Pulmonary and Paradoxical Embolism after Slow Pathway Ablation: A Thrombotic Disorder Unmasked by a Rare Complication.
Eiad Habib1, Amani Elshaer1, Azam Shafquat1,2, Bandar Al-Ghamdi1,2
1Heart Centre, King Faisal Specialist Hospital and Research Centre (KFSH&RC).2Alfaisal University, Riyadh, Saudi Arabia
.
Cardiac electrophysiology study (EPS) and catheter ablation procedure are established diagnostic and therapeutic procedures for cardiac arrhythmias.
Pulmonary embolism (PE) is a relatively rare but potentially fatal complication of Cardiac electrophysiology study (EPS). The paradoxical embolism (PDE) occurs due to an intracardiac defect with a right to left shunt with patent foramen ovale (PFO) being the most common cause. The simultaneous occurrence of PE and PDE is rare.
Here we present a case of PE and PDE after EPS and radiofrequency catheter ablation (RFCA) of the slow pathway in a patient with recurrent supraventricular tachycardia (SVT) due to atrioventricular nodal reentry tachycardia (AVNRT). To our knowledge, such a case of PE and PDE has not been reported after SVT ablation.
Key Words : Pulmonary embloism, Pardoxical emblism, Electrphysiology study.
Correspondence to: Bandar Al-Ghamdi, MD, FACC, FESC.FHRS, CCDS
Heart Centre, King Faisal Specialist Hospital and Research Centre, MBC-16, PO Box 3354, Riyadh, 11211 Saudi Arabia
Cardiac electrophysiology study (EPS) is an established diagnostic and therapeutic procedure for cardiac arrhythmias.[1] The catheter ablation procedure is a well-established treatment option for patients with supraventricular tachycardia (SVT) and has a high efficacy rate.[2] EPS procedure-related major complications occur in 1.1% of patients depending on the procedure type, patient’s age, and concomitant comorbidities. However, a higher complication rate of 3.1% is reported in patients undergoing radiofrequency catheter ablation (RFCA).[3] EPS major complications may include vascular damage, deep venous thrombosis (DVT), pulmonary embolism (PE), systemic embolism or systemic/ paradoxical embolism (PDE), AV block, cerebrovascular accident, tamponade, cardiopulmonary arrest, and rarely death. EPS minor complications include bleeding, hematoma, pericardial effusion with no hemodynamic compromise, and arteriovenous fistula.[3] The simultaneous occurrence of PE and PDE is rare. [4,5,6]
Here we present a case of PE and PDE after EPS and RFCA of the slow pathway in a patient with recurrent SVT due to atrioventricular nodal reentry tachycardia (AVNRT). To our knowledge, such a case of PE and PDE has not been reported after SVT ablation.
Our patient was a 25-year-old woman with a history of recurrent SVTs for several years presenting with intermittent palpitations. She had 3-5 episodes of palpitations every year. She had a documented narrow QRS tachycardia at 200 beats per minute (bpm) with no visible P waves that terminated with IV adenosine. Her past medical history was negative. Management options, including medical therapy versus EPS with RFCA with the exploration of the risks and benefits of each approach were discussed with the patient, and she decided to proceed with EPS and RFCA.Her physical exam was normal.
Her labs, including complete blood count (CBC), renal profile, hepatic profile, and Prothrombin Time (PT)/ Partial Thromboplastin Time (PTT), were normal. Hepatitis B and C and Human immune deficiency virus (HIV) 1 and 2 antibody screening were negative. The baseline electrocardiogram (ECG) was normal. Echocardiogram was normal with no visible patent foramen ovale (PFO) or evidence for an atrial septal defect.
The EPS procedure was performed with three venous accesses via the right femoral vein utilizing one 7, and two 6 French, introducer sheaths. Three catheters were placed in the right ventricular apex (RVa), coronary sinus, and His bundle positions. The sheaths were connected to heparinized normal saline flush. Her baseline intervals were normal, and SVT was easily inducible with programmed electrical stimulation. The SVT was consistent with a typical (slow-fast) (AVNRT). Slow pathway ablation was performed successfully. The procedure was then completed, and the catheters and sheaths were removed from the patient's body after a careful aspiration of all sheaths. The total duration of the procedure was 70 minutes. The patient was transferred to the cardiac holding unit (CHU) in stable condition with an order for bed rest for four hours Seven hours later, the patient became dizzy, and her level of consciousness deteriorated. On physical examination, she was conscious and oriented but lethargic. Her BP was 66/40 mmHg,Temperature 36.7 C, and SpO2 97 to 100% on room air. The cardiovascular and neurological exams were normal. Apart from sinus tachycardia at 109 bpm, her ECG was normal. Portable chest X-ray showed no significant abnormalities.
