |
378(4.9%) |
219338(8.50%) |
<0.01 |
| Hypertension |
1745998(68.1%) |
5075(65.3%) |
1751073(68.10%) |
<0.01 |
| Fluid and electrolyte disorders |
895405(34.9%) |
1904(24.5%) |
897309(34.9%) |
<0.01 |
| Liver disease |
69092(2.7%) |
133(1.7%) |
69225(2.70%) |
<0.01 |
| Neurological disorders |
205974(8.0%) |
324(4.2%) |
206298(8.0%) |
<0.01 |
| Peripheral vascular disorders |
354809(13.8%) |
843(10.8%) |
355652(13.8%) |
<0.01 |
| Renal failure |
996522(38.9%) |
2458(31.6%) |
998980(38.9%) |
<0.01 |
| History of stroke |
226751(8.9%) |
498(6.4%) |
227249(8.80%) |
<0.01 |
| Valvular Disease |
273245(10.7%) |
131(1.7%) |
273376(10.60%) |
<0.01 |
| Hospital Location |
|
|
|
|
| Rural |
304736(11.9%) |
303(3.9%) |
305039(11.90%) |
<0.01 |
| Urban Non-teaching |
943247(36.8%) |
1746(22.5%) |
944993(36.80%) |
|
| Urban Teaching |
1314162(51.3%) |
5725(73.6%) |
1319887(51.40%) |
|
| Bed size of the hospital |
|
|
|
|
| small |
345854(13.5%) |
611(7.9%) |
346465(13.5%) |
<0.01 |
| medium |
667961(26.1%) |
1506(19.4%) |
669467(26.1%) |
|
| large |
1548331(60.4%) |
5656(72.8%) |
1553987(60.5%) |
|
| Region |
|
|
|
|
| Northeast |
568435(22.2%) |
1722(22.2%) |
570157(22.2%) |
<0.01 |
| Midwest |
666681(26.0%) |
1913(24.6%) |
668594(26.0%) |
|
| South |
930343(36.3%) |
3024(38.9%) |
933367(36.3%) |
|
| West |
396686(15.5%) |
1115(14.3%) |
397801(15.5%) |
|
| Median household income percentile |
|
|
|
|
| 0–25th |
722541(28.7%) |
2038(26.5%) |
724579(28.7%) |
<0.01 |
| 26–50th |
677852(27.0%) |
2183(28.4%) |
680035(27.0%) |
|
| 51–75th |
615639(24.5%) |
1897(24.7%) |
617536(24.5%) |
|
| 76–100th |
498023(19.8%) |
1573(20.5%) |
499596(19.8%) |
|
† Atrial Fibrillation; ‡Heart Failure with reduced Ejection Fraction; §Agency for healthcare research and quality
A total of 2,569,919 patients with AF and HFrEF were identified from NIS dataset. Out of these, about 7,773 patients underwent AF ablation. Baseline characteristics of the study population are shown in Table 1]. Patients undergoing ablation tend to be younger when compared to patients not undergoing ablation (69.6 vs. 76.2 years, p < 0.01). 41.6% of the study cohort constituted female patients and ablation was performed in 34% of them. Median CHAD2VASC2sub> score was 4(2) for the non-ablation group and 3(2) for the ablation group.
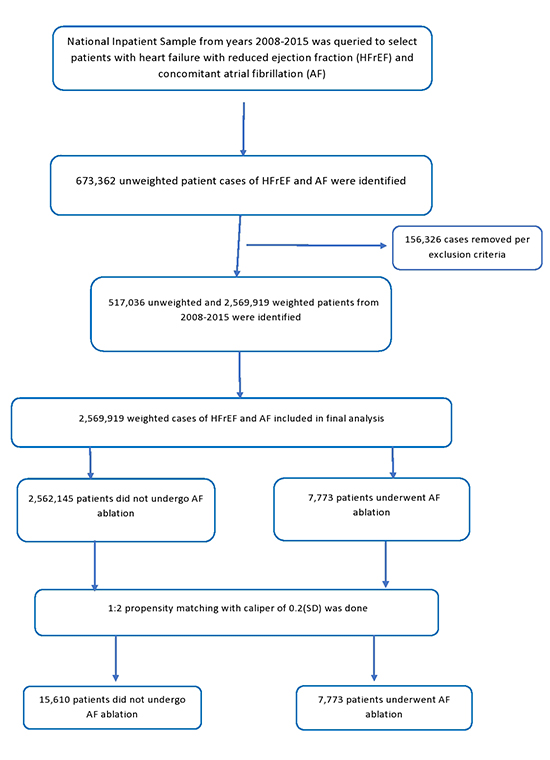
Table 2] illustrates outcomes and resource utilization of our study cohort based on raw unmatched data. A total of 124,765 (4.9%) patients in our study died at discharge. Mortality was significantly lower in the ablation group compared to no ablation group in both unmatched (1.2% vs. 4.9%, p < 0.01) and propensity matched groups (1.2% vs. 3.6%, p < 0.01). Please see Table 3] for detailed outcomes after propensity matching. Mortality trend remained low and stable over study years in both ablation and no ablation group [Figure 2]. There had been a steady increased trend in mean cost for hospital stay over study years in both groups [Figure 3].
