Atrial Tachycardias Occurring After Atrial
Fibrillation Ablation: Strategies for Mapping and
Ablation
Stavros Mountantonakis, MD, Edward P. Gerstenfeld, MD
Hospital of the University of Pennsylvania, Philadelphia PA.
The occurrence of left atrial tachycardias (AT) after catheter ablation for atrial fibrillation (AF) is common, especially after more extensive ablation of persistent AF. These AT are invariably symptomatic and often do not respond to medical therapy. The initial strategy involves ventricular rate control, cardioversion, and observation as some tachycardias may resolve with time. For persistent ATs, effective management frequently requires catheter intervention. Careful characterization of the tachycardia mechanism is essential in designing an effective ablation strategy that would also avoid further creation of pro-arrhythmic substrate. With this review, we summarize the incidence, mechanism, diagnosis and treatment of ATs occurring after AF ablation.
Correspondence to: Edward P. Gerstenfeld, MD, 9 Founders Pavilion, 3400 Spruce Street, Philadelphia, PA 19104.
Since its first description in 19981 there has been an exponential increase in the number of ablations performed for atrial fibrillation (AF). Ablation of paroxysmal AF is now considered standard second line therapy after treatment with a single antiarrhythmic drug according to the AHA/HRS consensus document.2 Persistent AF has also become a more common target of ablative therapy and studies have suggested that a more extensive ablation may confer better results.3-5 However, with more extensive ablation strategies, the incidence of post ablation atrial tachycardias (AT) has increased. These ATs are often highly symptomatic and resistant to antiarrhythmic medication (AAD). This review summarizes the incidence, mechanism, diagnosis and treatment of ATs occurring after AF ablation.
The incidence of post ablation AT is associated with the strategy and extent of prior ablation. In our experience using an ablation approach that incorporates wide proximal circumferential pulmonary vein isolation confirmed with a circular mapping catheter, ablation of non-pulmonary vein AF triggers, and avoidance of any linear left atrial lesions or ablation of fractionated electrograms, the post ablation incidence of AT is quite low (3.2%).6 However, when a solely anatomical approach of circumferential ablation around the PV ostia is performed without confirming electrical isolation, the incidence of ATs is much higher, ranging from 10% to 24%.7,8 This has led to most centers abandoning the solely anatomic approach to AF ablation. Some investigators have attempted the routine use of roof and mitral annular lines as prophylaxis against post ablation macroreentrant tachycardias. Using this approach, AT incidence has ranged from 10%9 to 30%.3,8,10,11 When ablation of complex fractionated electrograms alone is performed, post ablation AT may occur in approximately 15-25% of patients.5,12 With a more extensive ablation involving PVI isolation, empiric lines and electrogram based ablation all performed during the same procedure, the incidence of AT has been reported as high as 50%.3,7,13,14
Early versus Late Recurrence
AT can occur early or late after AF ablation. Early ATs are defined as AT occurring within 3 months after ablation and late ATs those that occur after the 3 month post-ablation period. The incidence of AT is particularly high in the early period post ablation with 89% of all ATs occurring the first month, 9% in the second and 2% in the third month.13 Although early AT occurrence does predict late AT or recurrence of AF13,15 approx imately one-third of tachycardias may resolve with conservative management.16 The presence of hypertension, persistent AF, left atrial (LA) size>4 cm, presence of structural heart disease and the lack of a successful ablation of all targeted PV have been independently correlated with early atrial tachyarrhythmias occurrence.16,17 The much higher incidence of AT during the early post-ablation period may partially be related to radiofrequency related inflammatory changes. Until the development of well defined fibrosis, tissue edema and inflammation can be pro-arrhythmic. In addition, like in all cases of acute local cardiac inflammation, ablation is likely to mount a systemic inflammatory response. Inflammation both at tissue and systemic levels are related to the development of arrhythmias. ATs that persist after a 3 month period or occur late after AF ablation typically do not usually resolve spontaneously and warrant intervention.13