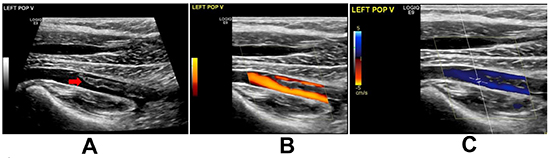
A pocket-sized ultrasound device (Vscan) study was immediately conducted to rule out pericardial effusion and tamponade. However, it revealed a mass in the right atrium (RA) and dilated RV. An urgent bedside echocardiogram revealed a large mass (16x17mm) likely thrombus in the RA crossing a PFO to the left atrium (LA) with filamentous thrombus and crossing the mitral valve. It was suggestive of PE with mild dilation of RV and elevated RV systolic pressure at 40-45 mmHg. The RA and (LA) were normal in size. The inferior vena cava was dilated and not collapsible. [Figure 1] The patient was transferred to the coronary care unit, and high dose heparin was initiated along with IV fluid resuscitation. The patient responded well to IV fluids with normalization of her BP. A whole body computed tomography (CT) scan was performed. The CT brain was clear, but the CT-PE study revealed a massive bilateral acute PE. [Figure 2A] Besides, there was a partially visualized filling defect within the right renal artery branch, associated with multiple bilateral renal wedge-shaped hypodensities, compatible with multiple renal infarctions and very indicative of embolic showering. [Figure 2B] However, the kidney function remained normal. Lower limb ultrasound was performed, and it showed a non-obstructive thrombus in the left popliteal vein. [Figure 3] Blood samples were sent for thrombophilia work up and autoimmune studies.
On the following day, repeated echocardiography revealed that the thrombus in the LA was large and highly mobile. It was felt that the safest approach would be to remove the LA thrombus surgically, so the patient was transferred to the operating room after signing informed consent. However, the pre-operative transesophageal echocardiogram (TEE) about 6 hours from the last transthoracic echocardiogram revealed the disappearance of the RA and LA thrombus. There were no clots within the RV, the main PA or the proximal right and lefts PAs. With these findings, the procedure was aborted, and the patient was returned to her bed in a stable condition. She was therefore started on oral anticoagulation. The patient was hemodynamically stable and discharged home a few days later.
The thrombophilia workup showed that her functional Protein C level was low at 0.5 IU/mL (normal 0.7-1.3 IU/mL), as was her Factor V activity which was 0.53 IU/mL (normal 0.6-1.5 IU/mL). Her functional protein S level was within the normal range. Genetic analysis was carried out and showed that the patient was heterozygous for C677T polymorphism.
Follow up echocardiogram a few months later showed normal LV and RV size and function and normal right-sided pressure.
The patient was maintained on oral anticoagulation with follow up in thromboembolic service. She was asymptomatic at 12 months follow up.
Figure 1. Echocardiogram (2D): A. Apical four-chamber view (A4C) showing right atrial mass crossing patent foramen ovale to the left atrium.B. Short axis view (SAX) at aortic valve level showing the right atrial mass measuring 16x17 mm. C. Parasternal long axis view (PLA) showing the mass protruding through the mitral valve.
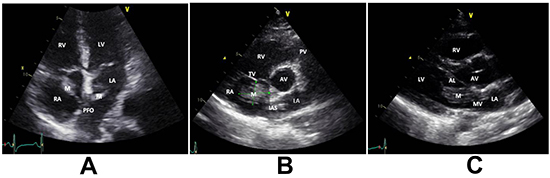
AL; anterior leaflet of the mitral valve, AV; aortic valve, LA; left atrium, LV; left ventricle, M; mass, MV; mitral valve, RV; right ventricle.
Figure 2. A. Computed tomography (CT)scan, PE study with axial cut showing distal filling defects in right and left pulmonary arteries(red arrows)RPA and LPA. B. Right renal artery with partial occlusion likely thrombus.
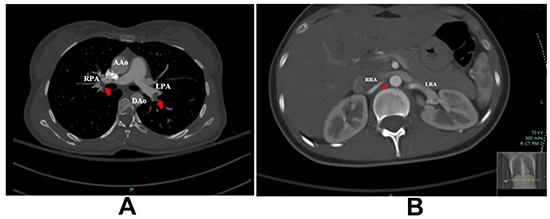
AAo; ascending aorta, DAo; descending aorta, LPA; left pulmonary artery, RPA; right pulmonary artery, LRA; left renal artery, RRA; right renal artery.
Figure 3. Ultrasound venous Doppler Scan of the left lower limb showing acute nonobstructive thrombus in the left popliteal vein (red arrow).
This case demonstrated a rare complication of EPS and RFCA with simultaneous PE and PDE.
