A Review of the Anatomical and Histological Attributes of the Left Atrial Appendage with Descriptive Pathological Examination of Morphology and Histology
Mark Hensey1, Louisa O’Neill1, Ciara Mahon1, Stephen Keane1, Aurelie Fabre1, David Keane1
1Saint Vincent’s University Hospital, Dublin.
The left atrial appendage (LAA) has a key role in the embolic complications of atrial fibrillation (AF). It has been studied extensively, from recent interest in the thrombotic implications of various LAA morphologies to LAA occlusion and ablation. We collected eleven post-mortem LAA samples for visual analysis, two were not included due to poor sample quality. On examination of the nine remaining samples, several common patterns of pectinate muscle orientation were noted. The LAA samples were noted to have a smooth circumferential neck of muscular tissue giving rise to a dominant singular smooth trunk of papillary muscle in 6 cases and two trunks in 3 cases. These trunks were either shallow (5 samples) or more muscular and raised (4 samples). Shallow trunks tended to be wider than the raised trunks and may even be circumferential (2 samples). The main trunk arborized to give off papillary muscle branches down to third or fourth order branches. The samples were visually assessed for the percentage of smooth papillary muscle versus non-papillary recesses and were found to have ≤50% smooth muscle in 3 samples, 50-75% in 3 samples and >75% in 3 samples. We performed histological analysis of further LAA samples collected during cardiac surgery in a parallel study. We identified a distinct pattern of myocyte orientation from the neck, mid-section and apical section of the LAA demonstrating arborization of myocyte fibers with minimal communication in distal segments of the LAA. We feel that this information may help understanding of the issues surrounding LAA ablation strategies.
Key Words : Left Atrial Appendage (LAA), Chicken Wing, Windsock, Cauliflower.
Correspondence to: Professor David Keane, Department of Cardiology, Saint Vincent's University Hospital, Elm Park, Dublin 4
The left atrial appendage (LAA) has a key role in the formation of thrombus and subsequent embolic complications in atrial fibrillation (AF) and flutter and has been shown to be involved in >90% of transient ischemic attacks (TIA) or strokes related to AF.(1) For a small anatomical structure, it has been studied extensively from the recent interest in the various morphologies of the LAA and their relative risk of thrombosis to LAA closure devices and the success of ablation procedures incorporating LAA isolation. In this paper we aim to review atrial anatomy relevant to the electrophysiologist, review the role of the LAA in AF initiation and propagation and we also provide a description of the macro and microscopic properties of the LAA based on post-mortem studies.
Embryology and anatomy of the left atrium
Embryologically the heart begins development by the formation of two tubes into a single tube in an inverted Y structure. Through folding of this structure, the two arms of the Y ultimately form the right and left atria. The left and right atrial appendages form from the respective left and right supero-lateral walls of the primary atrium. The folded structure is then separated into the four chambers along with the pulmonary artery and aorta by the formation of the endocardial cushion, intra-atrial septum, intra-ventricular septum and the aortic-pulmonary septum. When formed, the LAA is smaller and narrower than the extensive right-sided appendage. The LAA forms trabeculae due to cellular protrusion into the lumen. The development of the four pulmonary veins is a late process.(2,3)
The fully formed left atrium is composed of the postero-superior venous component, the vestibule forming the mitral valve orifice, the LAA and the body of the atrium. The LAA is separated from the left pulmonary veins venous component by the left lateral ridge. This is an infolding of the lateral atrial wall protruding into the LA endocardium.(4) The LAA lies anteriorly in the atrioventricular sulcus in close proximity to the left circumflex artery and the left phrenic nerve.
The LAA itself is divided into the os, the neck and the body; all of which can be variable in morphology. The body of the LAA is divided into a number of lobes which is again variable. The internal surface of the LAA is composed of raised pectinate muscles which results in an uneven surface – in direct contrast to the relatively smooth walls of the rest of the left atrium. This irregular surface with deep recesses likely increases its thrombogenicity.
