Sympathetic Denervation for Treatment of Ventricular Arrhythmias
Heajung L. Nguyen1, Marmar Vaseghi1
1UCLA Cardiac Arrhythmia Center, David Geffen School of Medicine at UCLA, Los Angeles, CA.
Ventricular arrhythmias are a major cause of morbidity and mortality in patients with heart disease. A growing understanding of the cardiac autonomic nervous system’s crucial role in the pathogenesis of ventricular arrhythmias has led to the development of several neuromodulation therapies. Sympathetic neuromodulation is being increasingly utilized to treat ventricular arrhythmias refractory to medical therapy and catheter ablation. There is a growing body of preclinical and clinical evidence supporting the use of thoracic epidural anesthesia, stellate ganglion blockade, cardiac sympathetic denervation, and renal denervation in the treatment of recurrent ventricular arrhythmias. This review summarizes the relevant literature and discusses approaches to sympathetic neuromodulation, particularly in the management of scar-related ventricular arrhythmias.
Key Words : Autonomic Modulation, Sympathetic Denervation, Ventricular Tachycardia.
Correspondence to: Marmar Vaseghi, MD PhD
UCLA Cardiac Arrhythmia Center
100 Medical Plaza, Suite 660
Los Angeles, CA 90095-7392
Sympathetic hyperactivity plays a critical role in the genesis and perpetuation of ventricular arrhythmias (VA) in patients with heart disease 1. Our understanding of the complex relationship and interactions between the substrate and autonomic nerves that lead to VA has grown significantly over the last few decades, with tremendous growth of interest and advancements in preclinical and clinical research. Sympathetic modulation has become an increasingly valuable target for the treatment of refractory VAs. Temporizing bedside procedures, such as thoracic epidural anesthesia (TEA) and percutaneous stellate ganglion blockade (SGB), may be particularly useful in hemodynamically unstable patients, where catheter ablation or surgical cardiac sympathetic denervation (CSD) may not be immediately feasible. More permanent interventions, such as CSD, can be used in addition to catheter ablation and have shown promise in decreasing the risk of recurrent VT/VF in high-risk patients. Renal sympathetic denervation (RDN), which can be used alone or as an adjunct to CSD, has also shown potential benefit for treatment of VT/VF.
This review briefly summarizes the role of the sympathetic nervous system in pathogenesis of VAs, discusses several approaches to sympathetic neuromodulation, especially in the management of scar-related VAs, and provides a review of the rationale and available clinical data for each approach.
Sympathetic Nervous System and Ventricular Arrhythmias
Neural control of the heart consists of several levels of feedback loops carrying a balance of sympathetic and parasympathetic signals between the heart and the peripheral and central nervous systems. Transmission and integration of afferent information at the level of the heart via the intrinsic cardiac nervous system, sympathetic ganglia, spinal cord, nodose ganglia, and brainstem play a key role in controlling efferent autonomic tone to the heart 2,3. With heart disease, neurohormonal activation and neural remodeling disturbs this intricate homeostasis, resulting in overall inhibition of parasympathetic tone and increased sympathetic afferent and efferent outflow to the heart, predisposing to ventricular tachycardia (VT) and ventricular fibrillation (VF) 4. Sympathetic activation leads to release of cardiac norepinephrine, whose electrophysiological role through activation of beta-adrenergic receptors is well-established, as well as sympathetic co-transmitters, including neuropeptide Y (NPY) and galanin. These sympathetic co-transmitters are found throughout the central and peripheral nervous systems with diverse roles in various physiological processes, including mood and appetite as well as cardiac autonomic control. Of the sympathetic co-transmitters, the role of NPY in arrhythmogenesis and heart failure is increasing recognized. NPY mediates vasoconstriction, angiogenesis, and cardiac remodeling in the cardiovascular system 5. Elevated NPY levels are associated with a poorer prognosis and increased mortality both after myocardial infarction and in the setting of heart failure 6,7. While low levels of sympathetic activation leads to predominantly NE, high levels of sympathoexcitation, as can occur in in pathologic conditions such as ST-elevation myocardial infarction, causes the release of NE and NPY from larger, more dense-cored vesicles 8,9. NPY can potentiate the effects of NE, but appears to also act on its own ventricular myocardial receptors to shorten action potential duration (APD) and refractory period, circumventing the effects of beta-blocker therapy and increasing the propensity for VAs 8,10,11. With sympathoexcitation, not only is APD shortened, but dispersion in APDs is also increased. In large animal models, stimulation of either the right or left stellate ganglia can increase dispersion of repolarization in the ventricles. In addition, sympathetic activation can cause early afterdepolarizations (EAD) and delayed afterdepolarizations (DAD) in normal hearts12-17. These changes are exacerbated in diseased hearts, as myocardial injury and inflammation lead to sympathetic axonal injury and denervation. This denervation leads to myocardial denervation supersensitivity and is followed by heterogeneity in sympathetic re-innervation 18-24. Thus, myocardial injury and scar lead to heterogenous sympathetic innervation of the myocardial substrate. In the setting of sympathetic activation, the differences in myocardial conduction and repolarization that exist as result of the mix of viable tissue in areas of myocardial fibrosis are further exacerbated, predisposing to VAs. In addition, decreased cardiac output from myocardial infarction (MI) and heart failure leads to activation of the renin-angiotensin-aldosterone system, which can increase myocardial fibrosis and alter gap-junctions, cause electrolyte abnormalities, and further lead to release of norepinephrine by acting on the prejunctional sympathetic angiotensin II receptors, contributing to the occurrence of VAs 2,25-29.
