HIGH VOLTAGE GUIDED PULMONARY VEIN ISOLATION IN PAROXYSMAL ATRIAL FIBRILLATION
Usama Boles1, Enes E Gul1, Andres Enriquez1, Howard Lee1, Dave Riegert2, Adrian Andres1, Adrian Baranchuk1, Damian Redfearn1, Benedict Glover1, Chris Simpson1, Hoshiar Abdollah1, Kevin Michael1
1Division of Cardiology, Heart Rhythm Service, Kingston General Hospital, Queen’s University, Kingston, Ontario, Canada..2Department of Mathematics and Statistics, Queen's University, Kingston, Ontario, Canada..
Ablation of the pulmonary vein (PV) antrum using an electroanatomic mapping system is standard of care for point-by-point pulmonary vein isolation (PVI). Focused ablation at critical areas is more likely to achieve intra-procedural PV isolation and decrease the likelihood for reconnection and recurrence of atrial fibrillation (AF). Therefore this prospective pilot study is to investigate the short-term outcome of a voltage-guided circumferential PV ablation (CPVA) strategy.
We recruited patients with a history of paroxysmal atrial fibrillation (AF). The EnSite NavX system (St. Jude Medical, St Paul, Minnesota, USA) was employed to construct a three-dimensional geometry of the left atrium (LA) and voltage map. CPVA was performed; with radiofrequency (RF) targeting sites of highest voltage first in a sequential clockwise fashion then followed by complete the gaps in circumferential ablation. Acute and short-term outcomes were compared to a control group undergoing conventional standard CPVA using the same 3D system. Follow-up was scheduled at 3, 6 and 12 months.
Thirty-four paroxysmal AF patients with a mean age of 40 years were included. Fourteen patients (8 male) underwent voltage mapping and 20 patients underwent empirical, non-voltage guided standard CPVA. A mean of 54 ± 12 points per PV antrum were recorded. Mean voltage for right and left PVs antra were 1.7±0.1 mV and 1.9±0.2 mV, respectively. There was a trend towards reduced radiofrequency time (40.9±17.4 vs. 48.1±15.5 mins; p=0.22).
Voltage-guided CPVA is a promising strategy in targeting critical points for PV isolation with a lower trend of AF recurrence compared with a standard CPVA in short-term period. Extended studies to confirm these findings are warranted.
Key Words : Circumferential pulmonary vein isolation (CPVA), voltage-guided ablation, radiofrequency ablation and AF ablation.
Correspondence to: Corresponding author: Dr Usama Boles
Consultant Cardiologist and Electrophysiologist,
Department of Cardiology,
Midland Regional Hospital,
Mullingar, Ireland,Email: bolesu@tcd.ie
Catheter ablation is an effective therapy for patients with symptomatic, drug-refractory atrial fibrillation (AF) [1]-[3]. The pulmonary veins (PV) play a major role in the initiation of AF [4] and pulmonary vein isolation (PVI) constitutes the cornerstone for ablative treatment of paroxysmal and persistent AF [5].
Histological studies demonstrate that the LA (left atrium)-PV junction is composed by discrete myocardial sleeves that extend from the LA into the PV, separated by gaps of connective tissue. Automated electro-anatomic mapping of cardiac tissue has been used to identify low voltage regions, which have been shown to be a surrogate marker of potential scar tissue [6]. Therefore, areas of high voltage are likely to identify areas of viable myocardium and hence, can be used to define putative muscular connections from PV antra into the pulmonary veins themselves. Scar or adventitial tissue would therefore be represented by relatively low voltage areas in the intervening regions [6], [7]. We hypothesize that an ablation strategy targeting regions of high voltage in PV antra could improve the outcomes as this allows for focused ablation on critical areas of muscular connections. This should also translate into a lower risk of reconnection and therefore less chance of early recurrence of AF.
In this single-centred prospective pilot study we enrolled patients undergoing a first catheter ablation for paroxysmal AF. Inclusion criteria were as follow: (i) age > 18 years; (ii) history of AF with documentation on 12-lead ECG, Holter or event monitor; (iv) refractoriness to at least one class I and/or III antiarrhythmic drug. Exclusion criteria were: (i) previous ablation for AF; (ii) intracardiac thrombus; (iii) contraindication to oral anticoagulants and (iv) unwillingness to participate in the study. Also there were no ‘’lone AF’’ patients in our study group.
