Comparison of Cryoballoon and Hybrid surgical Posterior Wall Isolation for Persistent Atrial Fibrillation to conventional ablation
Eric Nordsieck 1, Zhang Xin J 1, Pankaj Malhotra 1, Dali Fan 1, Nayereh G. Pezeshkian 1, Uma N. Srivatsa 1
1
Division of Cardiovascular Medicine, University of California Davis..
Optimalablation strategy for persistent atrial fibrillation (AF) is unclear;PWI of the left atrium may improve outcome.Our aim was to compare outcomes of posterior wall isolation (PWI) ablation for persistent AF achieved by cryoballoon ablation (CRYO) or hybrid surgical ablation (HABL) to matched patients undergoing conventional radiofrequency ablation (CRA).
In our single center retrospective study,patients underwent HABL and CRYO withcircumferential pulmonary vein ablation (CPVA),roof and floor lines to complete PW box lesion. CRA consisted of CPVA, roof line and lateral mitral isthmus line (MVI).
Of 61 patients (mean duration of AF 1.3 ± 0.4 yrs)who underwent ablation, after follow-up of 366 ± 62 days, AF recurrence was 10.5% and 48% (p=0.001) and the need for repeat ablation 5% vs 30% (p=0.007) in PWI and CRA groups respectively, without a significant difference in incidence of AT/FL 18 vs 26 % or cardioversion 5.2 vs 7.1 %. Total procedure time and fluoroscopic time were 242 ±70 min vs 279 ±53 min (p=0.08) and 20±9 min vs 12 ± 4 min (p=0.003) for PWI and CRA respectively. CRYO had less AF recurrence and complications than HABL. Mean length of stayfor CRYO patients was 41 hrs compared to 145 hrs in HABL group, who underwent two procedures.
PWI in persistent AF patients decreases recurrence of AF and need for repeat procedure compared to CRA; PWI by CRYO is superior to HABL due to less LOS and complications.
Key Words : Persistent Atrial fibrillation, ablation, posterior wall.
Correspondence to: Uma Srivatsa, MBBS, MAS, FACC, FHRS
Professor of Medicine, UC Davis, Sacramento, CA
Catheter ablationis indicated for treatment of drug refractory symptomatic atrial fibrillation (AF).[1]Circumferential Pulmonary VeinAblation (CPVA) for PV isolation (PVI)is the cornerstone of all AF ablations regardless of stage of disease; recurrence is common particularly in persistent AF compared to paroxysmal (PAF) patients. There is no consensus on the optimal ablation strategy persistent AF patients.
Cryothermal energy delivered by cryoballoon catheter [2]ablates a wider area including triggers from the antral tissue, leaving a small area of the posterior left atrium (LA) wall unablated. [3] Techniques have been described to create linear lesions in the LA with cryoballoonto avoid gaps.[4] We expandedthese techniques to create lesions in LA roof and floor for complete posterior wall isolation (PWI) in patients with persistent AF.
In this study, we compare the clinical outcomes in patients undergoing PWI by CRYO or hybrid surgical ablation (HABL)to historical controls from our institution with persistent AF who had been treated with our previous conventional endocardial radiofrequency ablation (CRA). Within the PWI group, we compare CRYO to HABL strategies for procedural and clinical outcomes.
After approval from Institutional review board, we evaluated the records of patients ablated for AF between 2014-2017. We included age and gender matched patients (> 18 yrs) with symptomatic persistent AFwith left ventricular ejection fraction (LVEF)>30%, who had failed at least one antiarrhythmicdrug. Patients with PAF were excluded. Direct oral anticoagulants were held onthe day prior to the procedure, resumed the same evening; warfarin was continued during the procedure. Class I agents, sotalol and dofetilide were held for five days and amiodarone for 30 days prior to the procedure. All patients had CT scan prior to evaluate PV anatomy and was used to navigate during mapping.
Electroanatomic mapping was performed in all cases with CARTO 3 system (Biosense Webster Inc) in sinus rhythmfor voltage map, in the setting of 0.05 to 0.5 mV to assess PVI and PWI. Activation window for atrial flutters was set at 50% -50% of the cycle length. Roof was the line drawn between the upper ends and floor was the line drawn between the lower ends of the right and left superior PVs. The area contained within roof, floor and PV antra were designated as the PW. The attenuated area < 0.05 mV after ablation divided by the total area of the PW was considered the percent of PWI.