Figure 2. Trends in mortality in heart failure with reduced ejection fraction and atrial fibrillation patients vs. not over the study years
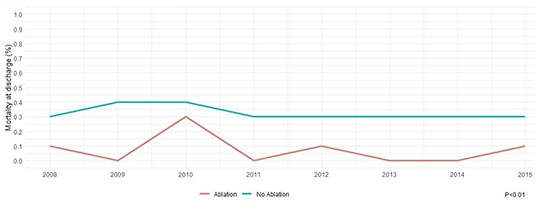
Figure 3. Trends in mean cost of stay in heart failure with reduced ejection fraction and atrial fibrillation patients undergoing ablation vs. not over the study years
Table 2. Hospital encounter outcomes and resource utilization of the study cohort
| Variables no. (%) |
No Ablation (n=2562145) |
Ablation (n=7773) |
All patients with Afib† and HFrEF‡ (n=2569919) |
P value |
| Died at discharge |
124674(4.9%) |
91(1.2%) |
124765(4.9%) |
<0.01 |
| <65 |
11280(2.7%) |
10(0.4%) |
11290(2.7%) |
<0.01 |
| 65-74 |
21181(4.1%) |
31(1.5%) |
21212(4.1%) |
<0.01 |
| >/=75 |
90688(5.7%) |
49(1.6%) |
90737(5.7%) |
<0.01 |
| Discharge Disposition of surviving patients |
|
|
|
|
| Routine/self-care |
1026629(42.1%) |
5420(70.5%) |
1032049(42.2%) |
<0.01 |
| Short-term hospital |
69374(2.8%) |
54(0.7%) |
69428(2.8%) |
|
| Another type of facility |
735372(30.2%) |
980(12.8%) |
736352(30.1%) |
|
| Home Health Care |
589475(24.2%) |
1219(15.9%) |
1219(15.9%) |
|
| Resource utilization, Mean (SD) |
|
|
|
|
| Length of stay, mean (SD), days |
6.1(5.7) |
6.2(6.4) |
6.1(5.8) |
<0.01 |
| Cost of hospitalization-mean (SD), $ |
46370(69554) |
92327(90984) |
46516(69680) |
<0.01 |
† Atrial Fibrillation; ‡Heart Failure with reduced Ejection Fraction
Table 3. Outcomes and resource utilization of the study cohort after 1:2 propensity matching
| Variables |
No Ablation (n=15610) |
Ablation (n=7773) |
All patients with Afib† and HFrEF‡ (n=2569919) |
P value |
| Died at discharge |
565(3.6%) |
91(1.2%) |
656(2.8%) |
<0.01 |
| <65 |
139(2.5%) |
10(0.4%) |
149(1.9%) |
<0.01 |
| 65-74 |
84(2.4%) |
31(1.5%) |
115(2.0%) |
<0.01 |
| ≥75 |
338(5.4%) |
49(1.6%) |
387(4.2%) |
<0.01 |
| Discharge Disposition of surviving patients |
|
|
|
|
| Routine/self-care |
8374(55.7%) |
5420(70.5%) |
13794(60.7%) |
<0.01 |
| Short-term hospital |
484(3.2%) |
54(0.7%) |
538(2.4%) |
|
| Another type of facility |
3127(20.8%) |
980(12.8%) |
4107(18.1%) |
|
| Home Health Care |
2998(19.9%) |
1219(15.9%) |
4217(18.6%) |
|
| Resource utilization, Mean (SD) |
|
|
|
|
| Length of stay, mean (SD), days |
5.9(6.6) |
6.4(6.9) |
6.0(6.7) |
<0.01 |
| Cost of hospitalization-mean (SD), $ |
47,900(100,799) |
93,535(92,919) |
63,071(100,572) |
0.04 |
† Atrial Fibrillation ; ‡Heart Failure with reduced Ejection Fraction
Overall, 10.2% patients had at least one complication associated with CA Table 4]. Complications associated with ablation included stroke (1.8%), myocardial infarction (3.6%), need for percutaneous coronary intervention (0.7%), cardiogenic shock (2.3%), cardiac tamponade (0.7%), vascular complications (0.7%), septic shock (0.7). The incidence of pericardiocentesis was 0.7% in our ablation cohort.