Anti-arrhythmic medications (except Amiodarone for a minimum of 4 weeks) were discontinued for 5 half-lives prior to the procedure and all patients provided written informed consent.
The procedure was conducted under conscious sedation with intravenous fentanyl and midazolam. An initial 12-lead ECG was performed to confirm sinus rhythm. Venous access was gained from the femoral veins. A deflectable decapolar catheter (Inquiry, St. Jude Medical, St. Paul, USA) advanced to the coronary sinus, a quadriapolar catheter (St. Jude Medical, St. Paul, USA) was placed at the right ventricular apex. Transseptal access was performed using a standard technique with a BRK Brockenbrough needle and SL1 sheath (St. Jude Medical, St. Paul, USA). Following this, a spiral multipolar PV catheter (AF Focus II, St. Jude Medical, St. Paul) and an open irrigated ablation Coolflex catheter (St. Jude Medical, St. Paul, USA) were used to perform the ablation.
EnSite NavXTM system (St. Jude Medical, St. Paul, USA) was used to construct the LA geometry and a voltage map of the antral region of the 4 PVs using a segmented cardiac CT template with/out fusion of the images.
Periprocedural Anticoagulation
Patient’s anticoagulated with warfarin the procedure was performed without interruption therapy with an INR level between 2 and 3. Patients on direct oral anticoagulants (DOACS) were instructed to withhold the doses for 48 hours prior to the procedure. After establishing LA access, intravenous unfractionated heparin was administered at 10 to 20 minute intervals to attain a target-activated clotting time of 300-350 seconds.
Voltage mapping and anatomical identification
Bipolar voltages of both the right and left antra were recorded during continuous pacing from distal coronary sinus poles using a spiral-mapping catheter (AF Focus II, St Jude Medical, St. Paul, USA).
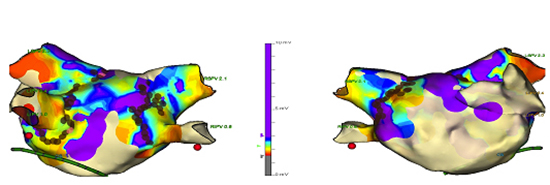
The voltage map was created and the readings were scaled. A voltage of 0.5 to 1.5 mV was considered to be viable normal myocardium and < 0.5 mV was considered to be scar or adventitia.[8] Voltages >= 1.5 mV were regarded as high voltage regions ([Fig 1]). Anatomical identification of high voltage points and areas were studied against the critical points and located in main 4 quarters (superior, inferior, anterior and posterior).
Critical points were identified as isolation or delay of PVPs.
Figure 1. This depicts voltage map in left atrium and corresponding antral RF ablation (brown dots). Both of posterior and anterior views of LA are demonstrated. The voltage map was set at 0.5 – 1.5 mV. The middle column illustrates the voltages ranges between 0 mV and 10 mV, coloured region represents areas ≥ 1.5 mV.
We aimed to compare 20 study patients with 20 controls. However, 6 patients from the study group were excluded from the study because -they were in AF at the time of procedure - to homogenize the voltage map techniques during pacing from the proximal coronary sinus catheter at CL of 600 ms. This was there as a consistent reference point allowing for a legitimate comparison of voltage points acquired in each antrum. If sampled in AF, oscillating and meandering rotors through the PV-LA junctions may have given an erratic activation pattern making it difficult to compare adjacent regions of varying voltages.
Radiofrequency (RF) energy was applied in the antral regions of the PVs, starting at the site of maximum voltage. Initially, we aimed high voltage areas because of fear of tissue oedema around the vein. When elimination of local electrograms was achieved, the adjacent remaining sites of maximum voltage were targeted in a sequential fashion (clockwise circumferential pattern) until isolation of the vein was achieved. If no isolation was achieved, the strategy of contiguous lesion application that is with completion of a full and complete circumferential ablation in the antrum with additional ablation guided by earliest activation on the PV spiral catheter was performed. The definition of adequate lesion was as follows: Under adequate power (30- 35 W) and irrigation with saline at a rate of 17- 25 ml/min, individual RF delivery was applied for approximately 40 seconds until the elimination of the atrial potential. In addition to that, signal resolution as Q formation or S>R was an end point to indicate successful lesion [9]. An impedance drop of 15 ohms is another confirmation of effective lesion in our study [10]. After ablation, the catheter was moved to the next site along the line. This was continued until ablation of the standard set of lesions was complete. Primary end point was considered as entry and exit block in all PVs. RF was delivered using a 4 mm externally irrigated-tip ablation catheter (Cool Flex 7F, St Jude Medical, St Paul, MN).