Post ablation ATs can be divided based on their mechanism as focal or macro-reentrant Figure 1. The mechanism of AT is usually related to the prior ablation technique. In our early experience using segmental PV isolation, the most common type of AT was a focal pattern originating from a segment of a previously isolated PV.18 These findings are in agreement with the findings of Ouyang and colleagues19 who found that more of 80% of left ATs had focal origin from the PV. In contrast, when linear ablation is performed in addition to PVI, the majority of AT are macroreentrant.16
Figure 1. Mechanisms of typical post ablation tachycardias based on electroanatomic mapping. Panels A and B show focal tachycardias originating from the left and right superior pulmonary veins respectively. Panels C and D show typical macroreentrant tachycardias with clockwise mitral annular flutter at panel B and counterclockwise mitral annular flutter in panel D. LSPV left superior pulmonary vein, RSPV right superior pulmonary vein, MV mitral valve
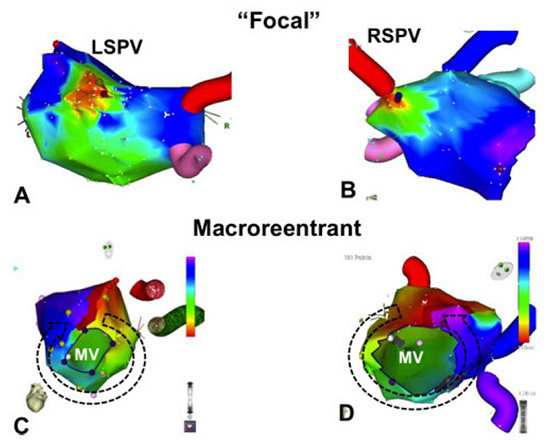
Although PV tachycardias often appear focal on electroanatomic mapping Figure 1, they demonstrate electrophysiologic characteristics consistent with reentry,6 including a flat-plus-increasing resetting curve, no change in tachycardia cycle length in response to adenosine, and the presence of mid-diastolic potentials.20 In support of a microreentrant mechanism, Oral and coworkers showed that an atrial input is often required for PV AT perpetuation.10 We believe that creation of such small reentrant circuits in the PV ostia is the direct result of non contiguous ablation resulting in slow conduction in a segment of the PV. In support of the above notion, reconnection of pulmonary veins is associated with both AT and recurrence of AF.18 Therefore, a circumferential approach to PV isolation should be performed and accompanied with confirmation of PV isolation to decrease the incidence of AT as well as AF recurrence after ablation.
Macroreentrant ATs seem to be more common after wide area of circumferential PV ablation or after linear ablation.3,21 Of the macroreentrant tachycardias, perimitral flutter is the most common, followed by roof dependent reentry and reentry around anatomic obstacles created by prior ablation including PVs.11,16 Linear ablation can lead to reentrant AT if block is not achieved facilitating slow conduction and reentry. In support of the proarrhythmic effect of non-complete linear ablation, Chae et al. reported that the critical isthmus in the vast majority of post ablation macroreentrant AT involves a prior ablation line and therefore consistent with a gap-related mechanism.9 Similarly, delivery of noncontiguous lesions in the LA during an electrogram based strategy may create isolated barriers that can serve as the center of a reentrant circuit, or create partially depolarized myocardium that might be a source of automatic or triggered rhythms.
It is therefore, apparent that the most important strategy in preventing post ablation ATs is to minimize the use of linear ablation during the index procedure. Isolation of PV should be the mainstay of therapy. In our experience, linear ablation should only be created to address clinical relevant macroreentrant tachycardias. In a recent paper, the best predictor of post-ablation mitral annular flutter was prior creation of a prior mitral isthmus ablation line.22 In cases where inducible or clinical arrhythmias dictate linear ablation, every effort should me made to confirm bidirectional block across any deployed lines.
Although extensive prior ablation may alter the surface ECG morphology of the tachycardia,23 the P-wave morphology may be quite useful when only PV isolation has been performed. One must obtain a clear P-wave that is not obstructed by a superimposed QRS or T wave. Examining the ECG after a premature ventricular complex or during AV block produced with ad enosine is extremely valuable. A clear isoelectric baseline among P-waves in all 12 leads of the ECG suggests a focal mechanism, whereas continuous activation suggests a macroreentrant mechanism Figures 2 and 3. At rapid rates, however, these two mechanisms may be difficult to distinguish.