Thoracic Epidural Anesthesia
TEA is a minimally invasive therapeutic modality for the immediate management of electrical storm. By injecting anesthetic agents into the thoracic epidural space, sympathetic signals carried by neuraxial afferent fibers via dorsal root ganglia and efferent fibers innervating the myocardium can be inhibited Figure 1. By blocking the C8 nerve roots, which forms part of the inferior cardiac sympathetic nerve, and T1-T4 segments containing the majority of cardioaccelerator fibers responsible for controlling heart rate and contractility, TEA has the potential to provide complete sympathetic blockade 30.
Figure 1. Sympathetic Neuromodulation for Ventricular Arrhythmias.
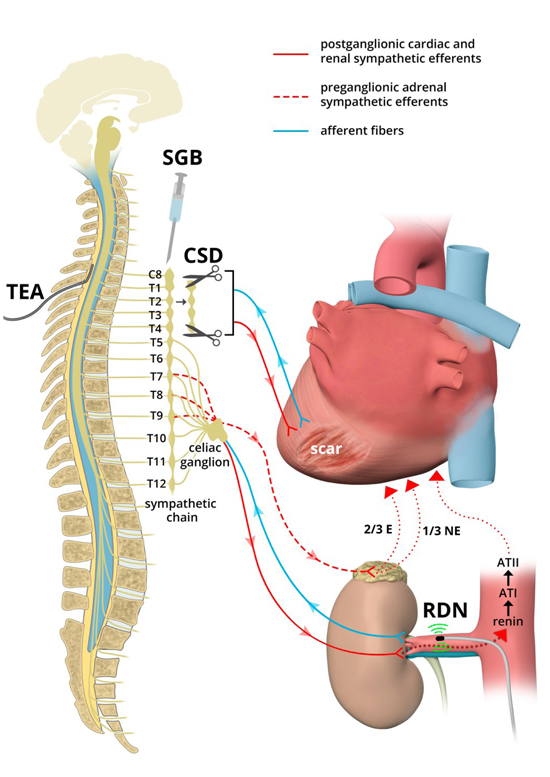
Neuromodulatory therapies targeting multiple levels of the cardiac sympathetic nervous system and cardio-renal-neuraxial pathways for the management of ventricular arrhythmias are shown. Note that these neuromdulatory therapies reduce or block both afferent (blue) and efferent (red) sympathetic fibers. Parasympathetic nerve fibers are omitted for simplicity. ATI/II = angiotensin I/II; CSD = cardiac sympathetic denervation; E = epinephrine; NE = norepinephrine; RDN = renal artery denervation; SGB = stellate ganglion block; TEA = thoracic epidural
Using a sterile technique, TEAs can be instituted at the bedside, though is preferably performed under fluoroscopic guidance. A 17-G epidural needle is inserted into the T1-T2 or T2-T3 interspace using a “loss of resistance” approach. Safe positioning is confirmed with absence of blood or cerebrospinal fluid on aspiration. An epidural catheter is advanced into the epidural space through the needle to the T1 level and is secured in place. Bupivacaine 0.25% or ropivacaine 0.2% are typically administered as a bolus followed by continuous infusion.