No additional linear ablation was performed in this group of patients with paroxysmal AF. Similar settings and techniques were employed in control group with standard circumferential PVI without voltage mapping. Acute reconnection was assessed in both groups at the end of the procedure and was defined if any gap (PV signals) were found after adenosine administration.
Oral anticoagulation was reinitiated 8 hours post-procedure if no bleeding complications (i.e. pericardial tamponed or access site significant haematomas) were noted. All patients were kept on either a class I or class III antiarrhythmic drug with/out a beta-blocker or calcium channel blocker and an oral anticoagulant (warfarin or DOAC) for at least 3 months post procedure.
First follow-up occurred at 12 weeks post PVI and subsequent visits at 6 month and 12 months. All patient had a 24-hour holter and 12 lead ECG at follow-up. Secondary end point is recognised as AF recurrence more than 30 seconds or atrial arrhythmia documented in ECG or holter monitor. We opted to exclude 6 patients From Voltage guided ablation arm (out of 20 patients) that were in AF on the day of the procedure.
Statistical analysis was performed with R version 3.2.1 (R foundation, Vienna University of Economics and Business, Austria). Data were expressed as mean ± standard deviation (SD) for continuous variables, and frequencies and percentages for categorical variables. T-tests (continuous variables) and chi-square tests (categorical variables) were used for statistical analysis. The logistic model for anatomical location of point-to-point analysis was used. Kaplan – Meyer cumulative rate occurrence of AF recurrence over the follow up period was calculated. The statistical significance results were considered if p <0.05.
Characteristics of patients
Baseline characteristics of patients and control group are depicted in [Table 1]. No differences in demographics or echocardiographic parameters were identified. Radiofrequency time, fluoroscopy time and total procedure time were not statistically different in both groups ([Table 1]).
Table 1. Study demographics of Voltage guided and control cohorts. Data is presented as a mean ± SD. Pre LVEF = pre procedure left ventricular ejection fraction. LA= left atrium. RF time = radiofrequency ablation time. BSA = body surface area.
|
|
Voltage-guided PVI (n=14)
|
Standard PVI group (n=20)
|
P value
|
|
Age (years)
|
59.8 ± 16
|
61.8 ± 11.8
|
0.66
|
|
Weight (Kg)
|
84.8 ± 17.6
|
89.2 ± 25.9
|
0.57
|
|
Height (cm)
|
166.8 ± 22.1
|
173.6 ± 9.6
|
0.29
|
|
BSA
|
2.0 ± 0.3
|
2.0 ± 0.3
|
0.93
|
|
Pre LV EF (%)
|
54.1 ± 9.6
|
55.2 ± 5.6
|
0.70
|
|
LA Diameter (mm)
|
40.2 ± 7.6
|
36.8 ± 4.2
|
0.36
|
|
LA Volume (ml3 )
|
66.2 ± 22
|
63.3 ± 13.6
|
0.65
|
|
Procedure Time (mins)
|
248.9 ± 53.7
|
233.9 ± 36.1
|
0.32
|
|
Fluoroscopy Time (mins)
|
33.6 ± 17.7
|
28.7 ± 8.9
|
0.36
|
|
RF Time (mins)
|
40.9 ± 17.4
|
48.1 ± 15.6
|
0.22
|
|
Initial Diagnosis (months)
|
36.8 ± 26.4
|
29.9 ± 25.8
|
0.46
|
|
Recurrence, n (%)
|
3/13 (23 %)
|
7/18 (39 %)
|
0.41
|
|
Acute reconnection, n (%)
|
3 (20)
|
7 (35)
|
|
|
Reconnected veins
|
|
|
|
|
LSPV, n
|
2
|
2
|
|
|
LIPV, n
|
0
|
1
|
|
|
RSPV, n
|
1
|
3
|
|
|
RIPV, n
|
1
|
0
|
|
Relationship of high voltage areas and critical points
Voltage point’s distribution and their frequency around each antrum are illustrated in [table 2]. The 50th quartile points correspond with voltages at 0.86 mV. The number and percentages of the points (to total points) at 10th, 25th, 50th, 75th and 90th percentiles are also documented. Total critical points identified were 526 points where, 369 (70.2 %) critical points were identified at 75th quartile or higher. On the other hand, no critical points were recognised in low voltages areas below the 25th quartile. ([Table 2])
These points were confirmed at the boundaries of the HV regions when the logistic model was applied (P = 0.06 at 1.5 mV).
