Cryoablation for the Treatment of Drug Refractory Symptomatic Atrial Fibrillation: A Regional Medical Center Experience
Robert Tonks, MD*1,3, Hiba-tul-kareem Sayed, MD2,3, Ashley Adams, BA3, William T. Smith, MD, FACC1
1Duke University, Division of Cardiology, Clinical Research Unit, Durham, NC.2Carolinas Medical Center in Charlotte, NC.3New Hanover Regional Medical Center, Wilmington, NC.4Cape Fear Heart Associates, Wilmington, NC.
PVI is an effective, guideline-based treatment for drug refractory symptomatic AF. Balloon cryoablation has been shown to be a safe and effective method for PVI. In the STOP-AF trial, data was produced from practitioners performing PVI with significant experience at high volume centers. This study evaluates the effectiveness and safety of treating symptomatic, drug refractory AF with PVI via cryoablation after implementation in a regional medical center.
This represents a retrospective analysis of outcomes after cryoablation treatment for AF in 71 patients over 354.7 +/- 164.4 days. Reported and recorded episodes of AF were categorized into a representative percent of AF “burden” for each 90 day period. Primary effectiveness and safety end points paralleled those of the STOP-AF trial.
Patients undergoing cryoablation had a 91% reduction of AF burden at 6 months following the procedure with an event-free survival rate of 45.5 % at a mean follow up of 12 months. The mean burden reduction was 3.21% per quarter. Anti-arrhythmic and anticoagulant medication use was reduced by 14.3% and 26.8% respectively. Significant complications included one report of pulmonary vein stenosis, one report of pseudoaneurysm and 5.5% of patients had transient pericarditis or pericardial effusion following the procedure.
The results of this study were comparable to those of the high volume multi-center STOP-AF trial. PVI via cryoablation is a safe and effective alternative treatment of drug refractory symptomatic AF in the setting of a regional medical center.
Key Words : Cryoablation, Atrial Fibrillation, Pulmonary Vein Isolation, blation, Arrhythmia.
Correspondence to: Robert Tonks, Duke University Heart Center,
Clinical Research Unit, 30 Duke Med Cir,
Baker House, Suite 183, DUMC Box 3963,
Durham, NC 27710.
Atrial fibrillation is a significant and growing health issue affecting more than an estimated 3.4 million people.1,2 Atrial fibrillation often has a negative impact on quality of life, is the leading cause of stroke, and increases the risk of kidney disease and heart failure.3,4,5,6 In addition, the cost to the United States healthcare system to treat atrial fibrillation is estimated at a staggering $12 billion.7
Pulmonary vein isolation via cryoballoon ablation was shown to be an effective treatment of drug refractory symptomatic atrial fibrillation in the STOP-AF trial.8,9 Cryoablation demonstrated up to 70% effectiveness rate among patients with drug refractory atrial fibrillation compared to only 7% among those treated with anti-arrhythmic therapy alone. The safety and effectiveness of balloon cryoablation demonstrated in, and following, the STOP-AF trial helped pave the way for cryoablation to become a widely accepted FDA approved treatment option for the treatment of symptomatic, drug refractory atrial fibrillation.9,10
The primary operators and centers which were included in the STOP-AF trial were referral centers with developed AF treatment programs, and large high-volume academic medical centers. There is limited data available regarding the effectiveness and safety of PVI cryoablation in the setting of mid-sized regional medical centers, since its implementation. The purpose of this study is to review the outcomes of the first series of patients undergoing balloon cryoablation following adoption of the technique at a regional medical center.
This study is an IRB approved retrospective analysis of 71 patients that underwent cryoablation between March 2012 and November 2013 at New Hanover Regional Medical Center in Wilmington, NC. These represent the first 71 patients to receive cryoablation at this institution. Procedures were collectively performed by four Electrophysiology Board Certified physicians, experienced at performing ablation, but none of whom had performed any cryoablation procedures prior to this study. Pulmonary vein isolation via radio frequency ablation was only rarely performed at this institution prior to the availability of the cryoablation technique. All physicians were trained at Medtronic, Inc. The cryoablation procedure was a single transeptal technique using an Achieve catheter. The length of the lesions with the initial “Arctic Front” balloon were goal two, 4 minute lesions performed at each site and minimum tolerated temperature of -60 degrees Celsius. With the newer generation “Arctic Front Advance” balloon, the lesions were goal two, 3 minute lesions performed at each site and minimum tolerated temperature of -55 degrees Celsius. Generally, no provocative testing was performed following demonstration of block, and additional lesions were only applied if an entrance/exit block was not demonstrated.
