Arrhythmia-Induced Cardiomyopathy: Prevalent, Under-recognized, Reversible
Rahul Dhawan, Rakesh Gopinathannair
Department of Internal Medicine, University of Louisville, Louisville, KY.
Arrhythmia-induced cardiomyopathy (AIC) is a clinical condition in which a persistent tachyarrhythmia or frequent ectopy contribute to ventricular dysfunction leading to systolic heart failure. AIC can be partially or completely corrected with adequate treatment of the culprit arrhythmia. Several molecular and cellular alterations by which tachyarrhythmias lead to cardiomyopathy have been identified. AIC can affect children and adults, can be clinically silent in the form of asymptomatic tachycardia with cardiomyopathy, or can present with manifest heart failure. A high index of suspicion for AIC and aggressive treatment of the culprit arrhythmia can result in resolution of heart failure symptoms and improvement in cardiac function. Recurrent arrhythmia, following recovery from the index episode, can hasten the left ventricular dysfunction and result in HF, suggesting persistent adverse remodeling despite recovery of left ventricular function. Several aspects of AIC, such as predisposing factors, early diagnosis, preventive measures to avoid adverse remodeling, and long-term prognosis, remain unclear, and need further research.
Key Words : Arrhythmia, cardiomyopathy, heart failure, arrhythmia-induced cardiomyopathy, left ventricular dysfunction, recovery.
Correspondence to: Rakesh Gopinathannair, MD, MA, FAHA, FACC, FHRS
Director of Cardiac Electrophysiology
Associate Professor of Medicine
Division of Cardiovascular Medicine
University of Louisville and Jewish Hospital
550 So. Jackson St., ACB/AL41
Louisville, KY 40202
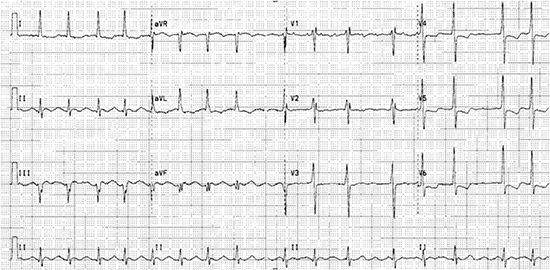
A 53-year-old man with history of severe mitral regurgitation and paroxysmal atrial fibrillation (AF) with preserved left ventricular ejection fraction (LVEF) undergoes mitral valve repair and MAZE procedure and presents a year later with a 2-month history of progressive fatigue, exertional dyspnea, and dry cough. His blood pressure was 116/74 mm Hg and, heart rate (HR) was 95 beats per minute (bpm). Physical exam revealed an irregular rhythm, bibasilar lung crackles and peripheral edema. Echocardiogram showed normal mitral valve function left ventricular (LV) end-diastolic dimension of 6.3 cm with severe LV dysfunction (LVEF 20-25%) and global hypokinesis. Coronary angiogram showed no obstructive disease. A 12-lead electrocardiogram (ECG) is shown [Figure 1].
Figure 1. 12-lead ECG showing atrial flutter with variable AV block suggestive of left atrial origin. Ventricular rate is about 95 beats per minute.
What is the optimal management strategy for this patient?
Heart failure (HF) is increasing worldwide and identifying a potentially reversible cause for HF and cardiomyopathy (CMP) can lead to appropriate management with improvement in symptoms and LVEF.[1] Arrhythmia-induced cardiomyopathy (AIC) is an important, potentially reversible cause of HF. The first description of AIC dates back to 1949 when Philips and Levine recognized that rapid AF can lead to HF in patients with otherwise normal hearts.[2] Despite the awareness in medical literature for many years, AIC still remains under-recognized.
AIC can be defined as a clinical entity in which a pathologic tachyarrhythmia or frequent ectopy ultimately leads to LV dysfunction and systolic HF.[3] Cardiomyopathy from arrhythmias can happen de novo in a structurally normal heart (arrhythmia-induced) or can be superimposed on pre-existing structural heart disease (arrhythmia-mediated).[1] The characteristic feature of AIC is recovery of LV function, either complete or in part, following elimination or adequate suppression of the arrhythmia.