Figure 2. Characteristic P wave morphology of focal tachycardias originating from pulmonary vein ostia. Notice that these tachycardias are inferiorly directed and positive throughout the precordial leads. Leads I, aVL and V1 are important in distinguishing left from right pulmonary vein origin and amplitude in the inferior leads help differentiating superior from inferior pulmonary veins. See text for details. LSPV left superior pulmonary vein, RSPV right superior pulmonary vein, LIPV left inferior pulmonary vein, RIPV right inferior pulmonary vein
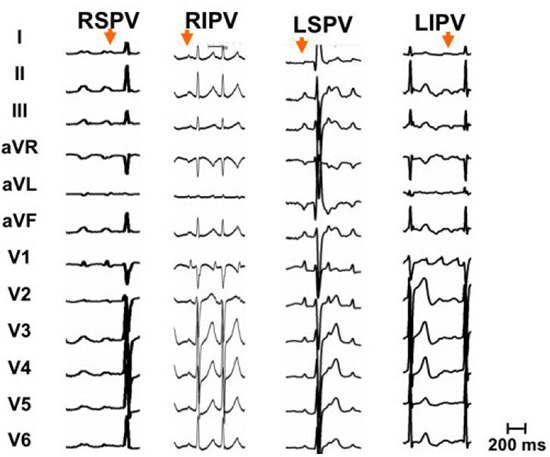
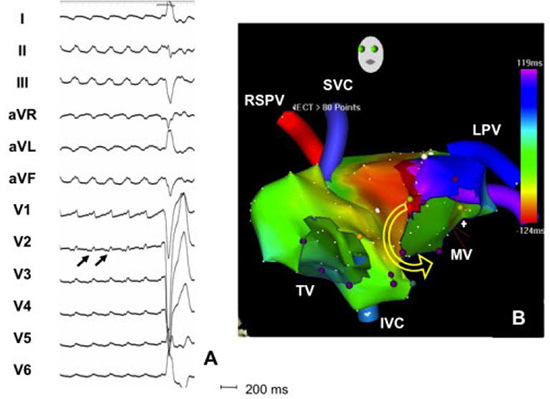
Figure 3. Surface ECG and biatrial activation map of counterclockwise mitral annular flutter. Continuous electrical activation without presence of isoelectric baseline between the flutter waves is suggestive of a macroreentrant tachycardia. Flutter waves are typically positive in all precordial leads with V2 of smaller amplitude that V1 and V3 since the mitral annulus is an anterior structure. In addition, flutter waves are inferiorly directed flat to negative in lead I. The right panel shows an electroanatonic biatrial map of the tachycardia that confirms the counterclockwise rotation of perimitral flutter with passive activation of the right atrium. RSPV right superior pulmonary vein, TV tricuspid valve, SVC superior vena cava, IVC inferior vena cava, MV mitral valve, LPV left superior pulmonary vein
Most focal ATs originate from the PV ostia. These can be readily identified as they are inferiorly directed and positive throughout the precordial leads, indicating a superoposterior left atrial origin. AT from the left PVs typically appear positive in V1 with a characteristic M shape P wave. In lead I, and are usually flat or biphasic and clearly negative in aVL Figure 2. Right PV ATs have a signature late peaking positive P wave in V1. In lead I they have a significant positive component, and lead aVL is flat or biphasic Figure 2. In the majority of cases inferior leads are positive for all four pulmonary veins as all PV are located fairly superiorly in the horizontal plain axis. Nevertheless, tachycardias originating from the very bottom of the inferior PVs may have a negative P wave in the inferior leads. Inferior leads may help in distinguishing left from right pulmonary veins as right veins typically give AT with a II/III ration of >1 as opposed to a ratio close to 1 for AT originating form left PVs.
The most common macroreentrant AT is mitral annular flutter, a largely iatrogenic arrhythmia. Counterclockwise mitral annular flutter (CCL MA) is positive in V1 and throughout the precordium. Flutter (F) wave in V2 typically has smaller amplitude than V1 and V3 and may have a small initial negative component. In addition, F waves are positive in the inferior leads and maintain negativity in lead aVL. Lead I can be negative, have a negative component or isoelectric Figure 3. Clockwise (CL) MA flutter has also positive F wave in lead V1 and the right precordial leads with often with a small initial negative component in the lateral precordial leads (V4-V6). The polarity in the limb leads is opposite to CCL MA flutter, thus negative in the inferior leads and positive in leads I and aVL. Due to continuous activation during atrial flutter, it is often difficult identify the onset of flutter wave. Based on our observations, the onset of flutter waves correlate with 80ms before the peak of the flutter wave in V1.