Patients were Caucasian (100%), predominately male (67.6%) with an average age of 65.4 years [SD 10.5] (Table 1). The vast majority of patients had paroxysmal atrial fibrillation (78.9%) and the rest were recently determined to be early persistent atrial fibrillation. Atrial fibrillation events were recorded most often with a wearable device (53.5%), and most patients had a normal ejection fraction (84.5%), a CHADS2 score less than 2 (59.2%) and a NYHA class of 0 (80.3%, Table 1). A history of atrial flutter was recorded in 36.6% of patients (26.8% had a history of atrial flutter ablation), and an enlarged left atrial diameter was noted in 49.3% patients (Table 1).
Table 1. Patient Demographics and Medical History Characteristics
| Characteristic |
Patients N = 71 |
| Age |
|
65.4 ± 10.5 |
| Gender |
Male |
48 (67.6) |
|
Female |
23 (32.4) |
| Race |
Caucasian |
71 (100%) |
| Atrial Fibrillation Burden Reporting Method |
Internal Device |
13 (18.3) |
|
External Device |
38 (53. 5) |
|
Patient Reported |
20 (28.2) |
| Left Ventricle Ejection Fraction |
Normal (≥ 55 %) |
60 (84.5) |
|
Abnormal (< 55 %) |
10 (14.1%) |
| CHADS2 |
0 |
13 (18.3) |
|
1 |
29 (40.9) |
|
2 |
14 (19.7) |
|
3 |
14 (19.7) |
|
4 |
1 (1.4) |
| NYHA |
0 |
57 (80.3) |
|
1 |
5 (7.0) |
|
2 |
4 (5.6) |
|
3 |
5 (7.0) |
| History of Artrial Flutter |
|
26 (36.6) |
| History of Artrial Flutter Ablation |
|
19 (26.8) |
| Left Atrial Diameter |
Normal |
33 (46.5) |
| Comorbidities |
Abnormal |
35 (49.3) |
|
Missing |
3 (4.2) |
|
Hyperlipidemia |
53 (74.7) |
|
Hypertension |
52(73.2) |
|
Tobacco Use |
37 ( 52.1) |
|
Coronary Artery Disease |
28(39.4) |
|
LVH |
24(33.8) |
|
Diabetes |
19 (26.8) |
|
Thyroid Disorder |
19 (26.8) |
|
OSA |
18 (25.5) |
|
Moderate to Severe Valve Disease |
17 (24.0) |
|
Congestive Heart Failure |
14 (19.7) |
|
Sick Sinus Syndrome |
13 (18.3) |
|
Low Testosterone |
5 (7.0) |
Data reported in N(%),Mean ± Std, or Median [Q1-Q3]
Atrial fibrillation burden was collected by patient reported symptomatic atrial fibrillation, through evaluation of external monitors and from interrogation of implanted devices. Symptomatic atrial fibrillation was extrapolated from patient reports in days per week, through emergency department visits for atrial fibrillation and through cardiology clinic appointments for symptomatic atrial fibrillation. These data were collected from two sources due to the hospital converting from paper charts to electronic medical record during the time frame of this retrospective study. However, all of the data was extracted from available medical records. The reported and recorded atrial fibrillation event days were divided by the number of days reviewed since the prior follow up to estimate the burden of self-reported atrial fibrillation. Over each period, the representative percent burden of atrial fibrillation that the patient experienced was recorded. Follow up data after cryoablation was also recorded in the same manner for reported and recorded data for the correlating time periods of the study.
Overall treatment success was determined similarly to that in STOP-AF. Acute procedure success was achieved through demonstration of electrical isolation in at least three of four pulmonary veins following the cryoablation procedure. All patients received pre-procedure CT or MRI vein assessment. If common ostium were discovered, they were ablated with cryo balloon ablation and if successfully isolated then each of the branches of the common ostia were considered as an acute procedure success. Freedom from recurrent atrial fibrillation after a post procedure 90 day blanking period was also assessed, as noted. Additional measures included the proportion of patients with subsequent elimination of anti-arrhythmic drug requirements as well as anticoagulation following the 90 day blanking period. Safety of the procedure was assessed by measuring rates of cryoablation related adverse events including pericarditis or pericardial effusion, pulmonary vein stenosis, phrenic nerve palsy, pseudoaneurysm, atrial esophageal fistulas, tamponade, stroke and death.
