The Wearable Cardioverter/Defibrillator – Toy Or Tool?
David Duncker, MD, Christian Veltmann, MD
Department of Cardiology and Angiology, Hannover Medical School, Carl-Neuberg-Str. 1, D-30625 Hannover.
After the success story of implantable cardioverter/defibrillator systems, prevention of sudden cardiac death (SCD) remains one of the main duties in cardiology. For patients with unkown or transient risk profile for SCD, a wearable cardioverter/defibrillator (WCD) has been established for temporary and effective prevention of sudden arrhythmic death. Several studies have shown safety and efficacy of the WCD, even though randomized studies proving a mortality benefit are still lacking. This review provides an overview of actual WCD data and usage, special indications and possible risks and complications. WCD use is effective and adequate for temporary prevention of SCD in chosen populations. In particular, it provides secured time for sophisticated risk stratification to identify patients at persistent risk for SCD. Nevertheless, prospective randomized trials seem mandatory to prove a prognostic relevance and the economic value of this device.
Key Words : Sudden Cardiac Death, Ventricular Arrhythmia, Wearable Cardioverter/Defibrillator.
Correspondence to: David Duncker Department of Cardiology and AngiologyHannover Medical SchoolCarl-Neuberg-Str. 1D-30625 Hannover Germany.
Sudden cardiac arrest (SCA) due to tachyarrhythmias remains a major cause of death in western countries.1,2 The implantable cardioverter/defibrillator (ICD) has been used for more than 30 years and is considered one cornerstone for primary and secondary prevention of sudden cardiac death (SCD) in high-risk patients.3 Decades have passed since the milestone trials in ICD therapy. The MADIT, MADIT-II and SCD-HeFT trial enrolled patients between 1990 and 2001.4-6 Since then, interventional and drug therapies for these patients have evolved tremendously and concomitantly may have affected risks for SCD. Additionally, ICD therapy may be accompanied by several device-related problems, especially lead failure.7 Recently, van der Heijden et al. described an overall-incidence for device related adverse events of 20% inappropriate shocks, device infections of 6% and 18% lead failures within 12 years of follow-up.8 ICD therapy has been shown to be cost-effective.9,10 However, a relevant proportion of patients getting implanted do not meet evidence-based criteria for implantation.11 Therefore, careful but however secured risk assessment before ICD implantation may be even more cost-effective.
A large number of possible risk markers like microvolt T-wave alternans, tests for autonomic dysfunction or signal averaged ECG have been proposed. Though, they did not find their way into clinical routine and are actually not supported by the guidelines.12 The only evidence-based risk stratifying marker today remains left ventricular ejection fraction, thereby reading “left ventricular ejection fraction (LVEF)”.12
Having said this, in a recent study, Sjöblom et al. investigated evolution of LVEF in 91 patients after myocardial infarction (MI).13 45% of the patients met the ICD criteria of LVEF ≤35% 40 days after MI. However, the authors found further significant improvement in LVEF in 6 more patients at 3 months follow-up (p=0.01), meaning that these patients no longer met the criteria for ICD implantation. These findings show that there is further improvement in LVEF beyond the initial 40 days post MI. Furthermore, 10% of the patients presented with life-threatening ventricular arrhythmias within the first 9 weeks post MI, emphasizing the arrhythmic risk and the need for antiarrhythmic prophylaxis in this early phase. Accordingly, in patients with recent onset non-ischemic cardiomyopathy, the IMAC study showed no benefit from early ICD implantation.14
Wearable Cardioverter/Defibrillator
For patients with unkown or transient risk profile for SCD, a wearable cardioverter/defibrillator (WCD) (LifeVest®, ZOLL, Pittsburgh, PA, USA) has been established for temporary but however effective prevention of sudden arrhythmic death (Figure 1). The WCD continuously analyzes the heart rhythm using 4 non-adhesive electrodes incorporated in a light garment. The ECG is registered via 2 non-standard leads (front-back and side-side). When a life-threatening arrhythmia is detected, the WCD runs an alarm cascade
including audible, visual and tactile alarms. If the patient is conscious,
he can withhold any therapy by pressing two response buttons on the
monitor. In case of unconsciousness and consequently release of the
response button, the WCD will deliver a biphasic shock after having
deployed contact gel through the contact electrodes. Programming
of detection rates can include a ventricular tachycardia (VT) and a
ventricular fibrillation (VF) zone, programmable from 120 to 250
beats per minute. The shock energy can be chosen from 75 to 150
J. All ECG with detected arrhythmias are stored in the device and
regularly transferred to a webserver where the attending physician
can review all episodes as well as the patient’s compliance. Since the
arrhythmia detection of the WCD is performed via surface nonadhesive
electrodes, there is a considerable risk for motion-related
noise artifacts.