For patients in group CRYO, ablation involved two applicationsto create lesions using cryoballoon (ArcticFront Advance™, 28 mm second generation, Medtronic, Inc., Minneapolis, MN) for 180 sec and 120 sec at all the PVs, after ensuring adequate occlusion by pressure monitoring and PV Doppler signals on the intra-cardiac echocardiogram.Then anchoring the Achieve mapping catheter (Abbott Inc.) in the PVs,sequential retraction and posterior rotation of the balloon was employed to create overlapping and contiguous stamp lesions spanning the LA roof and floor to complete PWI.[Figure 1]We incorporated the use of CartoSound™ (Biosense Webster Inc, Diamond Bar, CA) to integrate catheter position and orientation in real time into the electro-anatomical map during PW applications to draw “CartosOund Rings” (COR Imaging). [Figure 1D] Esophageal temperature monitoring [Figure 1A] and phrenic nerve pacing for right sided lesions were employed during ablation and applications were stopped for loss of phrenic capture or esophageal temperature drop < 29°C. Lesions on the roof and floor were created with single applications of 180sec and were stopped early if esophageal temperature dropped. Remapping with Pentarray multipolar catheter (Biosense Webster Inc) following ablation was used to confirm complete and well-demarcated PVI, roof, and floor lines to demonstrate dense PWI with no electrograms >0.05mV; if necessary additional ablation with thermo-cool radiofrequency catheter was applied to close gaps. If atrial flutter was inducible, additional RF ablation was applied to eliminate the atrial flutter circuit. Posterior wall was considered isolated if there was complete electrogram attenuation to < 0.05 mV.
Figure 1. A - Fluoroscopic view of Cryoballoon retracted from left upper pulmonary vein position at the roof; White arrowhead indicates esophageal temperature probe. B -Visualization of cryoballoon with intracardiac echo; C-CartosOund contours (COR imaging); D- Contours drawn on the cryo balloon yellow and blue lines represent non ablating and ablating segments respectively.
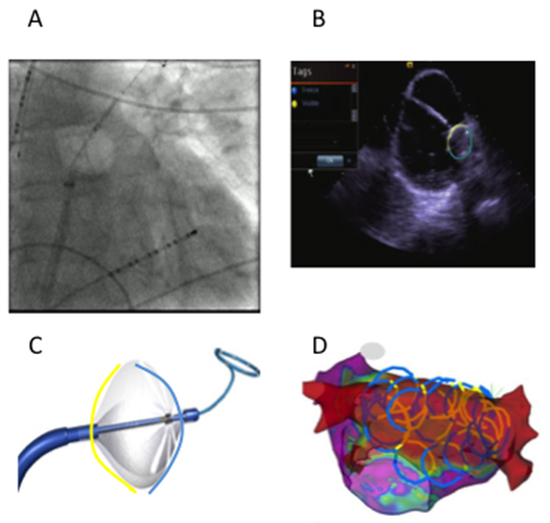
Patients in group HABL underwent surgical ablation: bilateral video assisted thoracoscopic surgery (VATS) with bipolar PVI (Atricure Inc), LA roof/floor lines, RA bicaval line, ablation of ganglionic plexi/the ligament of Marshall, and LA appendage clipping. After recovery from surgery, the patients returned for electrophysiology study/ablation for confirmation of PVI, PWI and additional ablations if required to achieve this as well as ablation of clinical or inducible atrial flutter.
Group CRAconsisted of patients who had received conventional endocardial ablation including CPVA for antral PVI with the addition of empiric linear lines in the LA roof and MVI with thermo-coolRF catheter. Confirmation of bidirectional block was documented for linear lesions. Inducible flutterswere targeted for further ablation.
Patients were seen post ablation at 1 week, 3 month, 6 month and 1 year and were monitored by ECG, Holter or event monitor or through existing implantable cardiac device. All patients discontinued antiarrhythmic therapy 8-12 weeks after the procedure; continuation of anticoagulation was based on their CHADS 2 VASC score.