Categorical data were reported in counts and frequencies, and analyzed, as appropriate, using Χ² test of independence or Fisher’s exact, as contingency tests used when evaluating two categorical variables. Numerical data were reported in means and standard deviations or least square means. Multi-factorial data was analyzed using one or two factor analysis of variance using a bonferroni adjustment to guard against error of bias due to overestimated p values caused by repeated testing effects, by using p values divisible by the number of factors being evaluated. Analysis was performed with SAS 9.0 (SAS Institute, Cary NC). P-values less than 0.05 were considered significant.
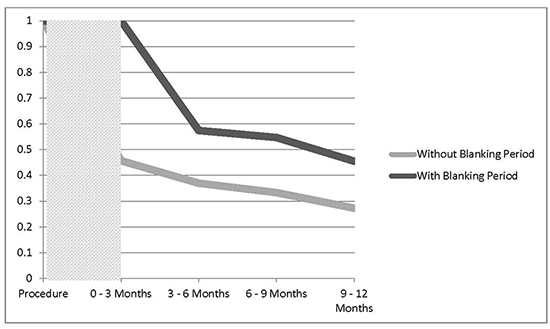
The acute procedure success was 98.6% and the absolute atrial fibrillation-free survival rate was 45.5% at 12 months after cryoablation. (Figure 1) shows the event-free survival rate following pulmonary vein cryoablation at New Hanover Regional Medical Center. Two survival curves are shown. One which represents the event free survival rate without the blanking period, and the other following the 90 day post procedure blanking period, as was performed in the STOP-AF trial. This blanking period is used to reduce the apparent skewing of the results due to a mild increase in arrhythmias which commonly follows ablation procedures.
Figure 1. Kaplan-Meier Survival Curve. Survival free from recurrent atrial fibrillation following cryoablation. Graphs shown represent the absolute event free survival of the population (grey) and the survival following a 90 day blanking period following ablation (black)
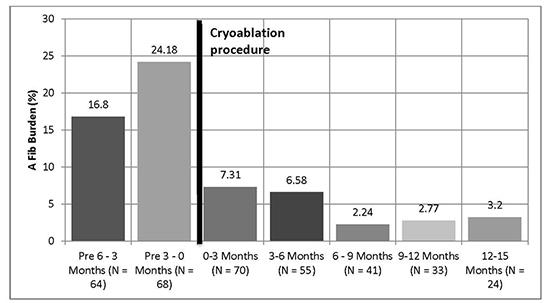
Although the absolute event-free survival rate was lower in our series, compared with that seen in the STOP-AF trial, there was a significant reduction in atrial fibrillation burden over time following PVI by cryoablation. (Figure 2) shows the burden of atrial fibrillation among the pooled population at baseline (24% in the 3 months prior to ablation) and for each three-month interval following PVI cryoablation. As shown in the figure, there was a decrease in mean atrial fibrillation burden from a baseline of 24% prior to cryoablation, to approximately 3% twelve months after cryoablation (p<0.0001). This represents an eight fold reduction in the overall burden of AF in the population. In addition, the modeled reduction of 3.2% per quarter was also a highly significant trend (p <0.0001).
Figure 2. Atrial Fibrillation Burden Over Time. There is a significant reduction in AFib burden from the pre-procedure time frames to the post-procedure time frames. Mean burden reduction of 3.21% per quarter. Generalized Linear Model, p<0.0001
Analyses were performed to assess for variables which may have contributed to procedural success rates. Ejection fraction, left atrial diameter, moderate to severe valve disease, sick sinus syndrome and prior atrial flutter ablation played no significant role in determining the change in AF burden. The only variable which was significantly related to procedural success rates in our series was the CHADS2 score. For each unit increase in CHADS2 score, the patient experienced an average increase in atrial fibrillation burden of 2.5% (p = 0.0085). This may have led to lower post procedure event free rates in patients with higher CHADS2 scores as only 12.5% of patients with scores of 2 or less had at least one episode of AF by six months following cryoablation compared to 73% of patients with a score of 3 or greater.