There has been recent interest in the variable morphologies of the LAA. Veinot et al. examined LAA’s from 500 normal autopsy hearts and found that 80% of specimens had more than one lobe (54% two lobes, 23% 3 lobes, 3% 4 lobes). They also found that 97% had pectinate muscles ≥1mm in size. Both of these are important considerations when examining the LAA with transesophageal echocardiography to rule out thrombus.(5) Di Biase et al used computed tomography (CT) and magnetic resonance imaging (MRI) to characterize the LAA morphology of 932 patients undergoing catheter ablation for AF and correlate this with a history of prior transient ischemic attack (TIA) or stroke. They identified four discrete morphologies – ‘chicken wing’ (48%), ‘cactus’ (30%), ‘windsock’ (19%) and ‘cauliflower’ (3%). Chicken wing morphology was found to have the least likelihood of previous TIA/stroke. In a multivariate model, cactus was 4.08 times, windsock 4.5 times and cauliflower 8.0 times more likely to have been associated with TIA/Stroke than a chicken wing morphology.(6)
The LAA is composed of both endocardial and epicardial components with variable myocyte orientation. The junction between the LA and LAA has a complex fiber orientation and may support anatomical reentry or anchor functional rotors.(7) LAA myocytes are more similar to ventricular myocytes than atrial myocytes in structure. There is variability in thickness between muscle bundles. Bachmann’s bundle runs in a sub-epicardial layer allowing conduction from the right atrial appendage through the inter-atrial septum and encircles the neck of the LAA. Histological changes in those with chronic AF have been noted with expansion of LAA size, loss of pectinate muscle volume and thickening and fibrotic change of the endocardium. Amyloid deposition has also been noted in chronic AF.(3)
There are several mechanisms proposed for the development and propagation of AF and it is unclear whether it is dependent on automatic focal or reentrant mechanisms. What is clear, however, is that the pulmonary veins are crucial sites of AF triggering and they have become the main target for AF ablation. Pulmonary vein isolation (PVI) has become a mainstream therapy for AF with guidelines supporting its use earlier in the disease process. (8) Current outcomes, however, are not ideal with multiple procedures often required and 5 year freedom from AF being reported as ≈80% in paroxysmal AF and ≈45% in permanent AF. (9-12)
Other myocardial sites have also been targeted for additional ablation such as the superior vena cava, ligament of Marshall, posterior left atrial wall, coronary sinus, crista terminalis and the LAA itself. Di Biase et al. have shown that the LAA has been found to be a site of recurrent AF after PVI in 27% of patients with excellent results from subsequent complete isolation of the LAA.(13) Empiric ablation of the LAA along with standard ablation in patients with permanent AF has been investigated in the BELIEF study. Early results have shown improved outcomes with 56% of patients with LAA isolation along with standard ablation being AF free after 12 months as compared to 28% of patients with standard ablation alone.(14)
We collected LAA samples from consecutive post-mortems being carried out in a tertiary referral centre with cooperation from the pathology department. Ethical approval was granted in advance of the study by the local ethics committee and written consent was obtained from next-of-kin. All available postmortem subjects were included. LAA’s were removed from the heart in a circumferential fashion by the pathologist performing the post-mortem. Once LAA samples were collected, they were subsequently opened up through a longitudinal dissection to reveal the inner surface with particular attention paid to the pectinate muscle orientation. The samples were then examined, photographed and disposed of. Some histological samples were taken for other projects with ethical approval and consent.
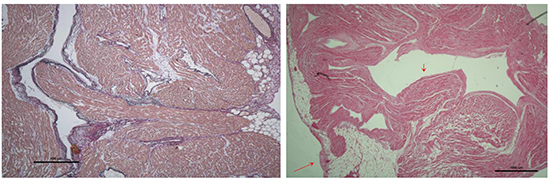
Eleven samples were collected for visual analysis, two were not included in this paper due to poor sample quality. On examination of the nine remaining samples, several common patterns of pectinate muscle orientation were noted. The LAA samples were noted to have a smooth circumferential neck of muscular tissue. This gave rise to a dominant singular smooth trunk of papillary muscle in 6 cases and two trunks in 3 cases. These trunks were either shallow (5 samples) or more muscular and raised (4 samples). Shallow trunks tended to be wider than the raised trunks and may even be circumferential (2 samples). (Table 1)
Table 1.