The differential diagnosis between left superior PV AT and CCL MA flutter can be challenging as both are positive in the inferior leads and across the precordium with a M-shaped P wave in V1. However, a negative component in lead I or an initial negative component in lead V2 suggests CCL left atrial flutter. In addition, MA flutters tend to have a cycle length of 240ms with less variation in cycle length than focal ATs however, there is a considerable overlap especially in patients with prior ablation lines.
Differentiation between CCL-MA and CL isthmus dependent right atrial flutter can also be challenging. Both are inferiorly directed; however, negative flutter wave in lead V1 is supportive of CL right atrial flutter. Finally, CL MA flutter is best distinguished from CCL right atrial flutter by lead I as CL MA flutter has a positive F wave in lead I, while CCL right atrial flutter is flat or biphasic in that lead.
Management of post AF ablation tachycardias largely depends on timing of occurrence, symptomatology and type of prior ablation. As previously mentioned, ATs often occur during the first three months after ablation without necessarily predicting procedure failure as up to a third of them may resolve with conservative management.21 In the case of severe symptoms, earlier intervention is sometimes warranted. Atrioventricular nodal agents should be maximized to achieve ventricular rate control. In our experience however, rate control is very difficult to achieve with pharmacologic agents. While AAD improve early recurrence of AF, its use has been associated with higher incidence of post ablation ATs (28% in patients receiving AAD versus 11% not receiving AAD; P<0.02).24 In addition, use of AAD was not superior to placebo in terminating ATs once they occur.24 The above paradox may be explained by AAD-related slowing of electrical conduction, a property inherent to Class IC antiarrhythmic agents. In our opinion, electrical cardioversion is indicated for early occurring persistent symptomatic AT. If the AT recurs soon after cardioversion, we would typically wait for at least 2 weeks for the ablation lesions to heal and for inflammation to subside before performing a repeat cardioversion. Although some have reported spontaneous resolution of AT occurring as late as 6 months after ablation, in our experience tachycardias that persist more than 3 months after ablation rarely resolve spontaneously and ablation is the most effective treatment.
Electrophysiologic Study – Diagnostic Maneuvers
Whenever conservative measures are ineffective, an electrophysiologic study (EPS) is the necessary next step for diagnostic and therapeutic purposes. The timing of the EPS depends again on the symptomatology, prior ablation strategy and type of AF. In cases of paroxysmal AF where the clinical AT seem to correlate with a previously identified PV trigger, subacute PV reconnection is the most likely mechanism. In those cases, we tend to repeat EPS earlier than 3 months to address PV reconnection. At the other end of the spectrum, in cases of persistent AF with extensive prior ablation we usually exhaust conservative measures during an initial 3-month period, awaiting for the atrial substrate to stabilize prior to repeating an EPS.
Electroanatomical activation mapping and entrainment maneuvers remain the cornerstone methods to identify the mechanism of the tachycardia and plan an effective ablation strategy. If a patient presents in AT, activation in the coronary sinus (CS) catheter gives rapid and valuable information about the origin of the tachycardia. Distal to proximal activation of the CS catheter excludes right atrial flutter or flutter around the septal pulmonary veins, whereas proximal to distal activation can be seen in right atrial flutter, CCL MA flutter, reentry around the septal PVs or focal ATs from the septal PVs Figures 4,5. Simultaneous activation of distal and proximal CS suggests ATs originating from the LA roof. We typically perform entrainment pacing from the cavotricuspid isthmus, prox imal and distal coronary sinus prior to transseptal puncture to exclude right atrial flutter and guide subsequent left atrial mapping. It should be noted that if a prior mitral isthmus line has been performed, then the CS activation pattern is less useful.