Several arrhythmias can result in AIC and are listed in [Table 1].[3] A clear threshold HR for development of AIC does not exist; however, several arrhythmia-related factors contribute to AIC.[1] A study in patients with focal atrial tachycardia (AT) showed that those with slower, unremitting tachycardia had a higher risk for developing AIC.[4] In terms of premature ventricular contractions (PVCs), a high daily burden of PVCs appears to be a major predictor for AIC.[5], [6]
Table 1. Reproduced with permission from Gopinathannair R et al, JACC 2015 (reference # 1).
| Causes of Arrhythmia-Induced Cardiomyopathy |
| Superventricular |
| Atrial fibrillation |
| Atrial flutter |
| Atrial tachycardia |
| AV nodal re-entrant tachycardia |
| AV re-entrant tachycardia |
| Permanent junctional reciprocating tachycardia |
| Junctional ectopic tachycardia |
| Ventricular |
| Idopathic ventricular tachycardia |
| Fascicular tachycardia |
| Ectopy |
| Frequent premature ventricular contractions |
| Frequent premature atrial contractions |
| Pacing |
| Persistent rapid atrial abd/or ventricular pacing |
AV=atrioventricular
Age is not a barrier for AIC development as it can affect patients in intrauterine life, infancy, childhood and adulthood.[1], [7] The common types of arrhythmias resulting in AIC do differ in adults and children. AIC is most commonly secondary to AF in adults.[8] Atrial ectopic tachycardia (59%) and permanent junctional reciprocating tachycardia (23%) are common causes of AIC in children.[1] AIC can occur in patients with structural heart diseas.[1] as well as in transplanted hearts.[9] Some role for genetic predisposition in development of AIC has been found but literature on this is limited.[1] It is anticipated that several other factors might also be playing a role in patient’s predisposition to develop AIC, but are not well understood. It is likely the interplay of arrhythmias and patient characteristics that determine the transition from arrhythmia to cardiomyopathy. In children, about 28% of patients with AET develop AIC.[1] Among the adult population, about 10% of those with focal AT[4], and about 9-34% of those with PVCs or non-sustained ventricular contractions have been shown to develop AIC.[1] There are inherent selection biases in these data. The accurate incidence and prevalence of AIC remains to be defined.
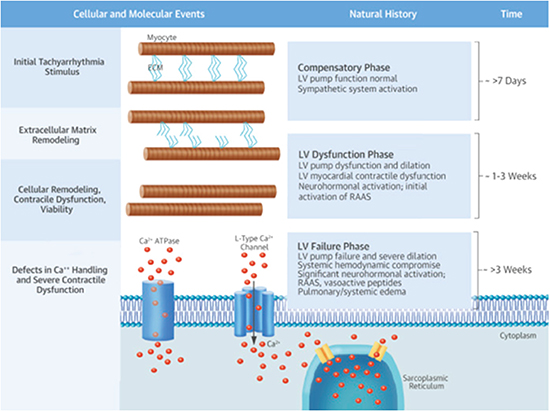
Diligent investigations on carefully constructed animal models have helped further the mechanistic relationship between arrhythmia and CMP. Much of the seminal work in this arena was conducted by Dr. Francis Spinale and colleagues[10]-[12], who used a model of rapid-pacing induced CMP. A full discussion of the pathogenetic mechanisms underlying AIC is beyond the scope of this review but a brief overview of the cellular, extracellular, biochemical and contractile changes in response to tachycardia is provided in [Figure 2]. Data from animal models, however, are not easily translated to clinical practice as several gaps in our knowledge exist regarding arrhythmia-specific pathophysiological mechanisms.
Figure 2. Figure explaining pathophysiology of arrhythmia-induced cardiomyopathy (AIC).It shows the time course of events that happen in response to rapid pacing in animals. Defects at level of ECM and cellular remodeling, along with defects in handling of calcium by cells and neurohormonal activation ultimately leads to left ventricular dysfunction and heart failure. Reproduced with permission from Gopinathannair R et al, JACC 2015 (reference # 1).
AIC can have varied presentations. In utero, AIC can present as hydrops fetalis.[7] In children and adults, the presentation of AIC can vary from asymptomatic arrhythmia with cardiomyopathy to end-stage HF. As the name suggests, there are 2 components of AIC- arrhythmias and an unexplained CMP. Arrhythmias may be apparent if the patient has symptoms or already have a pre-existing diagnosis. However, many patients might be asymptomatic from arrhythmias making early diagnosis difficult. Furthermore, expression of symptoms by children might be a challenge in this sub-group of patients. In these patients, AIC may not be detected till manifest HF develops.[1]
The relationship between arrhythmias and CMP can oftentimes create confusion as to whether the arrhythmia is the cause or culprit of CMP. Frequently, arrhythmias are not aggressively treated, resulting in worsening of CMP and HF. The following clinical scenarios should raise suspicion for either arrhythmia-induced or arrhythmia-mediated CMP.
a. Simultaneous presentation of a tachyarrhythmia/frequent ectopy and systolic HF from LV dysfunction in a patient with no prior cardiac disease.
b. Asymptomatic CMP in the setting of a persistent arrhythmia or frequent ectopy.
c. A patient with known structural heart disease or CMP now with worsening LV dysfunction and HF secondary to an arrhythmia.