TEA has an immediate onset of action and may forestall the need for intubation. The infusion can be titrated to effect (arrhythmic response). In addition, TEA can be safely performed even in critically ill patients, with little to no effect on mean arterial blood pressure, heart rate, cardiac index, or central venous pressure 31.
The antiarrhythmic effects of TEA were initially demonstrated in 1988 in a rodent post- MI model, where TEA significantly decreased the incidence of malignant VAs following coronary artery ligation (ischemia driven VAs) compared to a control group 32. A subsequent canine study in normal hearts showed that TEA causes prolongation of the APD in a cycle length-dependent manner 33. Mechanistic data on the effects of TEA in diseased hearts is sparse. However, in a recent porcine chronic MI model, TEA led to an increase in ventricular effective refractory period (ERP) and ventricular APD, and increased baroreflex sensitivity, indicating an improvement in parasympathetic function, likely due to interruption of sympathetic afferents neurotransmission 34. In this study, a significant prolongation in atrial ERP was also observed after TEA.
Following a case report of TEA successfully suppressing ischemic VT in a patient with refractory electrical storm over a decade ago, there have been a number of small clinical studies demonstrating similar efficacy with a low risk of complications 35. In a case series of patients with both polymorphic and monomorphic VT in the setting of ischemic and non-ischemic cardiomyopathy, 6 of 8 patients who underwent TEA had ≥80% reduction in arrhythmia burden Table 130. Similarly, in a multicenter series of 11 patients who underwent TEA for electrical storm, a complete response (defined as no recurrence of VAs) or partial response (defined as 80% to 99% reduction in VT episodes) was observed in the majority of patients (54%) Table 136. Patients who responded to TEA also tended to respond to intubation and sedation, although additional benefit beyond general anesthesia was observed with TEA in 2 of 3 patients with refractory VT 30,36. Importantly, TEA can allow for discontinuation of sedation and extubation of the patient, resulting in in the added benefit of patient participation in the decision-making process of more definitive therapies, which may include ablation, ventricular assist device, or cardiac transplantation.
Table 1. Clinical Studies on Sympathetic Modulation for Treatment of Ventricular Arrhythmias in Structural Heart Disease
|
Modality
|
Year
|
Citation
|
Study Type
|
N
|
Etiology of CMY
|
Type of VT
|
Follow-Up
|
Main Findings
|
|
Thoracic Epidural Anesthesia
|
|
TEA
|
2017
|
Do et al.
|
Retrospective, multicenter
|
11
|
NiCMY (45%), iCMY (27%), other (28%)
|
VT storm: PMVT (27%), MMVT (73%)
|
Acute
|
45% complete response*, 9% partial response*, 45% no response
|
|
TEA
|
2010
|
Bourke et al.
|
Retrospective, multicenter
|
8
|
iCMY (50%), NiCMY (25%), other (25%)
|
VT storm: PMVT (37.5%), MMVT (62.5%)
|
6.2±4.6 mo
|
≥80% reduction in arrhythmia burden in 6 of 8 patients
|
|
Percutaneous Sympathetic Ganglion Block
|
|
SGB (89% Left)
|
2017
|
Meng et al.
|
Retrospective review
|
38
|
NiCMY (18%), iCMY (45%), Unspecified (29%)
|
Mixed VT/VF (39%), PMVT (32%), MMVT (11%), VF (18%)
|
Hospital d/c (6-28 d)
|
83-95% relative reduction in VA burden
80.6% survived to hospital discharge
|
|
SGB (50% Bilateral, 50% Left)
|
2019
|
Tian et al.
|
Single center case series
|
30
|
iCMY (57%), NiCMY (33%), idiopathic (7%), LQT(3%)
|
VT storm (40%), VT+VF storm (50%), VF storm (10%)
|
72h + f/u of 22±16 mo
|
50% complete response^, 20% partial response^ at 72 hours post TEA. Overall 92% reduction in VA episodes (in patients with ICD)
|
|
Surgical Cardiac Sympathetic Denervation in Structural Heart disease
|
|
LCSD
|
2010
|
Bourke et al.
|
Retrospective, multicenter
|
9
|
NiCMY (22%), iCMY (22%), Sarcoid (22%), HCM (22%), ARVC (11%)
|
VT storm: PMVT (22%), MMVT (78%)
|
6.2±4.6 mo
|
33% complete response+, 22% partial response+,
44% no response
|
|
BCSD (66%), LCSD (34%)
|
2014
|
Vaseghi et al.