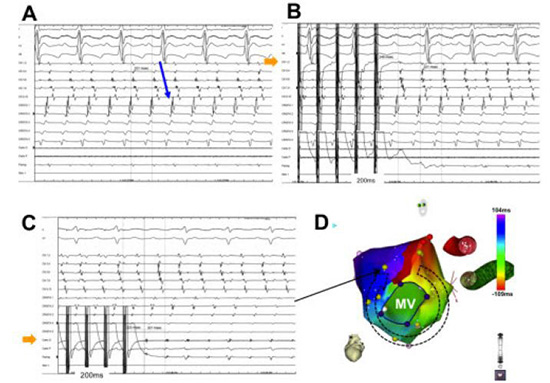
Figure 4. Focal atrial tachycardia originating from the left superior pulmonary vein (LSPV). Electrograms from two decapolar catheters and the mapping catheter (Map) are shown. One decapolar catheter is placed in the coronary sinus (CS) with proximal poles (CS9, 10) at the CS ostium and the other along the free wall of the right atrium (Crista). Panel A shows the CS activation sequence which is earlier at CS3, 4 suggestive of a tachycardia originating from the left atrium. Panel B shows entrainment from LSPV. Notice the similar LA activation sequence during pacing and the post-pacing interval within 20ms of the tachycardia cycle length. Panel C displays the electroanatomical map of the tachycardia
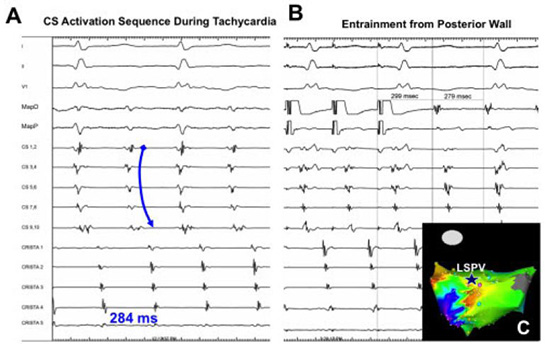
Figure 5. An example of counterclockwise mitral annular flutter. Panel A demonstrates that the activation pattern is distal-to-proximal in the coronary sinus (CS). Entrainment from the distal CS (Panel B, arrow) and superior mitral annulus (Panel C, arrow) with PPI-TCL <20ms establishes the diagnosis. The electroanatomic activation map is shown in Panel D
Once access to the left atrium is achieved, initial mapping usually begins with interrogating all four pulmonary veins for evidence of reconnection with a circular mapping catheter. Presence of mid-diastolic, low amplitude, fragmented or double potentials around the PV ostia is suggestive of an AT origin. Although, gaps in PV isolation are often encountered, the culprit vein can be identified by a characteristic reversal in activation with PV potential preceding the atrial potential at the reconnection site. Common areas of reconnection are the anterior ridge between the left PVs and left atrial appendage and inferior aspect of right inferior PV.
We next perform detailed activation mapping of the left atrium. A tachycardia with focal origin on the activation map with concentric spread originated from partially reconnected PV ostia is typically seen in patients with a PV reentry circuit Figure 4. Non-PV focal tachycardias can also be identified with the most common sites of origin include the posterior left atrium, and limbus of the fossa ovalis, left atrial appendage (LAA), CS ostium or inside the CS. If a macroreentrant AT is present, an “early meets late” pattern of activation is present, and the map incorporates the entire tachycardia cycle length Figure 1,3,5,6.