The above-mentioned case scenarios are not diagnostic for AIC, as all of them could have other causes for HF and/or CMP. But it is crucial that AIC is considered high up in the differential diagnosis list and the arrhythmia managed aggressively given the potential for partial or complete reversibility of HF and LV dysfunction in AIC, irrespective of underlying structural heart disease. In summary, whenever arrhythmias and HF co-exist, one should have a high index of suspicion for AIC and aggressive treatment of both becomes necessary.
The following clinical scenario illustrates the challenges of the above-mentioned possibilities, and the significance of a correct diagnosis.
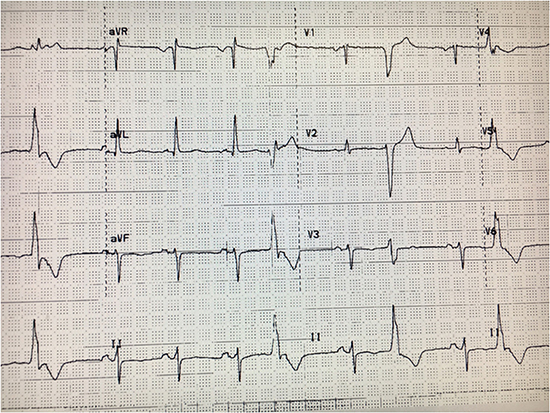
A 70-year-old female with history of dilated nonischemic CMP, ventricular tachycardia (VT) by records and status post implantable cardioverter defibrillator (ICD) implantation at an outside hospital 5 years ago, was evaluated for ICD repositioning for radiation therapy for breast cancer. Patient was on digoxin, beta-blocker and amiodarone. Clinical exam was unremarkable. 12-lead ECG showed HR of 64 bpm and no other abnormalities. Echocardiogram showed an LVEF of 60%. (LV end diastolic diameter of 5.1 cm). No arrhythmias were seen on device interrogation. Her ICD generator was removed, amiodarone and digoxin were stopped and lisinopril was added. The patient completed her radiation therapy for breast cancer but few months later, complained of palpitations and fatigue but no HF symptoms. 12-lead ECG [Figure 3] showed sinus rhythm with ventricular bigeminy of a left bundle branch block, inferior axis morphology. A 24-hour Holter monitor showed a PVC burden of 36%. Repeat echocardiogram showed an LVEF of 30% and global hypokinesis.
Figure 3. 12-lead ECG showing sinus rhythm with frequent premature ventricular contractions (PVCs) having a left bundle branch block, inferior axis morphology.
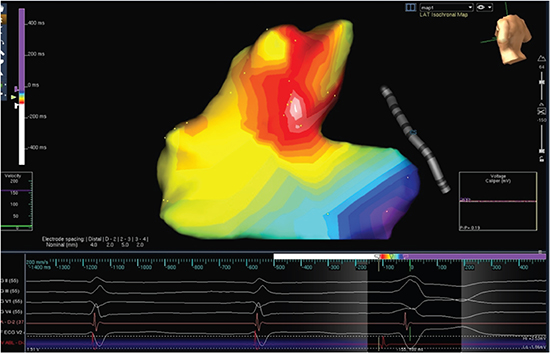
PVC-mediated AIC was suspected and patient underwent EP study where PVCs were mapped to the posterior right ventricular outflow tract and was successful ablated [Figure 4]. Her functional capacity significantly improved within a few weeks and TTE 8 months after ablation showed LVEF of 41%. Another TTE performed 28 months after ablation showed complete recovery of LVEF (56%) and right ventricular function. Beta-blockers and angiotensin-converting enzyme inhibitors (ACEI) were continued during this period.
Figure 4. 3D electroanatomic activation map of the right ventricular outflow tract showing the site of earliest activation and successful site of ablation (white colored area) in the posteroseptal right ventricular outflow tract.