Mortality predictors for AF patients with HFrEF are shown in [Figure 4]. Advanced age (OR 1.027, 95% CI 1.026-1.029), chronic pulmonary disease (OR 1.108, 95% CI 1.077-1.139), coagulopathy (OR 1.797, 95% CI 1.729-1.867), fluid and electrolyte disorders (OR 2.242, 95% CI 2.182-2.303), peripheral vascular disease (OR 1.128, 95% CI 1.088-1.17), valvular heart disease (OR 1.157, 95% CI 1.111-1.204) and renal failure (OR 1.302, 95% CI 1.267-1.338) were associated with increased mortality while ablation was independently associated with lower mortality in our cohort (OR 0.301, 95% CI 0.184-0.494).
Figure 4. Predictors of mortality in patients with heart failure and reduced ejection fraction and atrial fibrillation
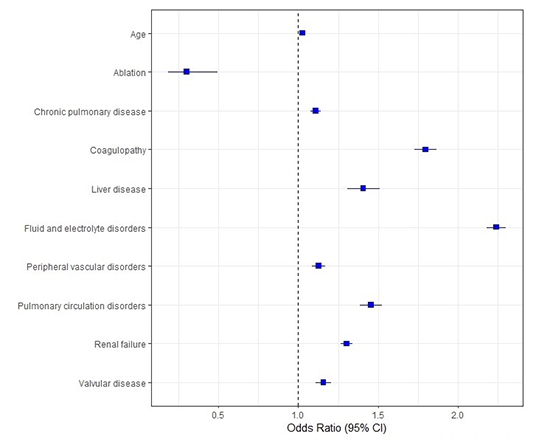
Table 4. Complications in AF ablation patients stratified on the basis of gender
| Variable no. (%) |
All complications (n=7773) |
Men (n=5135) |
Women (n=2637) |
P value |
| At least one complication |
788(10.2%) |
555(10.8%) |
233(8.8%) |
<0.01 |
| Iatrogenic cardiac complications |
121(1.6%) |
90(1.80%) |
31(1.20%) |
0.05 |
| Stroke |
138(1.8%) |
63(1.20%) |
75(2.80%) |
<0.01 |
| Vascular complications |
55(0.7%) |
30(0.6%) |
25(0.9%) |
0.07 |
| Pneumothorax |
21(0.3%) |
11(0.2%) |
10(0.4%) |
0.18 |
| Post-operative respiratory failure |
30(0.4%) |
20(0.4%) |
10(0.4%) |
0.94 |
| Need for pericardiocentesis |
57(0.7%) |
28(0.5%) |
29(1.1%) |
0.02 |
| Cardiac tamponade |
57(0.7%) |
28(0.5%) |
29(1.1%) |
0.07 |
| Cardiogenic shock |
181(2.30%) |
151(2.90% |
30(1.10%) |
<0.01 |
| Septic shock |
54(0.7%) |
44(0.9%) |
10(0.4%) |
0.02 |
| Myocardial infarction |
277(3.6%) |
204(4.0%) |
73(2.8%) |
<0.01 |
| Percutaneous coronary intervention |
53(0.7%) |
44(0.9%) |
9(0.3%) |
<0.01 |
| Pulmonary embolism |
44(0.6%) |
29(0.6%) |
15(0.6%) |
0.98 |
The main findings of current study include: (1) AF patients with HFrEF tended to have low mortality if they undergo ablation in both unmatched (1.2% vs. 4.9%, p < 0.01) and propensity matched cohorts (1.2% vs. 3.6%, p < 0.01). (2) CA was an independent predictor of reduced mortality in adjusted mortality analysis. (3) Approximately 10.2% patients had at least one procedure related complication with cardiac and neurologic complications being the most frequent in our cohort.
