The Mitral Inflow E/A Ratio Before The Procedure May Predict Late Recurrence in Patients With Atrial Fibrillation Undergoing Cryoballoon Ablation
Taner Ulus1, Ahmet Serdar Yilmaz1, Emre Şener1, Halit Emre Yalvaç1, Ezgi Çamlı1, Fatih Enes Durmaz1, Yüksel Çavuşoğlu1
1Department of Cardiology, Faculty of Medicine, Eskişehir Osmangazi University, Eskişehir, Turkey.
High recurrence rates after catheter ablation for atrial fibrillation (AF) is a major problem. Many predictors for recurrence after AF ablation have been described. Left ventricular diastolic dysfunction (LV DD) is directly related to most of such predictors. Some studies investigating the relationship between LV DD and late recurrence after AF ablation have found conflicting results. The mitral inflow E/A ratio is a simple and practical method commonly used to evaluate LV DD. We aimed to determine whether the E/A ratio before the procedure was predictive for late recurrence in patients with AF undergoing cryoballoon ablation.
A total of 99 patients undergoing AF ablation for the first time using second-generation cryoballoon were included. Only patients with paroxysmal AF and sinus rhythm the day before the procedure, and pre-procedural pro-B natriuretic peptide levels within normal limits were included. The patients who developed recurrence and those who did not were compared in terms of basal characteristics and procedural features. Any atrial tachyarrhythmia episode longer than 30 s after the blanking period was defined as late recurrence.
The patients [age: 58 (50-62) years, 53.5% female] were followed up for a median of 45.0 (15.0-63.0) months and late recurrence developed in 25 (25.2%) patients. The E/A ratio was lower in patients with late recurrence than in those without it [0.5 (0.4-1.2) vs 1.4 (0.6-1.7), respectively, p=0.001]. A multivariate analysis showed that female gender (HR: 4.44, 95%CI: 1.31-15.05, p=0.017), early recurrence (HR: 7.35, 95% CI: 2.28-23.71, p=0.001), and E/A ratio (HR: 0.28, 95%CI: 0.11-0.68, p=0.005) were independent predictors for late recurrence.
Female gender, early recurrence, and E/A ratio are independent predictors of late recurrence in patients with AF undergoing cryoballoon ablation. Measuring the E/A ratio in patients with sinus rhythm before AF ablation may help to predict future recurrences.
Key Words : .
Taner Ulus, MD, Prof.Department of Cardiology, Faculty of Medicine, Eskişehir Osmangazi University, Eskişehir, Turkey
Antral pulmonary vein isolation (PVI) is a mainstay for catheter ablation of atrial fibrillation (AF) 1. However, high recurrence rates after catheter ablation for AF are a major problem. Previous studies have identified some predictors associated with recurrence such as female gender, obesity, AF duration, early recurrence, elevated CHA2DS2-Vasc score, and left atrial (LA) diameter 2-6. The left ventricular (LV) diastolic dysfunction (DD) is related to most of the above predictors, and it has been shown that DD increases the risk of AF development 7.
Some investigators also investigated whether a relationship between DD and long-term recurrence after catheter ablation for AF, but they found conflictory results. Some of them found the relationship between DD and late recurrence 8 while others did not show such association 9. These studies had relatively short follow-up duration and they did not have a standard ablation protocol. Some patients were taken to PVI only, while additional linear lesions were added in localizations such as the LA roof, LA isthmus, and cavotricuspid isthmus in others 8,9. These issues are likely to have affected the results of such studies. The mitral inflow E/A ratio is a simple and practical indicator of DD 9. Cryoballoon (CB) is a widely used option for catheter ablation of AF, and it has similar efficacy and safety outcomes compared to radiofrequency ablation 1. In this study, we aimed to investigate whether the mitral inflow E/A ratio before the procedure was predictive for late recurrence in patients with AF undergoing pulmonary vein isolation (PVI) with cryoballoon.