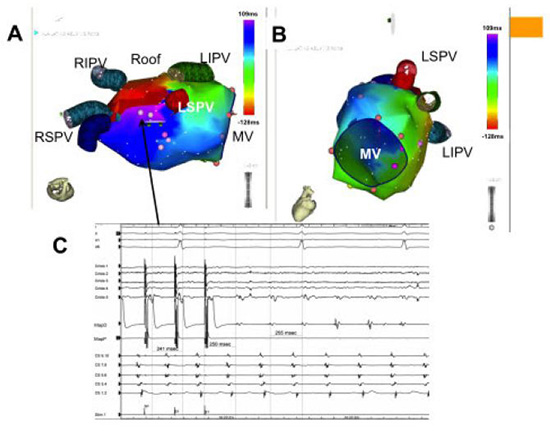
Figure 6. An example of a roof dependent left atrial flutter. Panel A: Electroanatomic map demonstrating “early-meets-late” on the roof of the left atrium. The range of the earliest and latest activation encompasses >95% of the TCL. Panel B shows passive activation of the mitral annulus excluding mitral annular flutter or figure-of-8 reentry. Panel C: Electrograms from two decapolar catheters and a mapping catheter (Map) are shown. One decapolar catheter is placed in the coronary sinus (CS) with proximal poles (CS9, 10) at the CS ostium and the other along the free wall of the right atrium (crista). The CS activation is predominantly distal-to-proximal but has a “fused” pattern consistent with a roof dependent left atrial flutter. Entrainment with PPI-TCL<20ms from the left atrial roof confirms the diagnosis of a roof dependent flutter. Ablation connecting the right and left superior pulmonary veins terminated the tachycardia
Once the activation map suggests a tachycardia mechanism, entrainment should be performed for confirmation prior to ablation. For a macroreentrant AT, entrainment from only 4 specific anatomic locations can often identify the tachycardia circuit. These sites include 1) the proximal CS 2) the distal CS 3) the septal MA and 4) the posterior LA roof. Entrainment with a post-pacing interval – tachycardia cycle-length (PPI-TCL) within 20ms from proximal and distal CS and septal MA is consistent with MA flutter Figure 5. When both the proximal CS and LA roof are part of circuit a macroerentry around the septal veins is suggested. Similarly, entrainment with PPI<20ms from the distal CS and roof suggests macroreentry around the left PVs. Finally, when the proximal CS, distal CS, septal MA and posterior LA roof are found to participate in the circuit one should consider figure-of-8 mitral flutter Figure 7. Figure 8 shows a diagnostic algorithm for approaching macroreentrant atrial tachycardias after AF ablation. For a focal AT, entrainment should be performed from the area of earliest activation or with mid-diastolic potentials to determine if it is a critical part of the reentry circuit. Higher output pacing may be required to capture in areas of diseased myocardium.
Figure 7. Double loop or figure-of-8 left atrial flutter. Electroanatomic map showing tachycardia rotating around the mitral annulus (panel A) as well as through the left atrial roof (panel B). Pacing from the proximal CS, distal CS, septal mitral annulus, and posterior roof all had PPI-TCL < 20ms. LSPV left superior pulmonary vein, RSPV right superior pulmonary vein, LIPV left inferior pulmonary vein, RIPV right inferior pulmonary vein, MV mitral valve, CS coronary sinus
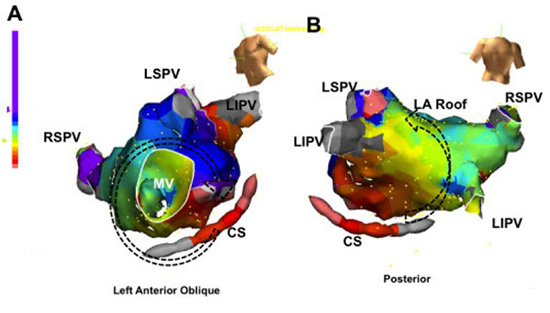
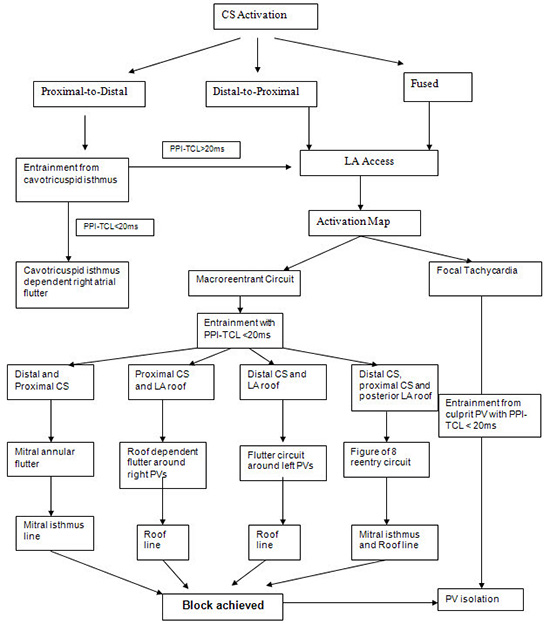
Figure 8. Algorithm to establish the diagnosis the mechanism of left atrial flutters occurring after AF ablation. Cavotricuspid isthmus=CTI; coronary sinus=CS; PPI post pacing interval; TCL tachycardia cycle length; LA left atrium; RA right atrium. PV pulmonary vein
Reviewing the prior ablation strategy is also important for mapping macroreentrant AT, as in the majority of cases the critical isthmus transverses an incomplete ablation line.19 Therefore, we examine all previously created lines for reconnected segments and look for low amplitude, fragmented or double potentials that represent slow conduction. In our experience and others, the reentry circuit with a protected isthmus can be identified in majority of cases.19
The mechanism of tachycardia is not identified with the above maneuvers. Common problems are a fast and unstable tachycardia, non-sustained tachycardia, multiple interchangeable circuits or non-inducible tachycardias. Cycle length variability of >15% of the tachycardia cycle length over a 1-minute observation is suggestive of a focal origin; however, a variability of less than 15% of the AT cycle length was non conclusive for either arrhythmia.25