Figure 1 Wearable Cardioverter Defibrillator (WCD)
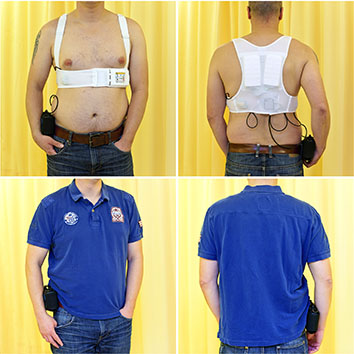
The WCD is used in patients at undefined or temporary risk for
SCD as well as in patients at known persistent high risk but with
transient contraindications for implantation of an ICD. Common
indications for WCD wearing are shown in table 1.
Table 1. Common indications for wearable cardioverter/defibrillators49
|
| Acute myocardial infarction with/without PCI and a LVEF ≤35% |
| Coronary revascularization (PCI or CABG) and a LVEF ≤35% |
| Non-ischemic cardiomyopathy, (acute) myocarditis, stress (Takotsubo) cardiomyopathy, peripartum cardiomyopathy, LVEF ≤35% |
| Waiting for heart transplantation |
| ICD explantation until re-implantation) |
| Post VT ablation in patients with only moderately reduced LVEF. |
WCD often gets misclassified as an alternative to permanent ICD
or a “bridge to ICD”. This does not give sufficient consideration to
the capabilities and the concept of WCD usage. This review provides
an overview of actual WCD data and usage, special indications and
possible risks and complications.
In 1998, Auricchio et al. reported the first 10 patients with
successful termination of ventricular arrhythmia by the WCD.15
Subsequently, WCD was shown to be safe and effective in detection
and termination of VF.16-18
Table 2 summarizes available data on WCD. Despite the manifest
gaps in evidence, based on these registries and case reports, the actual
ESC guidelines on prevention for SCD give a Class IIb level of
evidence C indication for the WCD “for adult patients with poor LV
systolic function who are at risk for sudden arrhythmic death for a
limited period, but are not candidates for an implantable defibrillator
(e.g. bridge to transplant, bridge to transvenous implant, peripartum
cardiomyopathy, active myocarditis and arrhythmias in the early
post-myocardial infarction phase)”.12
Table 2. Registries on WCD (MI: myocardial infarction, PPCM: Peripartum cardiomyopathy, NICM: non-ischemic cardiomyopathy, CHD: congenital heart disease, IA: inherited arrhythmias)
| Publication |
Year |
Patients (n) |
Etiology |
Prospective/ retrospective |
Appropriate shocks |
Compliance (h/d) |
Cumulative wearing time |
Unnecessary shocks |
| WEARIT/ BIROAD18 |
2004 |
289 |
Miscellaneous |
Prospective |
6 |
n/a |
Mean 3.1 months |
6 (2%) |
| Klein50 |
2009 |
354 |
Miscellaneous |
Retrospective |
27 (27.6%) |
Mean 21.3 |
Mean 106 days |
0 |
| Saltzberg38 |
2009 |
258 |
PPCM/ NICM |
Retrospective |
0/2 |
18.3/17.0 |
Mean 75±81/56±54 days |
0/0 |
| Dillon17 |
2010 |
2105 |
Miscellaneous |
Retrospective |
54 |
Median 21.3 |
Median 36 days (3-365) |
34 |
| Chung47 |
2004 |
3569 |
Miscellaneous |
Retrospective |
76 (2.1%) |
Mean 19.9±4.7 |
Mean 52.6±69.9 days |
67 (1.9%) |
| Rao32 |
2011 |
162 |
CHD, IA |
Retrospective |
0/2% |
Mean 19 |
Mean 27/29 days |
0/3% |
| Epstein19 |
2013 |
8453 |
Post MI |
Retrospective |
146 shocks in 133 (1.6%) patients |
Median 21.8 |
Mean 69±61 (median 57) days |
114 in 99 patients |
| Zishiri51 |
2013 |
4958 |
Miscellaneous |
Retrospective observational cohort study |
18 shocks in 11 patients |
n/a |
n/a |
n/a |
| Tanawuttiwat40 |
2014 |
97 |
Explantation of ICD |
Retrospective |
4 |
Median 20 |
Median 21 days |
2 |
| Sasaki52 |
2014 |
9 |
|
Retrospective |
1 |
Mean 23.7 |
Mean 21 days |
0 |
| Duncker39 |
2014 |
9 |
PPCM |
Prospective |
4 |
Mean 22.0±2.4 |
Mean 133±103 days |
0 |
| Opreanu34 |
2015 |
122 |
Candidates for cardiac transplant |
Retrospective |
7 |