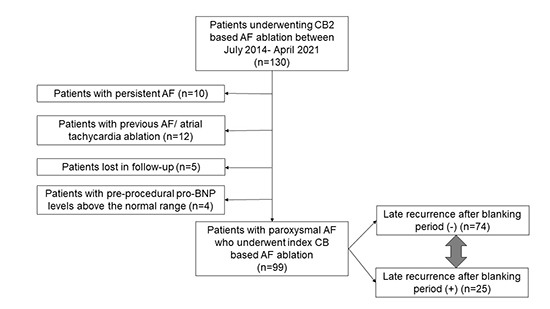
Among 130 patients undergoing PVI for AF using secondgeneration cryoballoon (CB2) at Eskisehir Osmangazi University hospital between July 2014 and April 2021, a total of 99 patients with paroxysmal AF undergoing ablation for the first time were included in the study. To measure the mitral inflow E/A ratio, only patients with paroxysmal AF and sinus rhythm the day before the procedure were included. Paroxysmal AF was defined by current guidelines 10. In addition, pre-procedural pro-B natriuretic peptide (pro-BNP) levels of all patients included in the study were within normal limits. Patients with moderate/ severe valve disease, recent decompensated heart failure (HF) attack, pseudonormal or restrictive LV filling pattern, acute myocardial ischemia, cardiac surgery within 3 months before ablation, LA thrombus, posteroanterior LA diameter >45 mm, active hyperthyroidism, pregnancy or serious comorbidities were excluded. [Figure 1] shows the flow chart of the study protocol.
Figure 1. Flow chart of the study. AF: Atrial fibrillation, pro-BNP: pro-B natriuretic peptide, CB: Cryoballoon, CB2: Second-generation cryoballoon.
Detailed medical history including cardiovascular diseases, cardiovascular risk factors, and medications was recorded. The CHA2DS2-Vasc score was calculated and the duration of AF before ablation was recorded. Pro-BNP levels were studied one day before catheter ablation. The study was performed according to the Declaration of Helsinki principles and the local ethics committee approved the study.
Transthoracic echocardiography was done to assess LV ejection fraction (LVEF), valvular pathologies, intracavitary dimensions, and LV mass index. To evaluate LV diastolic functions, the sample volume was positioned at the mitral leaflet tips in the apical four-chamber view. Transmitral early and late diastolic peak flow velocities (E wave and A wave, respectively), deceleration time of E wave (DT), and isovolumic relaxation time (IVRT) were recorded using pulse wave Doppler. The E/A ratio was calculated. All measurements were made in sinus rhythm the day before the procedure in accordance with the recommendations 11,12. All patients underwent pre-procedural multidetector computed tomography to evaluate LA and PV anatomy. If the patients were taking warfarin and the international normalized ratio was <2.5 before the procedure, ablation was performed without discontinuing warfarin. New oral anticoagulants were interrupted 24 h before the procedure.
Antiarrhythmic drugs were ceased at least 5 days before the procedure. Transesophageal echocardiography was performed within 24 hours before catheter ablation to exclude the presence of LA thrombus in all patients.