Looking at this case retrospectively, it is likely that her initial diagnosis of dilated CMP and VT was PVC-induced. Amiodarone use might have resulted in decline of her PVC/VT burden leading to improvement of LVEF. The patient developed high PVC burden several months after amiodarone was stopped, resulting in CMP. The longer recovery time for LVEF could suggest persistent ultrastructural changes from her initial diagnosis of CMP.
Diagnosis of AIC can be challenging at times. Physical exam findings can often be non-specific and standard imaging may not be able to differentiate AIC from dilated CMP and consequent arrhythmia.
Several studies have reported findings that might help in early diagnosis. Absence of late gadolinium enhancement on cardiac magnetic resonance imaging might rule out underlying structural heart disease.[13] LVEDD has been noted to be lower in AIC patients, when compared to patients of dilated CMP.[14] Unipolar electroanatomic mapping can be helpful in differentiating AIC from irreversible CMP patients.[14] It was shown in a study that rapid decline of NT-pro B-type natriuretic peptide levels at one week following arrhythmia suppression favors the diagnosis of AIC.[15] Ultimately, recovery of LV function following successful treatment of the arrhythmias helps confirm the diagnosis of AIC.[1]
Management of AIC should involve the following:-
(1) Aggressive treatment of culprit arrhythmia with a goal for cure, (2) Acute treatment for HF, (3) Close surveillance and continuation of standard neurohormonal antagonists as patient’s LV function improves over time.
Interventions to Control Arrhythmias
This is critical for ensuring recovery and should be done as early as possible in these patients.
Different agents alone or in combination can be used to achieve rate control in AF. Although lenient rate control (<110 bpm at rest) [16] is comparable to strict rate control, there has been debate over target HR in AIC patients. However, in certain AF patients[17], irregular rhythm by itself can lead to LV impairment despite adequate rate control therapy. Thus, a rhythm control strategy may be needed in a large portion of AF patients.[1]
Rhythm control strategies include electrical cardioversion, anti-arrhythmic drugs (AAD), and catheter ablation. AADs are often used for pharmacologic cardioversion and for maintenance of sinus rhythm. They are also useful for PVC-mediated and other supraventricular arrhythmia mediated AIC if catheter ablation is not feasible, not desired, or was unsuccessful.[1]
A study of 58 patients with AF and HF [18] showed that pulmonary vein isolation improved LVEF and functional status regardless of underlying heart disease and adequacy of rate control prior to ablation. In patients with AF and HF, catheter ablation has also been shown to be more effective than atrioventricular (AV) node ablation and biventricular pacing.[19] A study on 203 patients with persistent AF and HF showed that catheter ablation is better than amiodarone in terms of long-term efficacy and LVEF improvement.[20] Catheter ablation should be considered the treatment of choice for atrial flutter, focal AT and other supraventricular arrhythmia-induced CMP.[1] A meta-analysis of 158 studies [21] showed that catheter ablation has approximately 91% success rate for atrial flutter and a low risk of complications. However, one should follow these patients closely given the risk for development of AF with time. Medi et al has shown excellent results with catheter ablation for focal-AT mediated AIC.[4]
For PVCs, catheter ablation has higher success rates than AADs in reducing frequency of PVCs and in improving cardiac function.[22] PVCs have been associated with poor response to cardiac resynchronization therapy (CRT) in some HF patients. Radiofrequency ablation of PVCs in these patients improves LV function and can enhance efficacy of CRT.[23]
Conflicting evidence exists as to the efficacy of rate versus rhythm control for patients with AF and CMP. The AF-CHF trial showed no difference[24] but had several shortcomings when it comes to AIC patients. The CAFÉ-II trial[25] showed that cardioversion and AAD therapy, when compared to rate control therapy, significantly improved LVEF and quality of life in patients with persistent AF and HF. Cardioversion with antiarrhythmic therapy should only be used in atrial flutter patients as an option if catheter ablation is not possible.[1]
Management Strategy for the patient in Clinical Vignette # 1:
The 12-lead ECG [Figure 1] shows atrial flutter with variable AV block and an average ventricular rate of 95 bpm. Flutter wave morphology is positive in V1 and inferior leads and negative in aVL and is strongly suggestive of a left atrial origin, especially given history of prior MAZE procedure. 24-hour Holter monitoring showed average ventricular rate of 114 bpm (range: 91-138 bpm). The patient was already on digoxin and beta-blockers for rate control. In this patient, who now has both atrial flutter and HF, further rate control and titration of HF medications, although needed, may not be enough to improve his symptoms and LV function. Moreover, in atrial flutter, it is difficult to achieve and maintain adequate rate control. Available data would support rhythm control in this situation.