Mean 17±7 (median 20) |
Mean 127±392 (median 39) days |
2 |
| WEARIT-II24 |
2015 |
2000 |
ICM, NICM, CHD |
Prospective |
120 in 41 patients |
Median 22.5 |
Median 90 days |
10 patients |
Epstein et al. analyzed 8453 patients wearing the WCD early
post MI 19 and found 133 patients (1.6%) receiving 309 appropriate
shocks. Beyond that, 114 inappropriate shocks occurred in 99
patients. Mean time from MI to WCD prescription was 9±9 days and
mean time from prescription to first shock delivery was 22±32 days.
This shows that commonly accepted waiting time between MI and
ICD implantation of 40 days and 3 months respectively nevertheless
may remain a period at risk for life-threatening arrhythmias. These
findings are consistent with those of the VALIANT study which
found the highest mortality in the first 30 days after MI.20 The
DINAMIT21 and IRIS22 study suggested no survival benefit from
ICD implantation early post MI, even though in both studies, the
rate of SCD was halved in the ICD group. Unfortunately, this
reduction of SCD was negated by an increased number of non-sudden
cardiovascular deaths.23 The reason for this difference has not been
clarified sufficiently. Furthermore, relevant differences are obvious
comparing the populations of these two studies with a population
of a recent heart failure trial, e.g. in terms of optimized heart failure
medication. Given the great improvement in heart failure therapy
and interventional development for the treatment of acute MI in the
last years, the amount of non-sudden cardiac deaths may therefore
be relevantly reduced, leaving the amount of preventable arrhythmic
death at the disposal of antiarrhythmic devices.
Despite the great amount of descriptive or cohort studies on
WCD and the presumed self-evident benefit of the WCD, most of
these studies lack a control group, not to mention a randomization.
Therefore, the net benefit of the WCD still remains to be proven.
The only randomized controlled trial on patients after MI is the
multicenter VEST trial with a total of 1900 randomized patients
(www.clinicaltrials.gov, NCT01446965), which is about to complete
enrollment. The primary endpoint is sudden death mortality.
The recently published WEARIT-II prospective registry24 enrolled
2000 patients receiving WCD for a median of 90 days. 40% of the
patients had ischemic cardiomyopathy, 46% had non-ischemic
cardiomyopathy and 14% had congenital heart disease. Authors
reported 120 ventricular tachyarrhythmias in 41 patients (2%) and
only 10 patients (0.5%) receiving an inappropriate shock. Even
though the WEARIT-II registry represents the greatest prospective
database on WCD published to date, it still does not answer the
relevant unsettled questions on hard endpoints in WCD use.
Especially for possibly transient circumstances which may
temporarily elevate the risk for life-threatening arrhythmias,
the WCD offers protected time for any further diagnostics, risk
stratification, re-evaluation or simply letting any sort of therapy work
without letting the patient at risk during this period.
Successful use of the WCD has been described in acute
or suspected myocarditis,25,26 stress cardiomyopathy, 27,28
noncompaction cardiomyopathy,29 alcohol toxic cardiomyopathy,30
congenital heart disease24,31,32 as well as in children.33 Candidates
for cardiac transplantation can likewise be provided a WCD until
transplantation.34 However, since these days waiting for a donor
organ may take several years, implanting an ICD has to be seriously
considered.