All procedures were performed conscious sedation with dexmedetomidine infusion or midazolam and fentanyl boluses. Details of the procedure have been described previously 13. One arterial and two venous sheaths were inserted from the right inguinal region. A 6F steerable quadripolar decapolar catheter (St. Jude MedicalTM) and a 6F pigtail catheter (AlvisionTM) were positioned in the coronary sinus and aortic root, respectively. Single transseptal (TS) puncture was performed by modified Brockenbrough technique using a TS needle (BRK-1TM, St. Jude Medical) and an 8F TS sheath was placed in the LA. Then, a bolus of 100 U/kg unfractionated heparin bolus was administered immediately and the activated clotting time was maintained for 300-350 s with repeated bolus administrations. The TS sheath was exchanged with a 15F steerable sheath (FlexCath Advance, Medtronic Inc., Minneapolis, MN, USA) over the wire. A 28-mm CB2 catheter (Arctic Front AdvanceTM, Medtronic) was used for PVI. A circular mapping catheter (Achieve™, Medtronic, Minneapolis, MN, USA) was inserted through the lumen of the CB catheter to record PV potentials and to maneuver the balloon. After confirming complete occlusion of the PV by application of 50% diluted contrast agent, a freezing cycle of 180-240 s was applied for each PV. An additional bonus freeze of 120 s was performed if PVI was not obtained within 60 s or early PV reconnection occurred 14. The right phrenic nerve was paced with a 1500/2000 ms cycle and a 12/15-mA output while freezing the right-sided PVs to avoid phrenic nerve paralysis. The phrenic nerve stimulation was monitored by intermittent fluoroscopy and direct palpation of the right hemi-diaphragmatic contraction. Acute procedural success was defined as the disappearance or dissociation of all visible PV potentials. Entrance and exit blocks were tested by pacing through the coronary sinus and circular catheter, respectively.
Transthoracic echocardiography was repeated immediately after the procedure. If no pericardial effusion was observed on echocardiography, oral anticoagulation was restarted on the evening of the procedure and continued for at least 3 months. Antiarrhythmic drugs were also continued for at least 3 months after the procedure. Routine follow-up visits were programmed at 1, 3, 6, and 12 months after ablation and every one year thereafter. If the patients had any complaints compatible with atrial tachyarrhythmia (ATa) or procedure-related complications, they were evaluated earlier. Follow-up visits are composed of the evaluation of arrhythmia-related complaints, physical examination, 12-lead electrocardiogram (ECG), and 24-h Holter ECG monitoring. Any ATa episode (AF, atrial flutter or atrial tachycardia) longer than 30 s was defined as recurrence. The first 3-month period after ablation was considered as the blanking period. Recurrences within and after the blanking period were defined as ‘early recurrence’ and ‘late recurrence’, respectively 13.
A one-sample Shapiro-Wilk test was used to determine whether continuous variables were normally distributed. Normally and skewed distributed continuous variables were presented as mean±standard deviation and the median (25th, 75th percentiles), respectively, and they were compared between the groups using the Student’s t-test and Mann-Whitney U test, respectively. Categorical variables were presented as frequencies and percentages and were compared using the chi-square test. The Cox proportional hazards model was used to find the predictors of long-term ATa recurrence. Variables with a P-value of <0.05 in univariate analysis were included in multivariate analysis. ATa-free survival was estimated by the Kaplan-Meier method and compared by log-rank test. An optimal cut-off value of the mitral inflow E/A ratio to predict the long-term recurrence was determined by the receiver-operating characteristic curve analysis. Statistical analyses were performed using statistical software (IBM SPSS Statistics for Windows, Version 23.0, Armonk, NY: IBM Corp., USA), and a twotailed P-value of <0.05 was considered statistically significant.
All patients underwent only PVI using CB2. No additional ablation was performed in the patients. The age of the patients was 58 (60-62) years and 53 (53.5%) were female. Fifty-three patients (53.5%) were hypertensive and 23 (23.2%) had diabetes mellitus. The median CHA2DS2-Vasc score was 1.0 (1.0-2.0). The baseline characteristics of the study population were summarised in [Table 1].