Primary outcomes were first recurrent AF> 30 seconds, Atrial flutter (AFL)> 30 seconds or any atrial arrhythmia (AF or AFL) at one year, after the three month blinding period. We also analyzedantiarrhythmic drug utilization, referral for repeat ablation, peri-procedural complications, length of stay and control of AF at one year follow up. Procedural time was as recorded in the Apollo LX (version 6.2.0) records and includes time from femoral venous access to the patient exiting the electrophysiology laboratory. If patients remained in sinus rhythm after the initial recurrence assisted by cardioversion or antiarrhythmic drug, they were considered to have controlled AF.
STATA 13.1 was used for all statistical analyses. Continuous variables are expressed as mean + SD or SE; categorical variables are presented as percentages, comparisons were performed with t-test and χ2 test respectively. Differences between three groups were assessed using ANOVA for continuous variables, and Kruskal -Wallis for nominal variables. Adjustment of comorbidities for the clinical outcomes were performed with models as: (1) demographics- age and sex; (2) consisted of cardiovascular risk factors- hypertension, diabetes, coronary artery disease/peripheral vascular disease , transient ischemic attack / stroke, heart failure; (3) included body mass index (BMI), chronic obstructive pulmonary diseaseand obstructive sleep apnea; (4)LA dimension and left ventricular ejection fraction. Kaplan Meier survival analysis was used to compare freedom from AF, or AFLamong the groups; p-value < 0.05 was considered significant.
([Table 1]) There were61 patients ([Table 1])with persistent AF who fit the inclusion criteria; mean duration of AF was 1.3 ± 0.4 yrs. Majority was anticoagulated,without significant differences among the three groups, however more patients in group HABL had long standing persistent AF.
Table 1. Baseline characteristics
|
Cryo (n=24) |
Hybrid (n=14) |
Minimaze (n=23) |
P value |
| Age (years) |
66 ±10 |
70 ± 6 |
63 ± 9 |
ns |
| Sex (male %) |
70.8 |
71.4 |
61 |
ns |
| Hypertension (%) |
71 |
57 |
61 |
ns |
| Diabetes (%) |
37.5 |
7.1 |
17.4 |
ns |
| Coronary artery disease (%) |
25 |
28.6 |
26.1 |
ns |
| Heart failure (%) |
29.2 |
28.6 |
34.8 |
ns |
| Sleep apnea (%) |
37.5 |
35.7 |
22 |
ns |
| Body mass Index |
33 ± 6 |
29 ±5 |
29 ±5 |
ns |
| Cerebrovascular disease (%) |
4.3 |
none |
none |
ns |
| Chronic obstructive pulmonary disease (%) |
8.3 |
14.2 |
4.3 |
ns |
| CHADS2VAsc |
2.4 ±1.3 |
2.5 ±1.5 |
2.3 ±1.5 |
ns |
| Newer anticoagulant /warfarin (%) |
66.7/29.2 |
42.8/50 |
30.4/69.5 |
ns |
| Antiarrhythmic drugs (class III/I) |
33.3/29.2 |
14.3/14.3 |
30.4/21.7 |
ns |
| Left atrium (cm) |
4.5 ± 0.67 |
4.2 ± 0.64 |
4.3 ±0.6 |
ns |
| Left ventricular ejection fraction(%)‡ |
53 ± 10 |
59 ± 6 |
54 ± 10 |
ns |
| Long standing Persistent AF (%) |
25 |
78.6 |
13 |
<0.0001 |
| Prior endocardial ablation (%) |
33.3 |
43 |
21.7 |
ns |
| Alcohol use |
46 |
57 |
65 |
ns |
| Smoking |
54 |
36 |
22 |
ns |
Procedural characteristics
([Table 2]) All patients had CPVA to accomplish PVI, more than half the patients utilized magnetic robotic navigation in HABL and CRA. There was more MVI in HABL and CRAvs CRYO (p= 0.001); there was no difference in cavo-tricuspid isthmus (CTI ablation). More patientsin HABL converted to SR during ablation compared to other groups; there was no difference in conversion to AFL. Majority of the patients had inducible AFL, without differences among the groups. Electrophysiology procedure time was longest at 321± 98 min in CRA, (p=0.02) without a significant difference in fluoroscopy time or Dose Area Product among the groups.
