Pharmacological cardioversion of atrial fibrillation is an evolving topic and current opinion tends to consider recent-onset and long-lasting atrial fibrillation as two different entities, separated by “electrical remodeling” phenomenon. At the time of design of this study, current ACC/AHA/ESC guidelines’ revision for management of atrial fibrillation (2006)7 considered this evidence, with different recommendations for rhythm control of recent-onset and long-lasting AF. In this second case, amiodarone had IIa recommendation class while propafenone and quinidine were in class IIb. The reason of such a weak recommendation was that “quinidine is used less frequently than other pharmacological agents, due to the perception that it is less efficacious and has more frequent side effects, although direct comparative studies are lacking”.
The present report is the first one directly comparing these three drugs on this particular subset
of patients and our population is bigger than previous papers on pharmacological cardioversion of long-lasting AF, even if the size of our study is not comparable to major pharmacological trials.
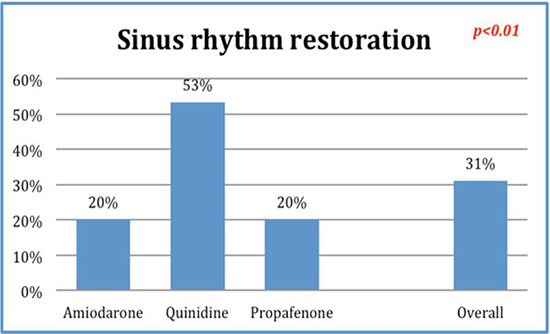
The main finding in this study is that conversion rate of patients treated with quinidine is more than twice that “conventional” drugs (53% vs. 20% p<0.01). There wasn’t any statistically significant difference among groups for parameters potentially predicting an increased risk of ADR or failure of cardioversion. In particular, atrial diameters weren’t significantly different among groups suggesting that anatomical remodeling (another important factor concurring to stabilize atrial fibrillation when it persists in time) did not influence our analysis, even considering the limitations of this method to estimate the phenomenon.
Multivariate in-group analysis did not find any correlation between historical or echocardiographic data and success rate for the procedure, showing that antiarrhythmic drug for pharmacologic cardioversion can probably be chosen considering its efficacy (and contraindications) only. The observation, albeit not statistically significant, of the trend to efficacy loss of propafenone with longer-lasting AF can be interpreted as a further confirmation that this drug should be kept for paroxysmal AF only. About quinidine’s side effects, acute oral loading of this molecule did not cause any significant adverse effect in studied patients. In particular, no case of tachyarrhythmia or syncope was recorded, neither any disturbance requiring the suspension of the therapy.
Concerns about quinidine began with the observation of “quinidine syncopes”8 associated with torsade the pointe and sudden cardiac deaths due to paroxysmal ventricular fibrillation. First described in 1848 by Van Heymingen and named by Pasteur in 1853, quinidine (class IA) has a long history as an antiarrhythmic. It has been used for decades for maintenance of sinus rhythm after cardioversion with an efficacy comparable to flecainide, but it has been progressively abandoned after the discovering of its proarrhythmic effect causing torsade de pointes and ventricular fibrillation, even if evidence is controversia.9 In 1990s several meta analyses posed severe doubts about safety of quinidine in chronic usage10,11, showing an increased risk of death if compared to other antiarrhythmics and placebo, leading to progressive discontinuation of this drug. Several recent trials12,13 and meta-analyses (including one from Cochrane collaboration14 noticed that fixed association of low dose quinidine and verapamil wasn’t inferior to sotalol in term of safety and efficacy, even if the second one is widely used for long-term sinus rhythm maintenance.
Concerning acute loading of quinidine for pharmacological cardioversion, there is no report in literature of a higher risk for patients than other antiarrhythmic drugs. Moreover, a recent retrospective analysis proved quinidine’s efficacy and safety on a large group of 501 patients with recent-onset atrial fibrillation.15 Also in this case, no life-threatening ADR associated with this kind of administration was reported and diarrhea was the only frequent side effect (13% of patients). In addition, quinidine is being studied for prevention of sudden death in Brugada syndrome16 as well as short QT syndrome17 and idiopathic ventricular fibrillation.18
Figure 1 Efficacy comparison among the studied drugs
There were two previous studies in literature comparing quinidine and amiodarone.19,20 Both showed similar conversion rates between drugs, but quinidine dose was not adequate and the studied populations were very small (40 and 30 patients respectively). Di Benedetto and colleagues compared propafenone with quinidine21 finding a clear superiority of the second one (24% vs. 84%) but patients enrolled had atrial fibrillation lasting no longer than six months. In another study,22 quinidine was compared to another IC drug (flecainide) in a population with a wide variability in duration of the arrhythmia. This last work did not find any significant difference within the two drugs but quinidine seems to have a higher conversion rate in the subset of patients with atrial fibrillation lasting more than 10 days.
During the writing of this paper a new revision of ESC guidelines for management of atrial fibrillation was released1. In this new edition, pharmacological cardioversion is considered for recent-onset cases only, while for the others DC shock is kept as the only recommended way to restore sinus rhythm. In our opinion, aside from patient’s major comfort, there are several situations (e.g. general medicine or geriatric departments, rehabilitation clinics, small structures etc.) where a monitored pharmacological approach can be a preferable solution, especially if drugs can be administered orally. Also, the absence of electrical shock avoids every risk of discomfort and device damage in paced patients. In conclusion, quinidine proved to be safe and effective for pharmacological cardioversion of atrial fibrillation and can be considered a first-line drug for this purpose.