|
Retrospective, single center
|
41
|
NiCMY (54%), iCMY (22%), HCM (7%), Sarcoid (5%), other (12%)
|
Refractory VT or VT storm: MMVT (80%), PMVT/VF (20%)
|
367 ± 251 d
|
48% of patients had 1-year survival free of ICD shock following BCSD (30% following LCSD)
90% of patients had reduction in ICD therapies
|
|
BCSD
|
2016
|
Saenz et al.
|
Retrospective, multicenter
|
75
|
Chagasic Cardiomyopathy
|
MMVT
|
7 mo
(IQR1-46)
|
Decrease in ICD shocks from median of 4 (range 2-30) before to 0 (range 0-2) after BCSD
|
|
BCSD (81%) LCSD (19%)
|
2017
|
Vaseghi et al.
|
Retrospective, multicenter
|
121
|
NiCMY (71%), iCMY (27%), Mixed CMY (2%)
|
Recurrent VT or VT storm
|
1.5±1.4 years
|
Reduction in ICD shocks from mean of 18±30 in the year before to 2.0±4.3 after CSD
|
|
BCSD
|
2019
|
Assis et al.
|
Retrospective, single center
|
8
|
ARVC
|
Refractory VT
|
1.9±0.9 years
|
Reduction in ICD shocks/sustained VT (12.6±18.2 to 0.9±1.4)
|
|
BCSD (80%) RCSD (20%)
|
2019
|
Okada et al.
|
Retrospective, single center
|
5
|
Cardiac sarcoidosis
|
Refractory VT
|
26 mo
(IQR 5–29)
|
Reduction in ICD shocks from median of 5 (in the 6 months preceding CSD) to 0
|
|
Renal Sympathetic Denervation
|
|
RDN
|
2014
|
Remo et al.
|
Retrospective, single center
|
4
|
iCMY (50%),
NiCMY (50%)
|
Recurrent VT
|
8.8 mo
(IQR 5-11)
|
Decrease in VT episodes from 11.0±4.2 during the month pre-RDN to 0.3±0.1/month post-RDN
|
|
RDN
|
2015
|
Armaganijan et al.
|
Prospective single center
|
10
|
Chagas CMY (60%), NiCMY (20%),
iCMY (20%)
|
Refractory VA
|
6 mo
(IQR 18 d, 6 mo)
|
Reduction in VT/VF episodes from 28.5 to 1
Reduction in ICD shocks from 8 to 0
|
|
RDN
|
2016
|
Ukena et al.
|
Retrospective, multicenter
|
13
|
iCMY (54%),
NiCMY (46%)
|
Refractory VAs: VF (62%), MMVT (54%), PMVT (46%)
|
12 mo
|
Reduction in VT/VF episodes from median of 21 in the month pre-RDN to 2 and 0 at 1 and 3 months post-RDN
|
|
RDN
|
2016
|
Evranos et al.
|
Propensity score-matched cohort
|
32
|
iCMY (62%),
NiCMY (38%)
|
Refractory VA
|
15 mo
(IQR 6-20)
|
Reduction in VT/VF/ICD therapies in RDN + ablation vs. ablation only group
|
|
RDN
|
2018
|
Jiang et al.
|
Prospective case series
|
8
|
DCM (63%), iCMY (25%), iCMY (12%)
|
VT storm or VA episodes on ICD interrogation
|
15 mo
(IQR 6-30)
|
Reduction in VA episodes from 3.2 to 0.1 per mo
|
|
RDN (10% Left sided only)
|
2019
|
Bradfield et al.