In CRYO, the total number of cryo applications to achieve PVI and PWI was 23.5± 5 for total 3675±776 seconds. Esophageal temperature nadir was -31± -2.6°C. Additional RF ablation was necessary to close small gaps for PWI in 46% of patients with mean RF time 136 ±42 sec (SE). Common locations for PWI gaps were in the mid floor and mid roof. No additional RF ablation was necessary to complete PVI. Additional ablations in LA for inducible AFL were required in 70%. COR imaging had a reduction in the procedure time (257 ± 52 vs 270 ± 59 min, ns), and fluoroscopy time (20 ± 5 vs 26 ± 10, p=0.08), and a reduced need for PW touch up with radiofrequency ablation catheter (33% vs 67%, ns) compared to those without COR imaging.
InHABL, EP/ablationwas performed 75 ± 26 (SE) days after VATS maze, when 59% of the PWarea had low voltage at baseline representing the extent of PWI due to surgery. Thegaps noted in roof and mid floor were closed (radiofrequency time 2754 ± 1572 sec) to accomplish > 90% area of low voltage in the PW [Figure 2b] and [Figure 2C]; due to gaps in PV, ablation for PVI was required in 43%.Four patients presented for the second stage in AF, three patients in AFL, which terminated on ablation. Although not statistically significant, there were more inducible AFL (mean 1.8 ± 0.8) in this group localized to CTI, MVI, and roof requiring additional LA ablation in all the patients.
All CRA patients had CPVA and roof line; 87% had MVI line with 95% achieving bidirectional block. Induced AFL was treated by activation mapping and ablation in 65% of the patients with a total of 5355 ± 1666 sec ablation time in this group.
Figure 2. A shows left atrial posterior wall voltage mapping with dense homogeneous isolation of the pulmonary veins and posterior wall. B shows voltage mapping (0.05-0.5mV) following surgical ablation (pre-endocardial). The bilateral pulmonary veins are isolated and posterior wall is partially isolated with gaps in floor line. C is a different hybrid ablation patient also pre-endocardial ablation; as compared to middle panel posterior wall is densely isolated. All voltage maps 0.05-0.5mV.
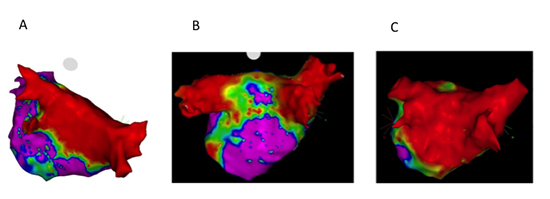
Table 2. Comparison of procedural characteristics:
|
Cryo ablation
(n=24)
|
Hybrid ablation n=14) |
Conventional ablation n=23) |
P value
(three group)
|
P value Group (Cryo vs Hybrid) |
P value (Cryo vs conventional ablation) |
| Procedure time (min) |
262 ± 54 |
208 ± 82 |
321 ±98 |
0.02 |
0.04 |
0.01 |
| Fluoroscopy time (min) |
22.5 ± 8 |
15.3 ± 9 |
20 ± 17 |
<0.0001 |
ns |
ns |
| DAP*dose |
3302 ±1912 |
3045 ±4306 |
2450 ±2719 |
0.003 |
ns |
ns |
| Magnetic Robotic navigation (%) |
4.2 |
57.1 |
74 |
0.0001 |
0.0002 |
<0.0001 |
| Mitral valve isthmus ablation (%) |
46 |
92 |
87 |
0.001 |
0.004 |
0.003 |
| Mitral Valve isthmus block (%) |
80 |
100 |
95 |
ns |
ns |
ns |
| CTI†ablation (%) |
71 |
71 |
74 |
ns |
ns |
ns |
| Additional Posterior wall ablation (%) |
45.8 |
7.1 |
- |
- |
0.01 |
- |
| Additional ablation for PVI ‡(%) |
none |
42.9 |
13 |
0.003 |
0.001 |
0.07 |
| Conversion to sinus rhythm during ablation (%) |
8.3 |
50 |
22 |
0.01 |
0.004 |
ns |
| Conversion to Atrial flutter during ablation (%) |
16.7 |
14.3 |
35 |
ns |
ns |
ns |
| Inducible atrial flutter (%) |
71 |
93 |
65 |
ns |
ns |
ns |
*DAP- Dose area product, † CTI- cavotricuspid isthmus, ‡ PVI- pulmonary vein isolation
Recurrent AFoccurred in 24.6%, recurrent AFL in 21.3%, any atrial arrhythmia in 31% and repeat ablation in 16.3% of the entire cohort. Age, sex, hypertension, diabetes, cardiovascular disease, prior stroke, heart failure, chronic pulmonary obstructive disease, sleep apnea, smoking and alcohol use did not predict recurrent arrhythmia. Patients with recurrent AF had lower BMI 27.8 ±3 (vs 31.4 ±6.3, p=0.05) and27% had coronary artery bypass surgery (vs 4%, p=0.01)compared to those without recurrence. Recurrent AFL was not predicted by any of these demographics or comorbidities.