| Patient |
Sex |
Age |
History AF |
No. muscular trunks |
Shallow/raised trunks |
% Smooth Muscle |
| 1 |
Female |
83 |
Yes |
1 |
Shallow |
<75% |
| 2 |
Male |
70 |
Yes |
2 |
Shallow |
<75% |
| 3 |
Male |
56 |
No |
2 |
Raised |
≤50% |
| 4 |
Female |
90 |
Yes |
2 |
Raised |
≤50% |
| 5 |
Male |
75 |
No |
1 |
Shallow |
50-75% |
| 6 |
Female |
71 |
No |
1 |
Raised |
≤50% |
| 7 |
Male |
63 |
No |
1 |
Raised |
50-75% |
| 8 |
Male |
66 |
Yes |
1 |
Shallow |
50-75% |
| 9 |
Male |
59 |
No |
1 |
Shallow |
<75% |
Figure 1. Sample with single shallow trunk (red arrow), ≤50% smooth papillary muscle
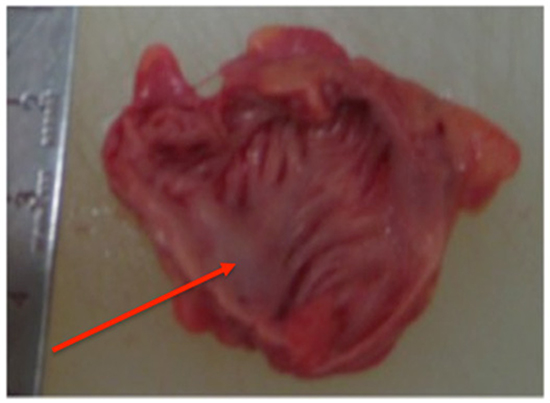
Figure 2. Sample with two raised trunks (red arrows) dividing early into second order branches, ≤50% smooth papillary muscle
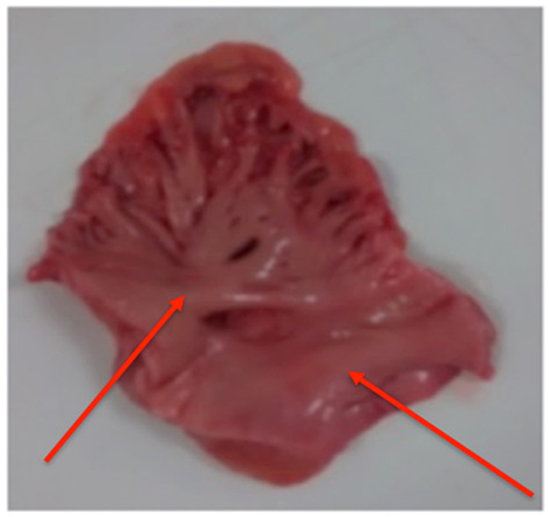
Figure 3. Sample with single smooth trunk (red arrow), >75% smooth papillary muscle
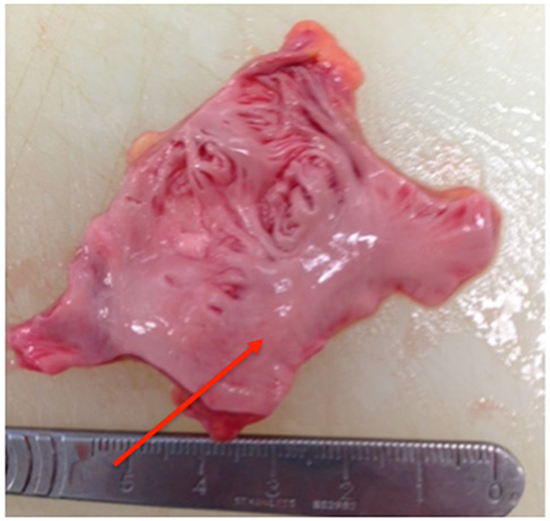
The main trunk arborized to give off papillary muscle branches down to third order or in some cases to fourth order branches. The samples were visually assessed for the percentage of smooth papillary muscle versus non-papillary recesses and were found to have ≤50% smooth muscle in 3 samples, 50-75% in 3 samples and >75% in 3 samples.