AF and CHF frequently co-exist and the prevalence of CHF is reported to be 42% in AF patients 14. AF is associated with frequent hospitalizations and mortality in CHF patients 4,5,6. Several trails have reported improvement in hard clinical end points of mortality and hospitalizations in AF patients with concomitant HfrEF if CA was employed as part of therapeutic modality 7,8. In the AATAC study 7, 203 patients with persistent AF and HFrEF were randomized to get either amiodarone or CA. At the end of follow-up, CA was found to be superior to amiodarone in maintain sinus rhythm and improving left ventricular ejection fraction (LVEF). The study also showed 45% relative risk reduction for unplanned hospitalizations and 56% relative risk reduction for mortality in CA patients when compared to amiodarone group. More recently, CASTLE AF 8 enrolled patients with paroxysmal and persistent AF and concomitant HFrEF to either CA or medical therapy with rate or rhythm control. The primary end-point taken in this trial was a composite of all-cause mortality or CHF hospitalizations. At the end of 37 months follow up, primary end point occurred in few patients who underwent CA compared to medical therapy (HR 0.62, 95% CI 0.43-0.87). In our real world analysis of AF patients with HFrEF, we have demonstrated significant improvement in mortality in patients in whom CA was employed as a therapeutic strategy for management of AF. The significant reduction in mortality was uniform in both matched and unmatched cohort. Additionally, in our adjusted mortality analysis, CA was found to independently predict improved mortality in our cohort (OR 0.301, 95% CI 0.184-0.494). Of note, due to limitation of NIS dataset, CA assessment was only done while patients are admitted to inpatient settings. These patients are speculated to be sicker when compared to their counterparts who get elective CA procedure as an outpatient and were the ones primarily enrolled in aforementioned trials. It is pertinent to point here that even in these sick patients, CA was associated with improved survival at discharge suggesting that due consideration should be given to this therapeutic modality for management of such patients.
In our study, about 10.2% patients sustained procedure related complications after CA ablation. In a study by Tripathi et al 12 on recent contemporary trends of CA in AF patients, the overall complication rate was reported at 5.46%. It is pertinent to point out that Tripathi et al. utilized all AF patients for their analysis and did not discriminate based on HFrEF status. Patients admitted with AF and concomitant HFrEF are particularly on the sickest end of spectrum in their disease process. The high complication rate of 10.2% in our study cohort probably reflected variable degree of institution experience in performing CA in these sickest patients. Our study also showed increased rate of myocardial infarction (3.6%) and cardiogenic shock (2.3%) in study population. Some degree of troponin elevation is frequently encountered post ablation due to localized myocardial necrosis consequent to creation of lesion sets 15, however, about 0.7% patients in our cohort did undergo coronary stenting indicative of type I myocardial infarction. Strong index of suspicion is therefore warranted for timely detection of these key cardiovascular complications as that may result in improved outcomes. In our cohort, about 0.7% patients were found to be septic during the particular hospitalization in which CA was performed. Sepsis typically is a late complication of CA and usually occurs within 30-days of procedure as demonstrated by recent study from Cheng et al. 16 and that may explain relatively low rate of this complication during our patients index hospitalization. The rate of vascular complications was 0.7% and that was similar to reports from earlier studies 12. Stroke happened in approximately 1.8% of our patients when compared to 1% of patients in Tripathi et al. study 12. AF perpetuates thrombus formation due to stasis of blood and it is speculated that HFrEF may accentuate this response by promoting further stagnation of blood. It is therefore advised that close attention should be paid to anti-coagulation regimen and activating clotting times during the CA procedure to minimize the risk of strokes in AF patients with concomitant HFrEF.
Our study has following key limitations: (1) NIS is an administrative claim-based database that uses ICD-9-CM codes for diagnosis that may be subject to error. However, the hard clinical end point of mortality and procedure code for ablation are less prone to error. Additionally, HCUP quality control measures are routinely performed on NIS dataset to ensure continued reliability and validity 13. (2) NIS collects data on inpatient discharges and do not reflect on outpatient related encounters. Currently, most AF ablations are done as an elective outpatient procedure and these patients are relatively less sick and expected to have lower mortality and complication rate when compared to our sample of admitted AF patients. Nonetheless, our study reflects real word data on outcomes in these sickest hospitalized AF patients after CA and largely representative of United States population sample. (3) Several patient related factors such as type and duration of AF, cardiac parameters such as chamber dimensions and ejection fraction and procedure related factors such as type of lesions performed (pulmonary vein isolation alone or in combination with left atrial roof and floor lines etc.) and type of energy used to create lesion sets could not be ascertained from present data set. (4) NIS does not collect longitudinal data on patients so long term follow up could not be assessed. To the same end, certain specific CA complications such as development of an atrio-esophageal fistula occurs weeks to months after the procedure and unfortunately the incidence of this complication could not be studied from NIS.
In this large nationally representative sample of United States population, we demonstrated that CA is associated with reduced mortality in AF patients with HFrEF in both matched and unmatched cohorts. The complication rate was 10.2% and primarily were cardiac and neurological in origin.