Table 2. This demonstrates the number of voltages points and their percentages at 10th, 25th, 50th, 75th and 90th quartiles. The critical points and their percentages (in comparison to the total critical points)
|
|
Total points
|
Voltages at 10th Quartile
(≤ 10th)
|
Voltages at 25th Quartile
(10th to 25th)
|
Voltages at 50th quartile
(25th to 75th)
|
Voltages at 75th Quartile
(75th to 90th)
|
Voltages at 90th Quartile
(≥ 90th)
|
|
Voltages values (mV)
|
NA
|
0.12
|
0.29
|
0.86
|
2.2
|
3.98
|
|
Voltage Points (N)
|
1479
|
145
|
361
|
458
|
369
|
146
|
|
Percentages of voltage
points (%)
|
100
|
9.8
|
24.4
|
30.9
|
24.9
|
9.9
|
|
Number of Critical points ≥ (n, %)
|
526
|
0
|
0
|
157, (29.8%)
|
223, (42.4%)
|
146, (27.8 %)
|
Follow-up and AF recurrence
The mean follow-up data was 46.4 ± 13.7 weeks. One patient in the voltage-guided group and 2 in the control group were lost to follow-up.
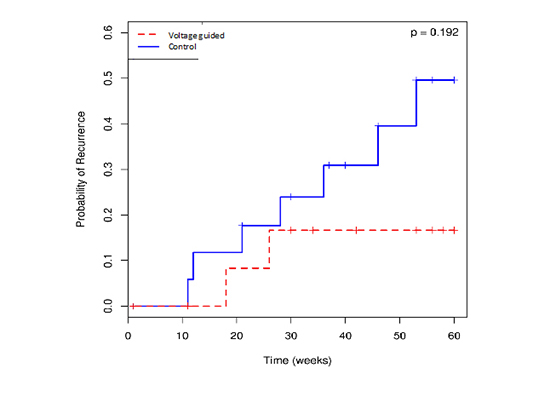
Cumulative recurrence rate using Kaplan-Meier curve showed that voltage guided ablation had a trend of lower recurrence than patients in the standard ablation group; 3/13 (23 %) vs. 7/18 (39 %), respectively ([Figure 2]).
Figure 2. Kaplan – Meyer cumulative rate occurrence of AF recurrence over the follow up period in weeks. Lower rate is observed in voltage guided CPVA group.
Anatomical frequency of critical points
The antrum of each PV was divided in 4 anatomical regions in a clockwise fashion: superior, inferior, anterior and posterior.
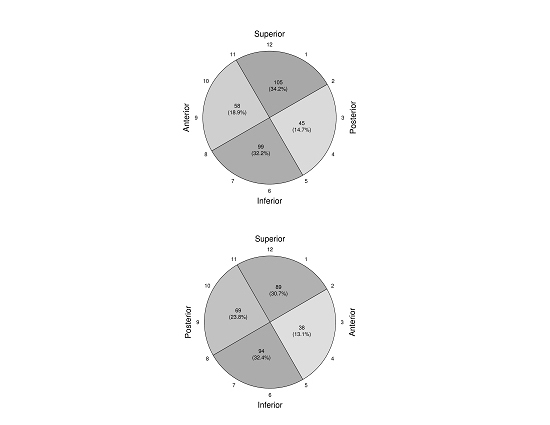
As shown in ([Figure 2]), the superior and inferior segments were more likely to harbour critically high voltage points.
Figure 3. this diagram depicts the frequency (%) of the critical points (sites of the isolation and delay of pulmonary vein potentials) around antral regions: A) left pulmonary vein antrum, B) right pulmonary vein antrum
This prospective study presented a new approach to identify critical areas of PV and antral connections in order to achieve superior results for PV isolation. Antral high voltage guided ablation, in clockwise fashion, corresponded with critical points and revealed lower trends for AF recurrence compared to standard CPVA procedure over a follow up period of one year.