Two patients required repeat ablation procedures, both of which occurred during the earlier portion of patients that had cryoablation in this series. Significant complications include one report of pulmonary vein stenosis, one report of pseudoaneurysm and 5.5% of patients had transient pericarditis or pericardial effusion following the procedure. In each case the symptoms resolved and there were no lasting side effects.
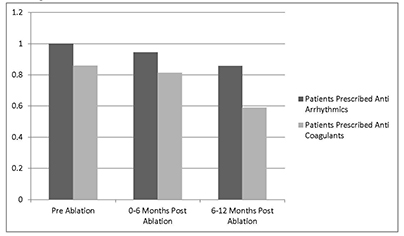
The use of anti-arrhythmic drug therapy and anticoagulation was also decreased following cryoablation procedure. At 12 months after receiving a cryoablation procedure in our medical center, approximately 15% of patients being treated with anti-arrhythmic therapy (p = 0.0033) and roughly 30% of the patients being treated with anti-coagulants (p = 0.0006), no longer required therapy as shown in Figure 3
Figure 3. Patients Prescribed Antiarrhythmics and Anticoagulants Pre- and Post- Procedure. Patients were prescribed significantly fewer anti-arrhythmic medications (shown in dark grey, p = 0.0033) and anti-coagulants (shown in light grey, p = 0.0006) in the months following cryoablation
The procedure success rate, as measured by event free survival without atrial fibrillation at 12 months, was lower in this series (45.5%) than in the STOP-AF trial (69.9%).9 The reasons for this discrepancy are likely multifactorial. As with any new technology or technique, there is a learning curve upon implementation. Procedural effectiveness for cryoablation has been correlated to the number of procedures which an operator has performed.11 All four physicians at New Hanover Regional Medical Center being represented in this series were performing their first cryoablation procedures. The patients in this series therefore represent a cohort when the operators were relatively inexperienced. It is possible that higher overall treatment success rates could be achieved as the operators and institution become more experienced at performing the technique, which echoes the importance of reviews such as this following implementation of a new technique.
There was also a change in the cryoballoon technology after approximately the first one third of the data was recorded during the collection period. The Medtronic Arctic Front “Advance” Cryoballoon replaced the first-generation balloon in the later cases. The second generation “Advance” balloon has more cooling jets resulting in a more expansive and homogenous cryoablation surface area spanning the entire front hemisphere. This improvement in balloon technology, larger treatment surface area compared to the equatorial cryoablation surface of the first generation cryoballoon and potentially more complete ablation lesions may have led to more effective ablation outcomes later in the study.12 Unfortunately, the exact timing of the change in balloon technology was not recorded.
As our series and the STOP-AF population were not from the same cohort, patient selection or patient characteristics may have contributed to different outcomes. In our series, a higher CHADS2 score correlated with lower rates of treatment success following cryoablation. This likely reflects the greater comorbidity burden in patients with higher CHADS2 scores. Those with more complex medical histories, such as CHF and hypertension, may be less optimal candidates for the procedure. Different baseline patient characteristics could have contributed to some of the difference between our series and the STOP-AF patients, but due to the small sample size of this series it is difficult to comment on this with any certainty.
Despite a lower absolute event-free survival procedural success rate, there remained a large reduction in the overall burden of atrial fibrillation. This is perhaps more important to the patient than the absolute event reduction rate. Though about half of the patients had recurrence of atrial fibrillation, their burden was dramatically reduced.
Limitations of this study include a change in the cryoablation procedure technology during the data collection period and the use of two different forms of medical records as previously described. Data abstraction was carried out with the same measures in each system, to maximize consistency. One of the major challenges in our series was to translate patient-reported and external monitor recorded information regarding the presence and amount of atrial fibrillation into a quantifiable “burden”. This required extrapolation and some estimation of reported burden into a quantifiable burden, as only a minority of patients had an implanted device to record a true burden.
In the setting of a regional medical center, pulmonary vein isolation via cryoablation for the treatment of drug refractory, symptomatic atrial fibrillation compares favorably to prior reports from larger hospital centers in the STOP-AF trial. Although a smaller event-free survival was seen, there was a dramatic reduction in the overall atrial fibrillation burden with minimal adverse events, and a reduction in antiarrhythmic and anticoagulation requirements.