|
Retrospective, single center
|
10
|
NiCMY (90%), iCMY (10%)
|
Recurrent VT/VT storm: MMVT (70%), PMVT (30%)
|
23 mo
|
Reduction in ICD therapies (from 29.5±25.2 to 7.1±10.1) 6 mo pre-to 23 mo post-RDN
|
Follow-up is given as mean ± standard deviation or median (IQR); d = days; mo = months of follow up. *complete response = complete suppression of VT episodes (48 hours after TEA), *partial response (80% to 99% reduction in VT episodes); ^partial response not defined; +complete response = no VA within 1 week of procedure, +partial response = recurrence of VAs that did not fulfill definition of VT storm. ARVC = Arrhythmogenic right ventricular cardiomyopathy; ATP = Anti-tachycardia pacing; BCSD/LCSD/RCSD = Bilateral/left/right cardiac sympathetic denervation; CMY = Cardiomyopathy; CPVT = Catecholaminergic polymorphic ventricular tachycardia; DCM = Dilated cardiomyopathy; HCM = Hypertrophic cardiomyopathy; ICD = Implantable cardioverter defibrillator; iCMY = Ischemic cardiomyopathy; LQTS = Long QT syndrome; MMVT = Monomorphic ventricular tachycardia; NiCMY = Nonischemic cardiomyopathy; PMVT = Polymorphic ventricular tachycardia; RDN = Renal sympathetic denervation; SGB = Stellate ganglion blockade; TEA = Thoracic epidural anesthesia;VF = Ventricular fibrillation; VT = Ventricular tachycardia.
Percutaneous Stellate Ganglion Blockade
Percutaneous blockade of the stellate ganglion with injection of a local anesthetic is an alternative bedside therapy for the acute management of VT or VF Figure 1. Initially described in 1934 for management of chronic pain syndromes, SGB inhibits both afferent and efferent sympathetic signals to and from the heart at the level of the stellate ganglion 37. It can be safely performed under ultrasound guidance alone or with a combination of fluoroscopy and ultrasound. The stellate ganglion can be accessed using a 22G needle at the level of the C6 transverse process. Once contact is made at the transverse process, the needle is slightly retracted before a bolus of a local anesthetic is injected. In contrast to a TEA where a catheter remains in place, percutaneous SG block procedures are repeated as needed by bolus therapy. Therefore, unlike TEA, SGB may also be performed in the setting of anticoagulation or antiplatelet therapy.
In a rabbit post-MI model, left SGB was shown to electrically stabilize the ventricular myocardium by prolonging transmural APD, reducing repolarization heterogeneity, increasing the ERP, and raising the VF threshold 38. Early evidence from case reports and small case series demonstrated that SGB can achieve a significant decrease in arrhythmia burden 39-43. A 2017 systematic review of 38 cases from 23 studies showed a clear reduction in the number of VA episodes and appropriate defibrillator therapies, regardless of the etiology of VT or the presence or absence of structural heart disease (SHD) 44. In 2019, Tian et al 45 described the use of SGB in 30 patients with predominantly SHD (90%) presenting with drug-refractory electrical storm. SGB resulted in VA suppression in 92% of cases, with 60% achieving complete response at 24 hours (Table 1). Similarly, in the latest case series of 20 patients who underwent bilateral SGB for refractory VA at a single center, 45% of patients were free of VAs at 48 hours, irrespective of VA subtype and etiology of cardiomyopathy 46.
TEA and SGB provide opportunities to stabilize the patient while precipitating triggers for and more definitive management of VAs are addressed. These therapies serve as a bridge to catheter ablation, surgical intervention, or consideration for advanced heart failure therapies and/or cardiac transplantation 47.
Surgical Cardiac Sympathetic Denervation
While TEA and SGB are both feasible and effective potential therapeutic options for the acute management of electrical storm, their effects are transient, limited by the pharmacokinetics of local anesthetic agents. Conversely, surgical CSD provides permanent autonomic modulation via resection of the lower half of the stellate (cervicothoracic) and the second through fourth thoracic paravertebral ganglia Figure 1. This results in a reduction in both efferent and afferent sympathetic neurotransmission to and from the heart, while still preserving some sympathetic innervation. Moreover, as CSD entails interruption of preganglionic sympathetic fibers along with removal of a portion of the stellate and thoracic post-ganglionic neurons, there is no regeneration or reinnervation with time, as is observed with sympathetic postganglionic denervation after heart transplantation 48.
Left CSD (LCSD) or bilateral CSD (BCSD) is performed under general anesthesia via video-assisted thoracoscopic surgical approach guided by intraoperative pathology. Three small surgical incisions are made in the sub-axillary area, followed by deflation of the ipsilateral lung. The sympathetic chain is identified under the parietal pleura, and the lower one-half to one-third of the stellate ganglion and the T2 to T4 thoracic ganglia are transected and removed. Major contraindications to CSD include severe chronic obstructive pulmonary disease or pulmonary disease prohibiting single lung inflation, anticoagulation that cannot be interrupted, and acute infection.