Table 1. Baseline characteristics and procedural features of all patients, those with and without late recurrence
|
Total (n=99) |
Late recurrence (-) (n=74) |
Late recurrence (+) (n=25) |
P value |
| Age (years) |
58.0 (50.0 – 62.0) |
58.0 (47.7 - 62.0) |
58.0 (54.0 - 66.0) |
0.329 |
| Female gender (n,%) |
53 (53.5) |
35 (47.3) |
18 (72.0) |
0.032 |
| Body mass index (kg/m²) |
27.7 (25.7 - 29.9) |
27.2 (25.7 - 29.5) |
28.6 (27.3 – 31.0) |
0.149 |
| Hypertension (n,%) |
53 (53.5) |
35 (47.3) |
18 (72.0) |
0.032 |
| Diabetes mellitus (n,%) |
23 (23.2) |
15 (20.3) |
8 (32.0) |
0.230 |
| CAD (n,%) |
9 (9.1) |
5 (6.8) |
4 (16) |
0.224 |
| HF with reduced EF (n,%) |
2 (2.0) |
1 (1.4) |
1 (4.0) |
0.443 |
| Current smoking (n,%) |
7 (7.1) |
6 (8.1) |
1 (4.0) |
0.675 |
| Alcohol intake (n,%) |
2 (2.0) |
1 (1.4) |
1 (4.0) |
0.443 |
| CHA2DS2-VASc score |
1.0 (1.0 – 2.0) |
1.0 (0 - 2.0) |
2.0 (1.0 - 3.0) |
0.013 |
| AF duration (months) |
23.6 ± 8.9 |
22.3 ± 8.3 |
27.8 ± 9.4 |
0.007 |
| Beta blocker (n,%) |
56 (56.6) |
42 (56.8) |
14 (56.0) |
0.947 |
| RAAS blocker (n,%) |
45 (45.5) |
30 (40.5) |
15 (60.0) |
0.091 |
| Statin (n,%) |
10 (10.1) |
7 (9.5) |
3 (12.0) |
0.710 |
| Propafenone (n,%) |
26 (26.3) |
19 (25.7) |
7 (28.0) |
0.819 |
| Amiodarone (n,%) |
15 ( 15.2) |
9 (12.2) |
6 (24.0) |
0.197 |
| Hemoglobin (g/dl) |
13.6 (12.6 – 14.6) |
13.6 (12.6-14.7) |
13.2 (11.9-14.2) |
0.085 |
| eGFR (ml/dk/1.73m²) |
88.9 ± 19.3 |
90.2 ± 19.6 |
85.3 ± 18.4 |
0.272 |
| pro-BNP (pg/ml) |
286.0 (186.0 - 366.0) |
281.0 (185.0 - 363.7) |
295.0 (185.5 - 417.5) |
0.635 |
| Left atrial diameter (mm) |
37.8 ± 3.9 |
37.5 ± 3.8 |
38.6 ± 4.0 |
0.231 |
| LV EF (%) |
60.0 (60.0-65.0) |
60.0 (60.0-65.0) |
60.0 (60.0-65.0) |
0.861 |
| LV mass index (g/m2) |
84.0 (71.0 - 94.0) |
82.0 (70.0 - 92.2) |
85.0 (72.0 - 104.5) |
0.176 |
| E/A ratio |
1.2 (0.5 – 1.6) |
1.4 (0.6-1.7) |
0.5 (0.4-1.2) |
0.001 |
| DT (msec) |
190.0 (170.0 – 230.0) |
180.0 (170.0 – 230.0) |
230.0 (170.0 - 242.5) |
0.079 |
| IVRT (msec) |
90.0 (75.0 – 110.0) |
85.0 (70.0 – 110.0) |
103.0 (90.0 – 111.5) |
0.016 |
| Early recurrence (n,%) |
8 (8.1) |
3 (4.1) |
5 (20.0) |
0.023 |
| Common trunk PV (n,%) |
32 (32.3) |
21 (28.4) |
11 (44.0) |
0.149 |
| Accessory PV (n,%) |
12 (12.1) |
9 (12.2) |
3 (12.0) |
1.000 |
| Procedure time (min) |
80.8 ± 18.5 |
79.2 ± 18.4 |
85.5 ± 18.2 |
0.143 |
| Fluoroscopy time (min) |
21.3 ± 6.5 |
20.7 ± 6.7 |
23.0 ± 5.6 |
0.120 |
| Follow-up (months) |
45.0 (15.0 – 63.0) |
42.5 (12.7 – 62.2) |
52.0 (22.0 – 65.5) |
0.373 |
AF: Atrial fibrillation, BNP: B natriuretic peptide, CAD: Coronary artery disease, eGFR: Estimated glomerular filtration rate, DT: Deceleration time, EF: Ejection fraction, HF: Heart failure, IVRT: Isovolumic relaxation time, LV: Left ventricle, PV: Pulmonary vein, RAAS: Renin-angiotensinaldosteron system.