ICD therapy in inherited arrhythmia syndromes like long QT
syndrome, Brugada syndrome or arrhythmogenic right ventricular
dysplasia is challenging due to the rather young patient age, limited
available data and elevated complication rate.35 WCD can facilitate
and cover time to diagnosis and risk stratification in these patients. In
the study by Rao et al., among 119 patients with inherited arrhythmias
receiving a WCD, the predominant indication was pending genetic
testing.32 Again, the WCD should not be considered as a bridge to
ICD in these patients, but rather as a tool for serving protected time
to exclude a diagnosis or to stratify the patient as low-risk and thus
omitting ICD implantation.
Peripartum cardiomyopathy
The concept of temporary prophylaxis for SCD of the WCD
is particularly attractive for transient pathologies leading to a
temporarily elevated risk for SCD. Peripartum cardiomyopathy
(PPCM) represents a rare idiopathic cardiomyopathy leading to
heart failure LV dysfunction towards the end of pregnancy or in
the months following delivery.36 Even if initial LVEF often is
severely affected at the time of diagnosis, there is a high potential
for LVEF recovery after starting an optimal heart failure medication
regimen.37 In a large cohort of 107 patients with PPCM wearing a
WCD, Saltzberg et al. did not report any arrhythmic event (though
3 patients died after WCD use).38 This may eventually be due to the
retrospective character of their analysis. On the contrary, we recently
reported 12 consecutively admitted patients with first diagnosis of
PPCM.39 7 patients presented with a LVEF of ≤35% and received
a WCD for 3 to 6 months. Among these 7 patients, we observed 4
events of VF in 3 of the patients. Patients significantly recovered in
LVEF during follow-up. Our data strongly suggest an elevated risk
for life-threatening arrhythmias in these young mothers early after
diagnosis of PPCM and warrant an uninterrupted use of the WCD
in all patients in the early phase of PPCM during recovery.
ICD explantation due to infection
Due to increasing numbers of ICD implantations and subsequent
generator exchanges, numbers of device infections with need of
system explantation are increasing, too. Especially for patients
with secondary prophylactic indications for an ICD, continuous
monitoring after explantation seems mandatory. Besides inpatient
monitoring for the period of antibiotic therapy, outpatient
management using a WCD for this period seems reasonable.
Tannawuttiwat et al. presented a retrospective analysis of 97 patients
wearing a WCD after ICD removal.40 2 patients received 4 shocks,
1 patient received 2 unnecessary shocks. 8 patients (8.2%) died (5
patients in hospital, 3 patients at home), no one was wearing the
WCD at the time of death. In a cost-effectiveness model, the WCD
was shown to be cost-effective in comparison to inpatient strategy
until re-implantation.41
WCD can successfully be used in cancer patients who often present
a contraindication for ICD implantation.42 Special considerations
can be raised on patients with planned radiotherapy adjacent to an
implanted ICD. Depending on the local findings and the planned
radiation protocol, an explantation of the ICD with temporary use of
a WCD for the radiation period and and subsequent re-implantation
may be a favorable strategy, as reported by Bowers et al.43
Patients who are in end-stage renal disease are known to have
a high risk for ventricular arrhythmias, but are as well known to
show reduced benefit from primary prophylactic ICD therapy due
to competing risks.44 Nevertheless, Wan et al. reported 84 SCA
episodes in 75 patients on hemodialysis showing the important
arrhythmic burden in these patients.45 Not all of these episodes were
tachyarrhythmias, but 18 episodes were described as asystoles not
treated by the WCD.
The actually enrolling WED-HED study (www.clinicaltrials.
gov, NCT02481206) is a multi-center, prospective, randomized
controlled clinical trial with 1:1 assignment of treatment and control.
It will evaluate the impact of WCD use on sudden cardiac death in
incident hemodialysis patients.
Risks and Complications of WCD Wearing
The WEARIT/BIROAD registry reported two patients with
unsuccessful defibrillation due to incorrectly placed therapy
electrodes.18 Accurate and thorough training and instruction of the
patients therefore seems mandatory. The effectiveness and reliability
of the WCD mainly depends on the patient’s compliance.
In the WEARIT/BIROAD registry 65 out of 289 patients
discontinued WCD use before prematurely, 30% did so due to
comfort or lifestyle issues.18
LaPage et al. reported one case of missensing a fatal ventricular
arrhythmia in a patient with unipolar pacemaker stimulation.46
Unipolar stimulation should be avoided in patients wearing the
WCD.
Sudden cardiac arrest may be due to asystole in some patients.
