Excellent symptom rhythm correlation in patients with palpitations using a novel Smartphone based event recorder
William George Newhama,b, Muzahir Hassan Tayebjeea
aLeeds Teaching Hospitals NHS Trust.bThe University of Leeds.
Definitive diagnosis of arrhythmia relies on “symptom-rhythm correlation” when electrocardiographic (ECG) evidence of the patient’s cardiac rhythm is obtained at the time of symptoms. The AliveCor smartphone App and device (AliveCor Inc, California, USA) has recently been introduced as an easy to use cardiac event recorder. The aim of this study was to investigate whether the smartphone based event recorder could be effectively used to achieve symptom rhythm correlation in unselected patients with palpitations.
20 patients (13 female, mean age 35±16 years) underwent evaluation of their palpitations for 12 weeks using 2nd generation AliveCor monitors.
Symptom rhythm correlation was achieved in 85% of patients with 45% detecting an arrhythmia. Of a total of 966 ECGs available for review 96% were interpretable.
The novel smartphone based event recorder is an efficient tool for achieving symptom rhythm correlation in patients with palpitations. By utilising their Smartphone, ECG recording is easily and readily accessible to patients when palpitations occur.
Key Words : Palpitations, Arrhythmia, Smartphone ECG, AliveCor.
Correspondence to: Dr Muzahir Hassan Tayebjee (Consultant in Cardiology and Cardiac Electrophysiology) MBChB (hons), MD, MRCP (UK) Department of Cardiology Ex Ward 39, Main Site Leeds General Infirmary Leeds West Yorkshire United Kingdom LS1 3EX Email:muzahir.tayebjee@nhs.net Tel:+44 113 3926619
The definition of palpitations is subjective and patients frequently complain of this symptom in normal sinus rhythm. Furthermore, patients often report symptoms that are transient or short lived in nature and therefore difficult to diagnose. In such patients, ambulatory ECG (AECG) monitors are commonly utilized.[1]-[3]
Recently a new AECG device has been developed which allows the user to record a single lead ECG on their smartphone. The AliveCor monitor incorporates two electrodes and attaches directly to the back of the user’s smartphone or its case. A 30 second real-time ECG equivalent to Lead I on a 12-Lead ECG is recorded by activating the App and placing at least one finger from each hand across the electrodes. The user then has the opportunity to annotate the recorded ECG with symptoms. Patients' personal data and recorded ECGs are stored on a secure server and accessed online by patient or healthcare professional via AliveCor's website.
Use of the AliveCor monitor in the ambulatory setting for patients with a primary complaint of palpitations has been evidenced in selected groups only; such as those with atrial fibrillation post ablation and in paediatric patients with documented paroxysmal arrhythmias.[4],[5] Although complementary of the device, further reports of its utility as an AECG device for patients with palpitations are largely anecdotal; reporting case studies and identifying potential benefits of the new technology in the larger population.[6]-[10] Given this potential we investigated whether the AliveCor monitor could be effectively used to achieve symptom rhythm correlation in an unselected group of patients with a primary complaint of palpitations.
Over a 3 week period, 23 patients with palpitations were referred from cardiology outpatient clinics for AliveCor monitoring. Patients were excluded if their palpitations were associated with syncope, they had an implantable cardiac device already in-situ, did not own a compatible smartphone, were unable to consent to us accessing their recorded ECGs, or were unable to use the monitor effectively.
Patients attended an outpatient AECG appointment where they received their monitor. Informed consent of the subjects for monitoring was obtained. Under instruction from the cardiac physiologist patients downloaded the ‘AliveECG’ App to their Smartphone and set up a ‘personal account’. All patients agreed to AliveCor's data protection policy, with personal data stored on AliveCor's secure server. The patient was required to grant us access to their account in order to view recorded ECGs. This was done by accepting an invitation email sent from the department’s ‘healthcare provider account’ accessed via the AliveCor website. The monitor was then attached to the patient's smartphone and the process of recording an ECG and annotating with symptoms explained to the patient, who was then required to record a test ECG confirming they were able to use the monitor effectively. Patients were instructed to record a 30 second ECG when symptomatic, annotate with ‘palpitations’ and or any other symptoms. Patients were instructed to annotate any ECGs made when asymptomatic as ‘test ECG’ and only use the device themselves. Patients were invited to fill in two short questionnaires, one at set-up and one when they returned the monitor, with questions structured to gain insight into ease of use of the monitor and App.