Procedural findings and complications
Total procedural and fluoroscopic time were 80.8±18.5 min and 21.3±6.5 min, respectively. Common trunk PV was observed in 32 patients (32.0%) (30 left common and 2 right common) [Table 1].
A total of 394 PV including common trunks were detected, of which 389 (98.7%) were successfully isolated. Freezing numbers were 2.0 (1.0 – 3.0) for the left superior PV, 2.0 (1.0 – 2.0) for the left inferior PV, 2.0 (1.0 – 2.0) for the right superior PV, 1.0 (1.0 – 2.0) for the right inferior PV, 3.0 (2.0 – 3.0) for the left common trunk, 1.5 (0.7 – 2.0) for the right common trunk.
As procedural complications, cardiac tamponade requiring pericardiocentesis was seen in 2 patients; while diaphragm paralysis occurred in 1 patient during cryoablation at the right superior PV. Groin complications were observed in 2 patients; the patient with femoral hematoma was followed up medically and the other with femoral arteriovenous fistula underwent surgical repair. Atrio‐esophageal fistula, PV stenosis, cardiac embolism, or procedure‐related deaths were not observed in any of the patients.
The median follow-up duration was 45.0 (15.0-63.0) months. Early recurrence was developed in 8 (8.1%) patients. Of these, 3 spontaneously reverted to sinus rhythm, 4 achieved sinus rhythm with pharmacological cardioversion, and 1 with electrical cardioversion. Late ATa recurrence was observed in 25 (25.3%) patients in the long-term follow-up. Of these, 17 were followed-up medically, RF ablation was performed in 7 patients, and 1 underwent second cryoablation with CB2. Among 8 patients who underwent a second procedure, typical atrial flutter was observed in 4 patients, PV reconnection was found in 2 patients, and atrial tachycardia (one roof dependent and one right atrial tachycardia) was detected in 2 patients.
Predictors of long-term recurrence after cryoballoon ablation
When the patients were compared according to the development of ATa recurrence after the blanking period, female gender (72.0% vs 47.3%), hypertension (72.0% vs 47.3%), and the frequency of early recurrence (20.0% vs 4.1%) were found to be higher in patients who develop ATa recurrence after blanking period than in those who did not develop (p =0.032, 0.032 and 0.023, respectively). The CHA2DS2-Vasc score was higher [2.0 (1.0-3.0) vs 1.0 (0-2.0)], AF duration was longer (27.8 ± 9.4 vs 22.3 ± 8.3 months), the mitral inflow E/A ratio was lower [0.5 (0.4-1.2) vs 1.4 (0.6-1.7)], and IVRT was longer [103.0 (90.0-111.5) vs 85.0 (70.0-110.0) ms] in patients with long-term recurrence than in those without it (p=0.013, 0.007, 0.001 and 0.016, respectively). The patients’ baseline characteristics and procedural findings according to the presence of late ATa recurrence are given in [Table 1].
The total procedure time, fluoroscopy time, and PV anatomic features were similar between the groups (p>0.05 for all) [Table 1]. There was no significant difference between the groups regarding freezing numbers and freezing durations for each PV (p>0.05).
The results of Cox regression analysis to determine predictors of long-term ATa recurrence are given in [Table 2]. In the univariate analysis, female gender (HR: 2.50, 95% CI: 1.04-5.99, p=0.04), AF duration (HR: 1.05, 95% CI: 1.01-1.09, p=0.012), hypertension (HR: 2.42, 95% CI: 1.01-5.81, p=0.047), early recurrence (HR: 4.12, 95% CI: 1.54-11.02, p=0.005), the CHA2DS2-Vasc score (HR: 1.50, 95% CI: 1.13-1.99, p=0.004) and the mitral inflow E/A ratio (HR: 0.27, 95% CI: 0.12-0.62, p=0.002) were significantly associated with long-term recurrence. The multivariate analysis showed that female gender (HR: 4.44, 95% CI: 1.31-15.05, p=0.017), early recurrence (HR:7.35, 95 % CI: 2.28-23.71, p=0.001) and E/A ratio (HR: 0.28, 95 % CI: 0.11-0.68, p=0.005) were independent predictors of late ATa recurrence.