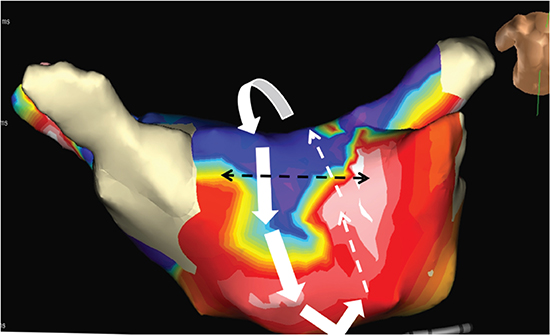
Treatment options including antiarrhythmic therapy with cardioversion as well as catheter ablation were discussed with the patient and patient chose to undergo catheter ablation. Electrophysiology study showed a macro-reentrant left atrial flutter with a critical isthmus localized to the anterior aspect of the left atrial roof [Figure 5]. Catheter ablation at this location terminated the flutter and made it non-inducible. Reconnected pulmonary veins were isolated again. There was complete resolution of HF symptoms following restoration of sinus rhythm. Echocardiogram performed at 3 months post-ablation showed normal LV size and function and with an LVEF of 50-55% Diagnosis of atrial flutter-induced AIC was made. Patient has not had any recurrent atrial arrhythmias over a follow-up period of 3 years.
Figure 5. 3D electroanatomic activation map showing a macro-reentrant left atrial flutter with a critical isthmus localized to the anterior aspect of the left atrial roof. Interrupted black lines show ablation line across the roof.
Appropriate therapy for HF is essential in these patients. ACEI and beta-blockers can aid in favorable LV remodeling but the duration of therapy is not well understood.
Continuation of HF therapy as patient’s LV function improves over time
Following arrhythmia control, LV function generally improves over time in these patients. Beta-blockers and ACEI have role in favorable remodeling, and it is not very clear as to when they should be stopped after initial LV improvement in AIC patients.[26]
Different studies point to different timelines for recovery of LV function following treatment of the culprit arrhythmia. A study on 24 patients with predominantly AF-mediated AIC showed that LV function improved in a mean of 5.8 months.[8] In a study of PVC-mediated AIC[27], in whom PVCs were adequately treated, 2/3rd of patients recovered LV function by 4 months. For the rest of the group, the time to recovery ranged from 5-45 months. Overall, all patients in the group had LV function improvement. It was noted that late responders of LVEF improvement usually had source of PVCs in the epicardial region.
It has been noted on cardiac imaging that despite improvement in LVEF, LV dimensions remain elevated when compared to age, gender and LVEF matched control group.[28] Moreover, one study reported cases of sudden cardiac death in AIC patients who had already recovered in regards to their LVEF.[8] It was noted that those patients had lower LVEF at presentation as compared to other patients in the study. This raises concern as to whether some degree of negative remodeling persists despite arrhythmia suppression and LV function recovery. This is also illustrated by the evidence that recurrent tachyarrhythmias following initial recovery can result in rapid decline in LVEF.[8] Thus, AIC takes months to years to develop, months to resolve, and recurrent arrhythmias can be calamitous.
The concept of AIC has been reported several decades ago, but has been more and more recognized recently. The trademark of AIC is the reversal of CMP following suppression or elimination of the culprit arrhythmia, and therein lies the significance of this fascinating disease entity for the practicing clinician. In a world where HF incidence keeps increasing, identifying and treating a reversible cause for HF and CMP is critical and AIC really exemplifies that old adage of “a stitch in time”. AIC remains under-recognized, probably diagnosed as idiopathic CMP in many cases with no focused monitoring for any culprit arrhythmias. In others, arrhythmias are often attributed to structural heart disease and/or HF. It is not to be forgotten that even in patients with a known structural heart disease, like ischemic disease, arrhythmias can worsen CMP and result in HF. A high index of suspicion and appropriate management of these patients can change their lives, as highlighted in the clinical vignettes in this article. However, negative remodeling may persist following initial LV function recovery in AIC patients and recurrent arrhythmias can hasten development of CMP. Close surveillance is thus needed in these patients.
Although significant progress has been made in understanding different aspects of pathogenesis, diagnosis and treatment of AIC, further studies are warranted to thoroughly fill in the gaps in knowledge of this disease entity, mainly with respect to arrhythmia-specific mechanisms, predisposing factors and long-term outcomes.