Comparison of group P to group CRA
([Table 3]) Since CRYO and HABL had PWI, they were together (group P) and compared to CRA. Group P had less recurrent AF (10.5% vs 47.8%, p=0.008), any atrial arrhythmia (23.7 vs 52.2%, p=0.02), but no difference in AFL [Figure 3B],[Figure 3D]; there was less repeat ablation (5.3% vs 30.4%, p=0.007) and more control of AF (95% vs 73%, p=0.02), compared to CRA. There was no difference in time to recurrence in both these groups.
Figure 3. Kaplan Meier survival estimates of freedom from recurrent AF (A and B) and Atrial flutter (C and D). A and C: comparison of three groups- cryo ablation (red line), hybrid ablation (blue line), and conventional radiofrequency ablation (green line). B and D: Comparison of posterior wall isolation (red line) vs conventional radiofrequency ablation (blue line)
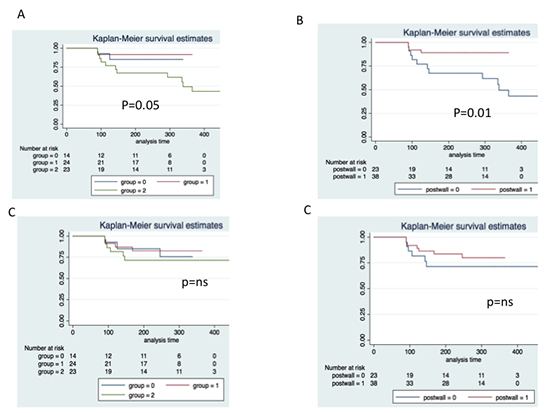
Table 3. Clinical outcomes
|
Cryo ablation
(n=24)
|
Hybrid ablation
(n=14)
|
Conventional ablation (n=23) |
P value |
PWI*-yes |
PWI*-no |
P value |
| Atrial arrhrythmias < 3 months after ablation (%) |
25 |
28.6 |
39.1 |
ns |
26.3 |
39.1 |
ns |
| AF†>3 months (%) |
8.3 |
14.3 |
47.8 |
0.005 |
10.5 |
47.8 |
0.008 |
| Atrial flutter > 3 months(%) |
16.7 |
21.4 |
26.1 |
ns |
18.4 |
26 |
ns |
| Any atrial arrhythmia |
28.6 |
20.8 |
52.2 |
0.03 |
23.7 |
52.2 |
0.02 |
| Control of AF (%) |
95.8 |
93 |
72.7 |
0.001 |
94.7 |
72.7 |
0.02 |
| Cardioversion <3 months after ablation (%) |
8.3 |
7.1 |
13 |
ns |
7.9 |
13 |
ns |
| Cardioversion > 3 months |
12.5 |
None |
8.7 |
ns |
7.9 |
8.7 |
ns |
| Repeat ablation within one year |
8.3 |
None |
30.4 |
0.01 |
5.3 |
30.4 |
0.007 |
| Antiarrhythmic Drug class I |
4.2 |
None |
8.7 |
ns |
2.6 |
8.7 |
ns |
| Antiarrhythmic Drug class III |
21 |
None |
30 |
ns |
13 |
30.4 |
ns |
| Follow up (days) |
347 ± 70 |
373 ± 48 |
381 ± 59 |
ns |
357 ± 63 |
381 ± 59 |
ns |
| Time to recurrence (days) |
245 ± 87 |
254 ± 90 |
269 ± 129 |
ns |
248 ± 87 |
269 ± 128 |
ns |
* PWI- Posterior wall isolation; †AF- Atrial fibrillation.