Chung et al. reported 23 patients showing asystole events, 17 of which
died.47 Asystole is a relevant cause of SCD in patients with reduced
LV function48 and to date, there are no data about the relevance of
backup pacing on mortality benefit in ICD patients.
Since the WCD is not able to provide pacing for bradyarrhythmia,
asystole may lead to SCD even though the patient is wearing the
WCD. Nevertheless, an asystole event will trigger the alarm cascade
and may call possible bystander’s attention to the patient.
Inappropriate therapies are another concern during WCD use.
They are reported occurring in 0.5-3% of the patients.24,32 Like in
implanted devices, there is the possibility of T wave oversensing. If the patient fails to press the response buttons, inadequate shock will
be delivered (Figure 2). Since the WCD is programmed to deliver a
“synchronized” shock in case of regular VT, a “synchronized” shock
triggered by an oversensed T wave may happen with a high risk
of induction of VF. This underlines again the need for a dedicated
training of the patient in handling the WCD.
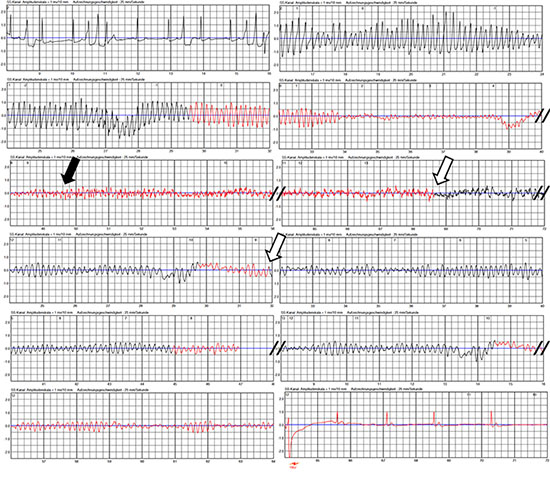
Figure 2 ECG shows normal sinus rhythm with T wave oversensing. The patient did not press the response buttons and therefore received an inappropriate therapy. Fortunately, the shock was triggered to the true R-wave
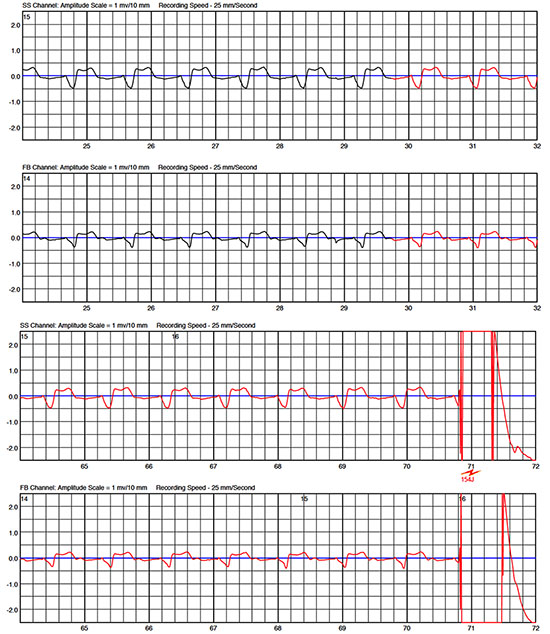
One of our patients presented a hemodynamic instable VT and fell
unconscious. VT was detected and shocked by the WCD. Immediately
after WCD shock, the patient developed irregular ventricular
rhythm. This rhythm was inappropriately classified as a ventricular
tachyarrhythmia by the WCD. Since the patient was still not fully
conscious, he failed to press the response buttons and therefore
received a second shock, which was inappropriate (Figure 3). Besides
the psychic and painful consequences, inadequate therapies bear the
risk for proarrhythmogenity by triggering malignant arrhythmias.
The concept of pressing the response buttons gives relevant safety,
but however there are possible scenarios in which the patient may not
be capable to withhold therapies, as shown in our case.