Rationale of the technique
In our cohort, we targeted antrum ablation, which previously proved superior outcome to ostial and intra-vein ablation, and carries a better long-term prognosis [11]. However, anatomically, the earliest activation seen on the spiral catheter during atrial pacing or sinus rhythm may not necessarily correspond with the optimal ablation site in the antrum, given that the muscle sleeves entering into each vein, may follow an often unpredictable course.[12] Significant voltage variation is also recorded at various extent proximal to the PV ostium at the level of the antral region[13]. A thickened PV-LA junction wall is a predictor of late recurrence and dormant conduction and hence these regions may represent critical areas for LA/PV conduction [14] Therefore, antral ablation to achieve PV isolation is challenging. Thus far ablation is essentially done empirically in a continuous anatomic fashion to encircle each pair of PV.
Though, over the last few decades, various approaches were implemented to improve PVI outcome. Of these defragmentation of complex fractionated atrial electrograms (CAFÉ), mainly in persistent AF, was proposed by Nademanee et al [15] [16]. However, the success of that approach comes at the price of possible iatrogenic macro-reentrant flutter [17] [18] as well as the risk of thermal esophageal injury.[19] Nevertheless, antral isolation is evidently effective to treat patients with paroxysmal AF when compared with CAFÉ defragmentation. [20] Interestingly, complex fractionations in normal atrial voltages may only represent the response of healthy atrial tissue to rapid pulmonary veins activation rather than a substrate to AF.[21]
Recovery of conduction in previously ablated muscle fascicles in the antrum is a common finding in patients with recurrent AF [22]. Circumferential ablation primarily achieves PV-left atrial block but also serves to reduce atrial ectopy decreasing the risk of AF recurrence.[23]
In our study, we proved most of the critical points (70.2 %) were located in the 75th quartile of voltage value or higher. In another word, most of the critical points were identified in higher voltage antral region and strikingly no critical points seen in voltages lower than the 25th quartile.
In our approach, by identifying sites of critical connection in high voltages areas in each antrum, the operator can focus ablation in terms of adequate power delivery, impedance changes as well as diminishing signal amplitude during the ablation. This will also avoid superfluous antral lesions that may raise significant edema preventing effective RF delivery to critical regions.
Our technique showed a trend to a reduction in the procedure time and radiofrequency time. This can be explained by the strategy of eventually completing a circumferential ablation set os lesions in each antrum, in the voltage guided group even after the PV isolation. This was to ensure that the staggered ablation lesions sets do not become the pro arrhythmic substrate for macro-re entrant tachycardia in this pilot group.
Although we eventually accomplished a complete CPVA lesion set in each antrum, the identification and prioritized ablation of HV critical regions made it possible to deliver effective transmural lesions at these sites before any reactionary oedema due to RF was invoked. This formed the basis of our strategy.
Anatomically, wide areas of interspersed connective tissue between the myocardial muscle fibers characterize myocardial sleeves extending into the PVs; this heterogeneous composition of the PV-LA junction is also reflected in their varied refractory periods. Then presents a milieu for micro-re entry as wave fronts of activation from triggers within the PVs encounter the PV-LA interface.[4] An ablation line at this critical junction has the potential to eradicate this critical substrate for re-entry and hence reduce recurrence of AF [7].
Collectively, this mechanistically explains the lower trends for AF recurrence - over almost a year follow up - in the voltage guided PVI group as opposed to unguided anatomical CPVA as was performed in our control cohort.
Some limitations can be noted in our study. First, the sample size was very small, however this pilot study was performed to investigate whether the results are promising. Therefore we think that it is reasonable to perform a larger randomized controlled study to obtain more precise and proper answers. Second, the study was not randomized and the comparison group consisted of a higher number of patients undergoing a standard approach of RF ablation. We elected to exclude those patients who presented in AF “in voltage-guided group” to ensure a constant study protocol. Both groups, however, were matched in terms of clinical characteristics and the primary operators were the same. Second, contact force technology was not used; however this applies to both groups.
A voltage guided CPVA strategy was useful in identifying critical points for PV isolation. Voltage-guided procedures showed lower trends in AF recurrence than anatomical CPVA in this pilot cohort. Further prospective evaluation will enhance our observation in this pilot study.