Electrophysiologic Study – Ablation Strategy
After confirming the mechanism of the AT we then proceed with an ablation strategy to address the specific mechanism. For a focal AT, ablation is typically focused on the “early” region with a post-pacing interval closest to the tachycardia cycle length. This will typically involve a reconnected PV ostium. If the site of earliest activation is inside the PV, ablation should be performed proximally at the PV antrum to reisolate the PV. Whether additional ablation lesions at the site of earliest activation inside the PV will reduce recurrences is unknown, however we typically avoid this to reduce the risk of PV stenosis and since most patients do well with PV reisolation alone.
For a macroreentrant tachycardia, a linear lesion connecting the nearest anatomic boundaries should be performed. If MA flutter is the confirmed mechanism, the mitral isthmus line can be performed by creating a line from the lateral mitral annulus to the left lower pulmonary vein. An irrigated ablation catheter is typically required with power delivery up to 40 watts.26 Epicardial ablation lesions inside the CS opposite the endocrdial line are usually required more than 50% of cases to create complete isthmus block. An irrigated ablation catheter with power settings of 20-25W is typically required inside the CS. Attention should be made during RF application inside the CS to maintain the catheter orientation toward the right atrium. Occasionally, lesions inside the CS may be limited by inability to deliver adequate power. In this case, cardioversion and completion of the line in sinus rhythm can be performed. Even if tachycardia terminates during ablation, additional ablation should be performed until bidirectional block is achieved. This can be achieved by noting a reversal in the CS activation sequence from distal-to-proximal to proximal-to-distal while pacing from the left atrial appendage Figure 9. Performing an activation map during LAA pacing is another way to confirm isthmus block Figure 10. During ablation, particular attention should be given to segment closer to the left inferior vein as this is the most common site of reconnection.27 If the original tachycardia was figure-of-8 reentry, then the flutter may change to a roof dependent flutter once complete mitral isthmus block has been achieved. Again, noticing the change in cycle length and/or activation pattern is important for recognizing the occurrence of a new tachycardia Figure 11. In the case of a roof dependant macroreentrant flutter, a roof line can be delivered by ablating between the left and right superior PVs. Catheter stability in this region is often challenging, and use of a steerable sheath should be considered. Conduction block across the roof line can be documented by recording inferior-superior activation of the posterior wall during LAA pacing, or by performing a repeat activation mapping the LA during LAA pacing. In some cases, figure-of-8 reentry around the mitral annulus and through the LA roof can be found. In this case, one can either perform 1) both the mitral isthmus and LA roof lines to interrupt the AT or 2) perform a long line from the RSPV to the MA. We prefer the 2 short lines, as it can be quite difficult to achieve block across the long RSPV to MA line and this line may distort left atrial conduction including delaying conduction to the LAA. If tachycardia persists after performing a mitral annular and roof line, a more anterior mitral annular line can be delivered. If tachycardia persists after creation of the mitral annular and roof lines, one should repeat entrainment maneuvers to be sure the tachycardia path has not changed. If the same tachycardia is confirmed, cardioversion to sinus rhythm can be performed. If block along the lines is not present, continued ablation in sinus rhythm or during LAA pacing to achieve block will typically render the tachycardia noninducible.