Figure 3 Episode of a patient with dilative cardiomyopathy. Patient fell on the floor and remained unconscious. ECG shows ventricular tachycardia that was adequately detected and WCD shock was delivered. After WCD shock, the patient remained unconscious and developed irregular ventricular rhythm going along with some noising leading to oversensing and delivery of another – inappropriate – WCD shock
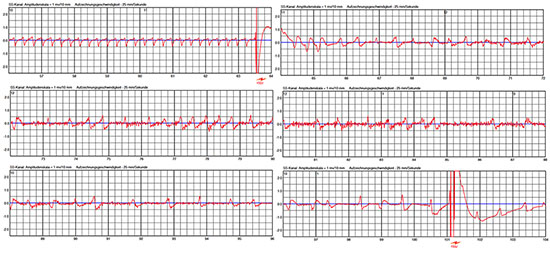
Undersensing due to low amplitudes during VF is a major concern
in ICD. Low amplitudes in VF may even more occur in surface
ECG. In one patient, we noticed VF undersensing due to very low
amplitudes during VF (Figure 4).
Figure 4 Spontaneous ventricular fibrillation (VF) occurred in this patient with peripartum cardiomyopathy. Amplitude of VF rapidly decreased followed by myopotentials (black arrow) which were possibly linked to hypoxia-related seizures. Due to this high-frequency lowamplitude signals, VF criteria were no longer satisfied and detection was cancelled (white arrows). VF was redetected several more times. Probably due to some increase in VF amplitude, VF was finally redetected and the life-saving shock was successfully applied 80 seconds after onset of the tachycardia
The LifeVest Network® (Zoll, Pittsburgh, PA, USA) permits
surveillance of WCD patients via remote monitoring. As a
result of increasing patient numbers being considered for WCD
wearing, the process of screening, prescription, training and remote
monitoring of the patients becomes more and more time consuming.
Increasing patient numbers require structured and optimized
patient management strategies to assure both, reliable logistics and
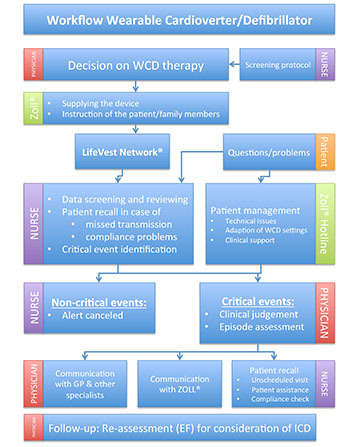
adequate handling of critical events. In our department, we have
established a workflow determining responsibilities and sequences
in the course of a WCD wearing (Figure 5). Screening possible
patients for WCD wearing is performed by a trained nurse using a
screening protocol considering the indications listed in table 1. The
decision for WCD wearing is then taken by a physician taking into
account device-related issues (efficacy and safety), patient-related
factors (compliance, aptitude, acceptance), and disease-related aspects (indications and survival benefit). Cautious instruction of
the patient (and, if feasible, family members) is performed by the
manufacturer when supplying the device. WCD data (arrhythmia
events, compliance, etc.) is available through the telemonitoring
system. Any event reported in LifeVest-Network® is verified by the
responsible nurse and submitted for further review to the physician,
if classified as “critical event”. “Critical events” were defined as: (1)
any therapy delivery, (2) any sustained or non-sustained tachycardia,
(3) any abnormality in the ECG not convincingly attributed to noise.
In order to identify patients at nontransient, enduring risk for SCD,
a careful follow-up for re-evaluation of LVEF is scheduled after 3
months. We use this standardized workflow to facilitate and optimize
patient management in clinical routine.
Figure 5 Workflow of the Wearable Cardioverter/Defibrillator (WCD) program at Hannover Medical School (GP: general practitioner, EF: left ventricular ejection fraction)
The WCD also offers new diagnostic options that may be used
in future versions. The exceptional chance of this device consists in
a continuous ECG monitor for 3 to 6 months. It already detects
asystole events (without giving any therapeutic options), but it could
just as well detect other arrhythmias. By detecting asymptomatic
atrial fibrillation, the WCD could enable stroke prevention at an
early stage. Continuous analysis of the ECG during this long period
in high-risk or assumingly high-risk patients has never been done
before. Additionally, the device can offer supplemental parameters,
such as heart failure indices or tests, which may be relevant in some
patients. Technicians and developer of the manufacturer should yield
this hoard to discover new unprecedented insights in rhythmic and
arrhythmic evolution in these patients. This tool offers completely
new options for future ECG risk stratification.
WCD use is effective and adequate for temporary prevention of
sudden arrhythmic death in chosen populations. In particular, it
provides secured time for sophisticated risk stratification to identify
patients at persistent risk for SCD. Nevertheless, prospective
randomized trials seem mandatory to prove a prognostic effect/
relevance and the economic value of this device.