Pioneered by Thomas Jonnesco in 1916, the first case of LCSD was performed in a patient with disabling refractory angina pectoris and ventricular tachyarrhythmias 49. Following CSD, he was noted to have complete resolution of not only his chest pain, but also his VAs. Despite this, it was not until the 1960s where interest in the antiarrhythmic potential of CSD re-emerged following reports of successful treatment of recurrent VT with CSD (left sided and bilateral) 50,51. Subsequent studies established the utility of LCSD in primary inherited arrhythmia syndromes, specifically congenital long QT syndrome (LQTS) and catecholaminergic polymorphic ventricular tachycardia (CPVT), in patients unresponsive to beta-blocker therapy 52-58. To date, the largest series of LCSD in LQTS was reported by Schwartz et al 52. In this series, 147 high-risk patients with LQTS, of whom 99% were symptomatic, 48% had a prior history of cardiac arrest, and 75% were symptomatic despite maximal beta-blocker therapy, underwent LCSD. Over a period of 8-years of follow-up, there was a 95% reduction in the mean number of implantable cardiac defibrillator (ICD) shocks and a 91% reduction in aborted cardiac arrests and episodes of syncope after LCSD. The efficacy of LCSD has also been reported in patients with CPVT. Several studies have shown that LCSD can reduce cardiac events and ICD therapies by 89% to 93% in this population 53,59,60. These data support the Class IIa and IIb recommendations for the use of LCSD in patients with LQTS and CPVT, respectively, who suffer from recurrent syncope and ICD shocks despite beta-blocker therapy 61.
More recently, there has been a growing body of evidence supporting the use of bilateral CSD in the setting of structural heart disease (SHD), specifically in patients with ischemic and non-ischemic cardiomyopathy who continue to experience recurrent VAs despite optimal medical therapy (Table 1). Initial case series describing LCSD for electrical storm in patients with SHD showed a modest benefit, with 5 of 9 patients showing clinical response, while a greater response was observed in patients who underwent TEA, which blocks both the left and right cardiac sympathetic fibers 30. This data suggested that bilateral CSD in this population may have more durable effects. Although the functional contributions of the LSG and right stellate ganglion (RSG) to the innervation of the left ventricular (LV) myocardium are not completely understood, innervation of the anterior LV wall by both stellate ganglia was demonstrated in a porcine model using APD and norepinephrine microdialysis measurements 62. Furthermore, Opthof and colleagues 63 observed electrical effects of both RSG and LSG stimulation in 42% of right and left ventricular sites examined in a canine model. An initial study in normal dogs reported increased ectopy with right stellate ganglion cooling (which blocks neurotransmission) in some animals in the setting of left anterior coronary artery (LAD) ischemia, though this study was limited by a significant number of animals that were excluded due to either VF with the first LAD occlusion before block or occurrence of no arrhythmias despite multiple occlusions, as well as lack of consistency of arrhythmias observed in other animals 64. A follow-up study to distinguish the antiarrhythmic potential of right vs. left stellate ganglionectomy in a canine post-infarct model, however, demonstrated that while left-sided ganglionectomy was more effective than right-sided ganglionectomy in reducing incidence of VF, both right and left stellate ganglionectomy independently reduced ischemia-induced VF and improved outcomes 65. Finally, in a porcine model of chronic MI, pathological neural remodeling was observed in both the left and right stellate ganglia, irrespective of infarct location 66. Neurochemical remodeling of both right and left stellate ganglia neurons has been observed in both infarcted pigs and in patients with VT and cardiomyopathy undergoing CSD, with an increase in neuronal adrenergic phenotypes and increased NPY immunoreactivity compared to controls. These studies provided a mechanistic rationale for performing BCSD as a therapy for VAs in patients with structural heart disease, and indeed, BCSD has shown more durable outcomes than a left-sided procedure in this population. In the initial single center study of 41 patients who underwent either BCSD (n=21) or LCSD (n=14) for refractory VT and VF, predominantly in the setting of SHD, those with BCSD appeared to have a longer freedom from ICD shock compared to those with left CSD at one year, despite similar reductions in ICD shock burden 67. This study was followed by a retrospective multi-center study of 121 patients with SHD who underwent either LCSD or BCSD for refractory VT or VT storm and showed a 58% freedom form ICD shocks/sustained VT at one year 68. Of note, patients with BCSD had significantly longer ICD shock-free transplant-free survival than those who underwent LCSD. Longer VT cycle lengths >400 ms also proved statistically significant for arrhythmia recurrence in multivariable analysis. Additional factors associated with the combined endpoint of ICD shock, death, and transplantation after CSD included advanced NYHA class (III-IV), age, diabetes mellitus, chronic kidney disease, and the use of >1 antiarrhythmic drug. Outcomes were similar in patients with ischemic and nonischemic cardiomyopathy. Taken together, these findings suggested that CSD in SHD appears to be most beneficial when performed bilaterally and earlier in the course of cardiomyopathy, before patients develop NYHA Class IV symptoms, and in cases where the clinical VT has a relatively shorter cycle length. A limitation of the above study was the heterogeneous population of patients that presented with both monomorphic and polymorphic VT. Recently, Dusi et al 69 quantified the value of CSD in scar-mediated monomorphic VT in a single center retrospective study, specifically excluding patients that presented with any polymorphic VT. This study showed a >30% reduction in the adjusted time to all VT recurrence rates (including shocks and anti-tachycardia pacing therapy) in patients who had previously undergone at least one VT ablation procedure. Other smaller case series have suggested benefit in specific etiologies of cardiomyopathy, including Chagas disease and cardiac sarcoidosis 70,71.