Figure 9. Electrograms demonstrating conduction block across the mitral annular line between the left pulmonary veins and mitral annulus. Electrograms from three decapolar catheters and ablator are shown. One decapolar catheter is placed in the coronary sinus (CS) with proximal poles (CS9, 10) at the CS ostium. Another catheter is placed along the free wall of the right atrium (crista) and a third circular catheter (Lasso) is inside the left lower pulmonary vein. Panel A: While pacing from the mapping catheter (MapD) located in the left atrial appendage, baseline CS conduction occurs in the distal-to-proximal direction. Panel B: After endocardial ablation along the mitral isthmus, pacing from the appendage continues to show distal-to-proximal activation consistent with continued conduction through the mitral isthmus. Panel C: After epicardial ablation inside the CS opposite to the endocardial line, mitral isthmus block is achieved as indicated by the reversal of CS activation while pacing from the left atrial appendage
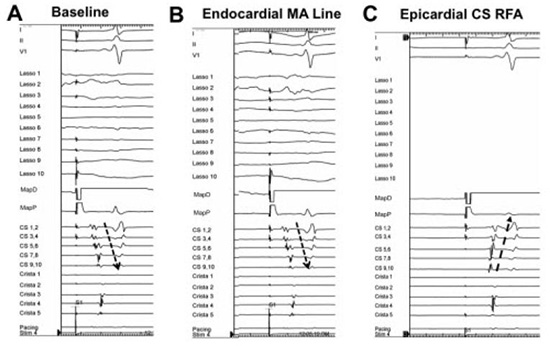
Figure 10. Intracardiac electrograms recorded from the coronary sinus (CS), right atrium (crista) and ablation catheter (ABL) in a patient with figure-of-8 reentry around the mitral annulus and through the left atrial roof. During mitral isthmus ablation, there is an abrupt change in coronary sinus activation without a change in tachycardia cycle-length. This suggests that mitral isthmus block has been achieved but the tachycardia persists using the second limb around the right pulmonary veins utilizing the left atrial roof. Linear ablation connecting the right and left superior pulmonary veins terminated the tachycardia
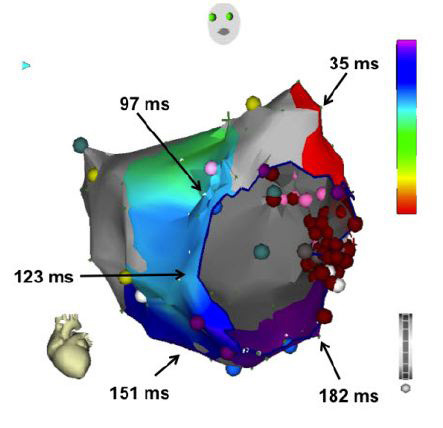
Figure 11. Activation map of the left atrium during pacing from the left atrial appendage after ablating the mitral isthmus with linear ablation between the left inferior pulmonary vein and mitral annulus (red dots). Note that activation occurs in a counterclockwise direction around the mitral annulus with latest activation lateral to the line, demonstrating mitral isthmus block
In cases where tachycardia is not inducible, un-mappable or unstable, one should proceed with re-isolation of the PVs, which may be the source of many focal tachycardias and can be required to trigger macroreentrant arrhythmias. The surface ECG may be the only useful tool to guide therapy in these cases. If the appearance was consistent with a focal PV AT, then PV reisolation is sufficient. If the ECG and AT CL is consistent with a macroreentrant mitral flutter, then creation of a linear lesion at the mitral isthmus may be warranted, providing that bidirectional isthmus block can be achieved.
In summary, post ablation ATs have become increasingly more common and constitute a significant clinical problem as they are typically very symptomatic. The occurrence of these ATs is a directly related to the extent of ablation performed in the initial procedure and are therefore more prevalent in persistent AF patients undergoing stepwise ablation that combines pulmonary vein isolation with linear and electrogram based ablation. Absence of conduction block across ablation lesions is of critical importance as it allows the generation of areas of slow conduction that provide the substrate for micro and macro reentrant circuits. Effective management of post-ablation ATs frequently requires catheter intervention, as medical therapy is frequently ineffective. Such intervention should always be guided by careful characterization of the tachycardia mechanism that would allow targeted ablation and avoid further creation of pro-arrhythmic substrate.