Monitoring was conducted over 12 weeks with uploads reviewed once a week by a cardiac physiologist. Each ECG recording was categorised as one of the following: sinus rhythm, sinus bradycardia, sinus tachycardia, ventricular ectopics, supraventricular ectopics, ventricular bigeminy/trigeminy, broad complex tachycardia, narrow complex tachycardia, atrial fibrillation, atrial flutter, high grade AV block, unusable ECG or test ECG.
Symptom rhythm correlation was expressed as a percentage of patients reporting palpitations during a recording with successful capture of ECG rhythm. Detection of arrhythmia was expressed as a percentage of patients reporting palpitations during a recording with successful capture of ECG rhythm with findings other than sinus rhythm, bradycardia or tachycardia only. ECG quality was expressed as a percentage of total recorded ECGs where the cardiac rhythm could be identified. Recorded ECGs were classed as 'unusable' simply when the rhythm could not be identified due to poor quality baseline and or presence of artifact. Variables are expressed as mean±standard deviation and data was stored on a password-protected trust computer using Microsoft Excel. The study complied with the Declaration of Helsinki and the research protocol was approved by the local Research and Innovation Department as a service evaluation project.
Twenty patients [Table 1]received an AliveCor monitor. During the 12 weeks, 19 patients (95%) recorded at least 1 ECG, 1 patient (5%) did not record any ECGs. 6 patients (30%) were fully compliant with the instructions given at set-up. Three patients (15%) deleted uploads, 13 patients (65%) uploaded a proportion of their ECGs without annotating with symptoms; these accounted for 66% (n=639) of the total ECGs available to review. At follow-up 11 of these patients reported that they were in fact symptomatic and had failed to consistently annotate with symptoms as instructed at set-up (203 (32%) of the blank uploads). These ECGs were re-classified as symptomatic with palpitations. The remaining 68% of blank ECGs (n=436) were uploaded by 2 patients whilst asymptomatic. These uploads were subsequently re-classified as test ECGs.
Table 1. Patient baseline clinical characteristics
| Characteristic |
Values |
|
|
| Number of patients |
20 |
| Age (years) |
35±16 (range12-64) |
| Female |
13 (65%) |
| Male |
7 (35%) |
|
|
| Baseline symptoms |
|
| Palpitations |
20 (100%) |
| Pre-syncope |
6 (30%) |
| Fatigue |
5 (25%) |
| Breathlessness |
4 (20%) |
| Chest Pain |
1 (5%) |
| Hypertension |
1 (5%) |
| CAD |
1 (5%) |
| WPW |
1 (5%) |
| Previous EPS |
6 (30%) |
|
|
| History of palpitations (years) |
2.6±3.5 (range 0.25-13) |
| Frequency of palpitations |
|
| Daily |
3 (15%) |
| Weekly |
8 (40%) |
| >Weekly |
6 (30%) |
| Monthly |
1 (5%) |
| >Monthly |
2 (10%) |
|
|
|
|
CAD - Coronary artery disease, WPW - Wolf Parkinson White, EPS - Electrophysiology study*Continuous data presented as mean ± SD and range (in parenthesis), Categorical data as number of patients (percentage of sample in parenthesis).
A total of 1145 uploads were made during the 12 week monitoring period. 179 uploads did not have stored ECGs, as patients (n=3) had deleted them from their personal account. 42 (4%) were deemed unusable as the ECG was of poor quality, 462 (48%) were test ECGs and 157 (16%) were recorded with symptoms not including, or other than palpitations. 7 (<1%) were uploads where patients (n=4) documented their palpitations had subsided before recording the ECG. 340 uploads were recorded by patients when symptomatic with palpitations of which 37 (11%) were unusable. A sample of recorded ECGs uploaded with the AliveCor monitor is shown in [Figure 1].
Patients achieving symptom rhythm correlation was found to be 85% with 17 patients recording at least 1 ECG whilst reporting palpitations. For detection of arrhythmia 9 patients (45%) recorded at least 1 ECG while reporting palpitations with findings other than sinus rhythm, bradycardia or tachycardia only. Identification of cardiac rhythm was possible in 96% of the total ECGs uploaded (922/966) and in 89% of symptomatic uploads (303/340). ECG recordings deemed unusable were through poor quality baseline and interference, examples of which are displayed in [Figure 1] (ECGs i and j). The irregular 'spiked' interference shown in [Figure 1] (ECG - j) was present in 3 patients' recordings.