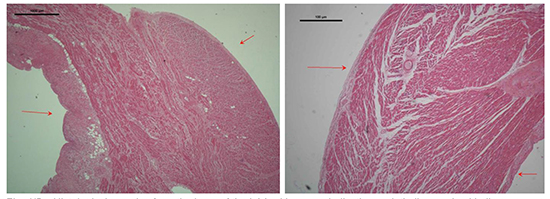
We performed histological analysis of further LAA samples from a parallel study gathered during surgical LAA excision. Using these samples we identified a pattern of myocyte orientation of the neck, mid-section and apical section of the LAA. In the images, the epithelial surface can be identified by the presence of adipose tissue. We found that the neck or proximal portion of the LAA had dense circumferential orientation of the myocytes.
Figure 4. Histological samples from the base of the LAA with arrows indicating endothelium and epithelium
In the mid-section of the LAA, arborization of the papillary muscles can be appreciated with fibrous separation between myocyte bundles.
Figure 5. Histological samples from the mid-section of the LAA with arrows indicating endothelium and epithelium
Finally, in the apical segment of the LAA, myocytes can be seen reaching the epithelial surface in discrete bundles with very little communication between each bundle, often limited to one or two individual myocytes.
Figure 6. Histological sample of LAA apex showing myocyte bundles reaching the epithelial surface with minimal communication between bundles (red arrow)
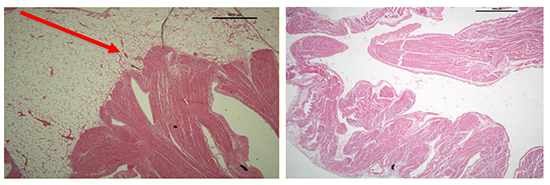
We have identified via the examination of a small number of left atrial appendages a common pattern of macroscopic papillary muscle orientation; namely a circumferential smooth ostium with the emergence of one or two papillary muscle trunks. Histological analysis of separate LAA samples have shown arborization of myocyte fibers with minimal communication in distal segments of the LAA. Perhaps the presence of prominent or multiple muscle trunks could result in difficulty in full isolation of the appendage during LAA ablation. These muscular bundles may also serve as a source of triggered arrhythmias. We believe, although our study is limited by small sample size, further study in this area is warranted and that this information could be used to investigate ablation strategies.
It is known that longer p wave durations are associated with increased risk of AF and a reduction post PVI are associated with improved outcomes.(16, 17) Kawamura et al. examined the resultant effects of left atrial ligation on atrial conduction. They found that LAA exclusion in 15 patients with paroxysmal and persistent AF by the LARIAT device, which results in LAA necrosis, results in a significant reduction in p wave duration and amplitude.(18) There were greater reductions in p wave duration and amplitude in those who remained free from AF than those who had recurrence. These p wave changes are consistent with decreased atrial mass and decreased atrial dispersion that may represent reverse electrical atrial remodeling. This may explain in part the positive results seen in LAA isolation.
Concerns regarding empiric ablation of the LAA include the loss of LAA contraction and hence increase thrombogenicity and stroke risk. Indeed LAA ablation has been shown to result in an increase in LAA thrombus formation.(15) Interestingly, although there was no increase in TIA/stroke, in the cohort of patients in the BELIEF trial who underwent LAA isolation, 52% had impaired LAA function.(14) It is noteworthy that in some patients who have previously undergone LAA isolation by endocardial catheter ablation, reconduction to the LAA can occur with relative high LAA exit velocities on transesophageal echo. The use of concomitant LAA occlusion devices could be used as a strategy to overcome this issue. Another concern is the risk of perforation due to the thin walls of the structure. There has been found to be a 1.8% risk of cardiac tamponade with LAA ablation.(13)
Further investigation as to the macroscopic variations within the LAA and their potential thrombogenicity may reveal pectinate muscular characteristics associated with increased thrombotic risk post ablation.
The left atrial appendage plays a key role in the stroke risk associated with AF, however, its role in the initiation and propagation of AF may be underestimated. We believe that left atrial appendage muscular bundle characteristics are worthy of further investigation as to their potential role in atrial fibrillation with potential implications for ablation strategies.