Table 2. Cox regression analysis to identify predictors of late recurrence after catheter ablation for AF
|
Univariate |
Multivariate |
|
Beta |
HR (95% CI) |
P value |
Beta |
HR (95% CI) |
P value |
| Female gender |
0.917 |
2.50 (1.04-5.99) |
0.040 |
1.491 |
4.44 (1.31-15.05) |
0.017 |
| Hypertension |
0.886 |
2.42 (1.01-5.81) |
0.047 |
0.895 |
2.44 (0.77-7.77) |
0.129 |
| CHA2DS2-VASc score |
0.409 |
1.50 (1.13-1.99) |
0.004 |
-0.16 |
0.85 (0.55-1.30) |
0.459 |
| AF duration |
0.052 |
1.05 (1.01-1.09) |
0.012 |
0.036 |
1.03 (0.97-1.10) |
0.287 |
| Early recurrence |
1.418 |
4.12 (1.54-11.02) |
0.005 |
1.995 |
7.35 (2.28-23.71) |
0.001 |
| E/A ratio |
-1.277 |
0.27 (0.12-0.62) |
0.002 |
-1.26 |
0.28 (0.11-0.68) |
0.005 |
AF: Atrial fibrillation, HR: Hazard ratio.
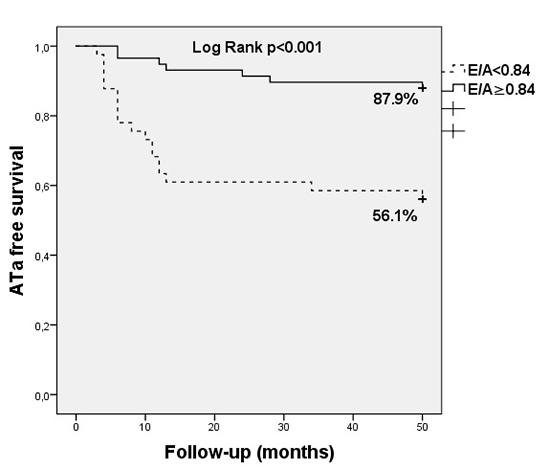
Receiver operating characteristic analysis showed that the optimal cut-off level of the E/A ratio was 0.84 (Sensitivity: 72%, specificity: 70%). Kaplan-Meier curves comparing ATa free survival according to cutoff E/A ratio of 0.84 after AF ablation are presented in [Figure 2]. Any ATa free survival rate was significantly lower in patients with E/A ratio <0.84 than in those with ≥0.84 (56.1% vs 87.9%, log-rank p<0.001).
Figure 2. Kaplan-Meier survival curve showing the comparison of ATa free survival rates according to cut-off mitral inflow E/A ratio of 0.84 after AF ablation. AF: Atrial fibrillation, ATa: Atrial tachyarrhythmia.
In this study, we investigated whether the mitral inflow E/A ratio was a predictor of ATa recurrence in patients with paroxysmal AF who underwent PVI using CB2 during median 45 months follow-up. Our main findings were: 1) Clinical success without ATa recurrence was 74.7% at long-term follow-up. 2) Female gender, hypertension, the median CHA₂DS₂-Vasc score, AF duration, the presence of early recurrence, and E/A ratio were significantly associated with late ATa recurrence after the blanking period. 3) Female gender, early recurrence, and E/A ratio were independent predictors of late recurrence.