Figure 1. Example ECG rhythms uploaded with the AliveCor heart monitor
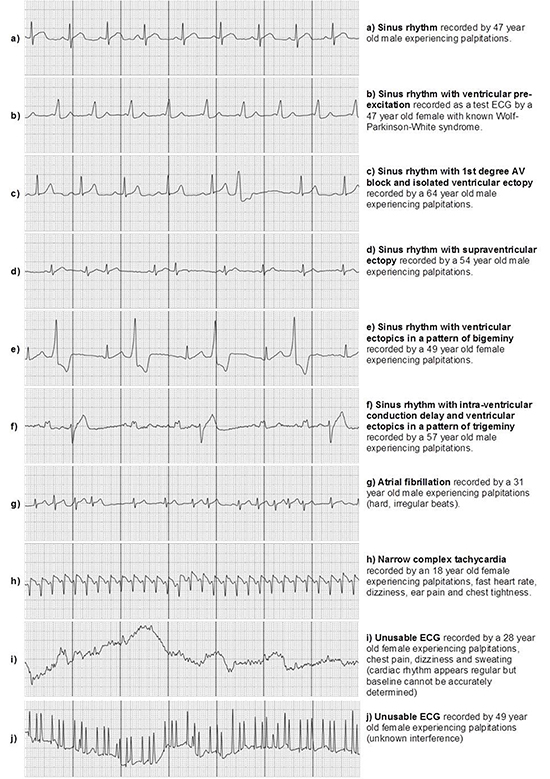
20 patients (100%) completed the questionnaire at set-up and 17 (85%) at follow-up. 18 patients (90%) reported the process of finding and downloading the AliveCor App as very easy/easy, while 2 patients (10%) found it difficult. 20 patients (100%) found the instructions given at set-up very easy/easy to follow. 16 (94%) found using the monitor very easy/easy. 3 patients (18%) reported problems using the monitor (n=1 encountered the unknown interference displayed in [Figure 1] (ECG - j) on a number of their ECGs causing distress as the interference resulted in an inaccurate very high heart rate reading, n=2 felt they were unable to activate device in time to record ECG during short lived palpitations).
This study demonstrated that the smartphone based event recorder used under medical supervision can be effectively used to record good quality ECG and achieve symptom rhythm correlation in patients with palpitations. In contrast with the historically reported poor quality ECG recordings of conventional event recorders,[1] in this study the overall quality of ECG recordings was good and consistent with other studies in the ambulatory setting reporting between 87% and 99% of ECGs recorded with the AHM as being interpretable.[5],[6] Other studies where the AliveCor monitor was used as a screening tool have reported similar high quality ECG recordings.[12]-[15]
Patient training by the cardiac physiologist is likely to have increased the overall quality of recorded ECGs than if patients had purchased and used the device independently. Even under these circumstances, complete adherence was modest with many patients not consistently annotating the ECGs with symptoms. Furthermore, some patients elected to record multiple recordings without symptoms. This can in future be dealt with by better patient education when prescribing the device. In addition, short lived palpitations may not be recorded as the arrhythmia may terminate before activation. This is a commonly reported limitation of non-looping event recorders.[3]
At £74.99 unit cost of the 2nd generation AliveCor monitor (2015) is cheaper than most other conventional forms of AECG monitoring, especially implantable loop recorders and could even be a single use device. Another potential cost saving is the online ECG reviewing system which is free to use. Conventional forms of AECG monitoring often require costly analysis systems with associated maintenance costs.[10] Furthermore, current conventional patient activated devices are often limited to 3-4 weeks of monitoring as well as requiring continued maintenance and battery changes.[1] In contrast maintenance of the AliveCor monitor is not required and battery life is reported at 12 months with typical use. This could allow for external (non-invasive) monitoring over long periods of time free of continued device up-keep, previously unavailable without use of implantable loop recorders which are both costly and have associated risks of an invasive procedure.[3]
With conventional patient activated devices there is evidence that if symptom rhythm correlation is not achieved within the first 2 weeks of monitoring, then further diagnostic yield will be low.[11] A potential reason is that patients lose interest and do not carry their monitor.[8] This problem is obviated with the AliveCor monitor as it is attached to the patient's smartphone. Interestingly, Ofcom (The independent regulator and competition authority for the UK communications industries) recently branded the UK a "smartphone society" with an estimated 66% of adults owning a smartphone in 2015.[16] Together with the fact that the 3rd generation monitor has extended compatibility, this monitor is likely to be suitable for a large number of people.
The novel smartphone based event recorder is an efficient tool for achieving symptom rhythm correlation in patients with palpitations. By utilizing their Smartphone, ECG recording is easily and readily accessible to patients when palpitations occur. In order to ensure appropriate use and quality of recorded ECGs, use of these devices under the supervision of a healthcare professional is advised.
Biosense Webster (research grant and travel grant), St Jude Medical (research grant and travel grant), Medtronic (research grant, travel grant and proctorship), Boehringer Ingelheim (research grant).