Predisposing factors for thromboembolic events such as deep venous thrombosis (DVT) in patients undergoing EPS include placement of the venous sheath(s) leading to vascular endothelial injury and stasis.[7] The placement of multiple femoral venous sheaths could be a contributing factor. In one study, the placement of multiple femoral venous sheaths was associated with a high incidence of non-occlusive DVT (19.2%) detected through duplex ultrasonographyembolized.on the next day following the procedure. However, none of these patients experienced symptomatic PE.[8] Another study reported a significant increase in the incidence of femoral vein thrombosis following the use of multiple femoral venous sheaths without administering heparin in patients who underwent EPS (62.5%); however, fixed prophylactic dose with body weight independent low molecular weight heparin significantly decreased the risk of femoral thrombosis in these patients (18%).[9] Similar to the previous study, none of the patients showed clinical features of PE.[9]. The catheters manipulation in blood vessels and cardiac chambers is another risk factor with activation of the coagulation cascade.[7] A thrombus may develop inside the sheath(s) or on the surface of the catheter(s) and attach to the vessel walls, valves or cardiac chambers, and then embolize. Besides, post-procedure compression of the puncture site(s), subsequently leading to venous stasis and an accentuated thrombogenic state.[7] Bed rest with immobilization post-procedure is another contributing factor.
PE is a relatively rare but potentially fatal complication of EPS. The overall incidence of PE with reviewing five studies, including 14,205 patients, underwent EPS was 0.12%.[10] Although the incidence of PE post EPS is low, it carries a mortality rate of greater than 15% during the first three months after diagnosis.[11] The risk of systemic embolism and PDE post EPS and RFCA is less established. PE after routine right cardiac catheterization is likely underestimated.[12] In one study with a small sample size of 57 patients, 12% of the patients had new perfusion defects consistent with PE, which were detected by ventilation-perfusion lung scans just one day after right-heart catheterization.[12] This finding suggests that PE may be more common than previously anticipated.
The PDE occurs due to an intracardiac defect with a right to left shunt. The most common intracardiac defect associated with PDE is the patent foramen ovale (PFO) defect with a venous embolus that crosses the PFO to the systemic circulation. [3-6] Patients with PFO are usually asymptomatic. However, if RA pressure increases, a right to left shunt occurs with risk for PDE. Our patient had elevated right heart pressure following the acute PE and resulted in the PDE. The Brain arteries are the most frequent localization of PDE.[6] However, embolization may happen in any other systemic (not systematic)arteries.
On the full thrombophilia workup, our patient was discovered to have a heterozygous C677T polymorphism, which is an additional risk factor. The C667T genetic mutation is the most common methylenetetrahydrofolate reductase (MTHFR) mutation variant. It is correlated with reduced enzyme activity, which in turn leads to elevated levels of homocysteine. High levels of homocysteine showed an increased risk for atherosclerosis, which may result in a heart attack and/or stroke, and DVT. [13] This polymorphism may be present either in a homozygous or heterozygous state. In the heterozygous, there is reduced enzyme function to about 65% of normal, and in homozygous there is only 30% of normal enzyme function. [13] This mutation is extremely common in certain ethnic and geographic populations. [13] In Saudi population, the presence of the C677T heterozygous and homozygous variants are 25.25% and 2.25% respectively. [14] The association of C677T polymorphism with the development of Venous thromboembolism (VTE) is not universally accepted. [13] A recent meta-analysis supported the association of MTHFR C677T polymorphism with VTE risk. [15] Our patient was also found to have a low level of Factor V and Protein C. A low level of Protein C was shown to be an independent risk factor for VTE in multiple studies, and it predisposes patients to recurrent VTEs.[16] The patient did not have a factor V Leiden or G20210A prothrombin gene mutation.
With the exclusion of atrial flutter, routine anticoagulation therapy is not routinely recommended before, during, and after a right-heart procedure unless other risk factors for systemic embolism are present.[7] However, There is a need for further evidence-based guidelines concerning anticoagulation and prevention of VTE during and after routine EPS procedures. Risk stratification and early intervention for such cases, along with DVT prophylaxis when indicated, may decrease the incidence of VTE and its complications.
The authors declare no conflict of interest related to this manuscript.
DVT is not uncommon after EPS, and subsequent PE and/or PDE although rare is a serious complication with possibly dramatic consequences. Venous stasis, vascular endothelial injury with activation of the coagulation cascade resulting from multiple venous sheaths, and prolonged hemostatic mechanisms may all contribute to the formation of DVT. PE should be suspected with prompt diagnosis and management in patients who develop acute respiratory distress after a right-heart ablation procedure. There is a need for updating evidence-based guidelines on antithrombotic therapy in the setting of EPS procedures.