Adjustment for co-morbidities
AF recurrence remained less for group P (< two times less likely) compared to CRA, after adjusting for models one (p=0.01), two (p=0.03) and three (p=0.003), model four (p=0.01) and duration of AF (p=0.002). Please see methods section for details regarding the models. After adjustment of individual comorbidities of hypertension (p=0.005), diabetes (p=0.005), coronary artery disease/ peripheral vascular disease (p=0.005), heart failure (p=0.004) and TIA/ stroke (p=0.001), the recurrent AF remained lower by multivariate logistic regression analysis.
Comparison of three groups
[Table 3] For a follow-up of 366± 62 days AF recurrence was highest in CRA compared to CRYO and HABL (p=0.005), [Figure 3A] without a difference in recurrent AFL among the groups.[Figure 3C]Occurrence of any atrial arrhythmia was highest in CRA mainly due torecurrent AF; there was no difference in cardioversion among the groups. While repeat ablation was not required in HABL (who already had the second ablation as a planned procedure), there were more repeat ablationsand antiarrhythmic drug use use in CRA compared to other groups.
Length of stay was longest in HABL(145 ± 53 hours) as it included both stages vs CRYO (42 ± 58 hours, p<0.0001) or CRA (38 ± 25 hours, p<0.0001). At the end of follow up control of AF was achieved in 95.8, 93% and 72.7% (p=0.001) of the patients in CRYO, HABL and CRA respectively.
Conversion to sinus rhythm or AFL, inducibility of AFL, achievement of MVI block, CTI ablation and prior ablation did not predict recurrence of AF or AFL. Robotic magnetic navigationused predominantly in HABL and CRA had higher risk for recurrent AF 40% vs 14.3% in manual ablation cases (p=0.02). Patients with PWI were 1.9 times less likely to have recurrent of AF (p=0.002) after adjustment for robotic magnetic navigation catheter utilization.
In CRYO, small hematoma and transient phrenic nerve palsy resolving by hospital dischargeoccurred each in one patient. In group HABL, therewas drug reaction in onepatient, abdominal ileus in one patient, pleural effusion in two patients, and pneumonia in three patients one of whom died within one year post procedure due to non cardiac problems. In CRA, two patients had prolonged hospitalization (> 23 hours) for heart failure exacerbation one of who also had pneumonia. Other complications such as atrial-esophageal fistula, CVA, pericardial effusion/ tamponade, or peri-procedural mortality did not occur in this cohort.
The main finding in our study is the improved efficacy of PWI compared to conventional ablation in patients with persistent AF regardless of the method of ablation. Of the two methods used to isolate the posterior wall,CRYO had less complications,required less number of procedures and had less total length of stay to achieve the same result compared to HABL.
With wide area of antral CPVA, gaps in the LA at roof or MVI can be a source of recurrent atrial arrhythmias, which have been targeted for ablation.[5,6]Triggers in the PV antrum, PW and non PV locations have been shown to be significant source for target inpersistent AF.[7]However, additional ablations beyond CPVA for persistent AF has been questioned after the results of the STAR-AF II trial. [8] A meta analysis of PVI plus substrate ablation vs PVIand a recent randomized trial of PVI vs PWI + PVIhas revealed superiority of PVI plussubstrate strategy compared to CPVA.[9,10]
LA PW is known to have a shared embryology and histology with the pulmonary veins and as such may represent similar substrate to that of the PVs.The demonstration of complete PWI by mapping is important to confirm absence of gap.[11] Our findings are consistent with this concept in that regardless of CRYO or HABL, PWI had higher success rate to prevent recurrence and for control of AF. Though any recurrence > 30 sec is considered a failure of ablation, we have also included “control of AF” as an alternative outcome for those who have persistently been in AF.