Diastolic dysfunction is a condition that develops as a result of abnormalities in LV relaxation and compliance during diastole 12. As a result, the LA pressure increases to ensure adequate filling of the LV and atrial wall tension increases. Enlargement of the LA and PVs due to increased wall stretch may induce ectopic firing from PVs. Another mechanism is stretch-induced atrial remodeling, which consists of changes such as Ca+2 channel downregulation, myocytolysis, and atrial fibrosis. All of the factors mentioned above may contribute to the development of AF in DD 12, 15-17.
The transmitral flow velocity measurements including E wave, A wave, E/A ratio, DT, and IVRT are practical and useful tools to assess diastolic functions. Impaired LV relaxation with normal LA and LV pressures is an early form of DD. At this stage, the E/A ratio is <1, DT is >240 ms, and IVRT is >110 ms 12. In our study, we used the E/A ratio to evaluate LV DD. We did not include patients with recent decompensated HF attacks or pre-procedural pro-BNP levels above the normal range. Again, we did not include patients with the pseudonormal DD pattern or restrictive type DD by excluding patients with pre-procedural pro-BNP levels above the normal range or DT<160 and IVRT<60 msec 12,18. For this reason, we think that the low E/A ratio in our study reflects the early stage of DD.
We showed that the E/A ratio before the procedure was an independent predictor of long-term ATa recurrence after AF ablation. Some studies have investigated the relationship between DD and late recurrence and they found different results. Kosiuk et al. showed that preprocedural E/A ratio was associated with early recurrence after AF ablation, but not with late recurrence 9. Kim et al and Li et al found that DD, as measured by the E/e’ ratio was an independent predictor of late recurrence 19,20. However, a standard ablation method was not used in any of these studies. Some patients underwent PVI, while others underwent linear ablation of the LA or cavotricuspid isthmus. The lack of a standard ablation procedure is likely to have affected the results of such studies. However, we performed a standard procedure in all patients, PVI with CB2. In addition, the follow-up period of the patients was relatively longer than the others. In patients who will undergo AF ablation, the E/A ratio can be measured before the procedure to predict long-term recurrences. Closer follow-up against the possibility of recurrence and more strict control of modifiable risk factors to reduce recurrence may be considered in patients with a low E/A ratio.
The CB2 has been shown to have favorable clinical outcomes compared to first-generation one, probably due to extensive wide-area circumferential antral lesion creation 13,21. We also found that female gender and early recurrence were independent predictors of long-term recurrence in the study. Our results were compatible with previous studies 2,4. The frequency of hypertension, the median CHA₂DS₂-Vasc score, and AF duration were significantly higher in patients with late recurrence, but they were not independent predictors. The LA diameter is similar between the groups in our study. The exclusion of patients with persistent AF and LA diameter >45 mm, and the low number of patients may have been responsible for this result.
This study had some limitations. First, the study was single-center and the number of patients was small. Second, the study was of a retrospective design, and E/e’ was not included in the analyzes because mitral annular diastolic peak velocities (e’) were not recorded in most patients. Third, regular follow-up was based on clinical examination, ECG, and 24-h Holter monitoring, and some asymptomatic nonsustained ATa episodes may have been overlooked. Fourth, our study results cannot be applied to all patients undergoing AF ablation. However, our results suggest that a low E/A ratio may be an independent predictor for late recurrence after AF ablation in patients with DD in the early stage. Fifth, we did not define whether atrial substrate existed with methods such as magnetic resonance imaging in our patients. Sixth, we did not include LA volume measurements in the analyzes. Finally, the study had no follow-up data on diastolic functions after AF ablation.
The mitral inflow E/A ratio, female gender, and early recurrence are independent predictors of late ATa recurrence in patients with AF undergoing PVI with cryoballoon. Measuring the E/A ratio in patients with sinus rhythm before AF ablation may help to identify patients who may develop recurrence in the future. In this regard, there is a need for further studies with a larger number of patients, including other parameters showing diastolic functions, and in which the standard ablation method is applied in all patients.