A hybrid approach to ablation in the atria can replicate the original cox maze IV surgery.[12]Mid term follow up after such an approach has shown that about two thirds of the patients maintain sinus rhythm without the need for antiarrhythmic drugs leading an increasing interest in this procedure.[13]
Due to limitations ofthe conventional ablation for persistent AF[8], and thepotential replication of Cox maze IV with hybrid approach, we started performing the latter for long standing persistent AF in our institution.When these patients were brought to electrophysiology laboratory for the second stage, mapping revealed gaps in roof and floor lines, which required ablation to achieve PWI; gaps in PV also required re-ablation consistent with prior investigation.[14]Sinus rhythm was maintained in 86% off antiarrhythmic drugs, without repeat ablation. While this experiencewas encouraging, our patients reported significant intercostal pain, had longer length of staywith other morbidities.Persuaded by the early reports of ability to close roof gaps with cryoballoon[4], we started using this approach to isolate PW in patients with persistent AF. We found that the total area of the antra and posterior wall was more extensively and homogeneously ablated with the cryoballoon.Potential reentry circuits due to non homogenous scaras noted in MRI studies[15], perhaps were eliminated contributing to the success in maintaining sinus rhythm.
Our results in group P are similar to a recent investigation comparing PVI to PWI + PVI;[10] our patients have similar antiarrhythmic drug utilization during follow up. We had more cryo-ablation time as we performed two freezes for every PV; we also found from early experience that a cold temperature of <-30o C and a duration of >180 seconds was needed to avoid gaps in the LA roof and floor if nadir <-300 C occurred later than 30 sec after onset of freeze. We stopped ablation if esophageal temperature dropped to <29o C. This was arbitrarily chosen as we noted up to 3-4o C reduction after cessation of freeze. Although esophageal temperatures up to 15o C have been recommended vs lower nadir, they do have incidence of esophageal ulcer.[16]Since we only have the conventional esophageal probe [Figure 1A] at our institution, and could miss a wider area of collateral damage, we chose a higher nadir for safety reasons. Locations of drop in esophageal temperature were posterior wall or pulmonary veins in proximity to the esophageal temperature probe.
Despite additional ablation targets in HABL, the clinical outcomes at one year wassimilarto CRYO group, reinforcing the significance of posterior wall for thepathogenesis ofpersistent AF. Surgical group also had LAA clipping which would electrically isolate the LAA. While the significance of this step is unclear from our study, other investigators have shown it to be relevant to prevent recurrence in persistent AF.[7,17]The benefit of LAA isolation beyond complete homogenization of posterior wall remains to be investigated. Less ablation of MVI in CRYO group but similar results to HABL and better results than CRA reiterates the need for MVI only if there is inducible mitral flutter.
We used CARTO mapping system for voltage and activation map of the left atrium and the atrial flutters. Even though pentarray catheter added to the cost, the dense multipolar map created by this catheter helped reduce the time needed to diagnose and ablate atrial flutters; the latter also justified utilization of radiofrequency ablation catheter in addition to cryo balloon. Overall, the shorter procedure time and reduced EP lab utilization time can be cost effective and warrants further study.
To our knowledge, ours is the first study to compare surgical or endocardial PWI vs conventional ablation for persistent AF and first to report the benefit of PWI regardless of the method of ablation.
Our study is small, from a single center and has limitations attributable to retrospective analysis. Besides this, historical control is limited by selection and lead time bias. Future large multi-center randomized controlled trials regarding the efficacy of PWI using CB forpersistent AF compared to surgical or RF ablation would be a valuable area of future research.
Complete PWI is safe and feasible with cryoballoon ablation; the efficacy of cryothermal PVI/PWI is comparable in our experience to that of hybrid surgical technique,which replicates Cox maze IV, with additional advantage ofshorter length of stay and less morbidity. PWI is superior to conventional ablation as defined in this study in patients with persistent AF for recurrence and control of AF.