Mechanisms behind the benefit of CSD in diseased hearts include an increase in APD in infarcted hearts and beneficial changes in the heterogeneity of ventricular activation 72. In addition, Ardell and colleagues 73 recently showed that in a canine model, stellate decentralization, which interrupts both efferent and afferent sympathetic neurotransmission from the brain and spinal cord, reduced norepinephrine release during ventricular ischemia and increased survival from VF from 72% to 92%. It is important to note that the beneficial effects of CSD are mediated not only by blocking efferent sympathetic tone, but also by interrupting cardiac afferent tone, This may have beneficial effect by improving parasympathetic tone, reducing efferent sympathetic outflow, and improving cardiac remodeling in heart failure 74-76.
Renal Sympathetic Denervation
RDN has been described as a novel catheter-based therapy for treatment of refractory Figure 1 VT. Originally developed for resistant hypertension, RDN has been shown to reduce systemic sympathetic activity, as evidenced by a 42% reduction in whole-body norepinephrine spillover, reduction in renal sympathetic efferent nerve activity, and significant improvement in insulin resistance 77,78.
When initially reported in two heart failure patients presenting with electrical storm, RDN resulted in acute reduction in arrhythmic burden, with minimal effects on basal hemodynamic parameters 79. Subsequent small case series have suggested similar safety and efficacy in both ischemic and non-ischemic cardiomyopathy patients, with sustained results observed at 6 months of follow-up 80,81. In a retrospective matched cohort study of 32 patients who underwent catheter ablation alone or a combination of ablation and RDN, there was a significant decrease in both arrhythmic burden and associated ICD therapies in patients who received RDN as an adjuvant therapy 82. In another series of 10 patients who underwent RDN after prior catheter ablation and CSD (9 bilateral and 1 left sided), patients who demonstrated an initial response to CSD and underwent a staged outpatient RDN had a marked reduction in VT/VF burden and associated ICD therapies at a median follow-up of 28 months. In contrast, patients who required salvage RDN in the acute setting did not appear to derive significant benefit, suggesting that RDN may be more beneficial when performed earlier in the course of disease 83. Relevant studies are provided in Table 1.
The mechanisms by which RDN reduces VAs have been elucidated by a number of studies in animal models. RDN significantly reduced spontaneous premature ventricular contractions (PVCs) and the incidence of VF during ischemia 84. In a chronic porcine infarct model, RDN performed 2 weeks after MI reduced sympathetic co-transmitter NPY and nerve growth factor levels in the infarcted myocardium, reduced ventricular sympathetic nerve density, and reduced occurrence of spontaneous VAs 85. In rodents, RDN was shown to reduce VA inducibility, myocardial fibrosis, and sympathetic neural remodeling after chronic MI 86. In a recently published study, RDN in a canine model of chronic MI resulted in prolonged ventricular ERP, increased VF threshold, flattened restitution curves, and decreased VAs 87. Furthermore, tissue analysis in the RDN group revealed a significant decrease in neural remodeling of both the heart and bilateral stellate ganglia.
Procedurally, RDN is performed percutaneously under fluoroscopic and electroanatomic mapping guidance. Selective renal angiography is performed at the beginning of the procedure via femoral arterial access to assess the anatomy and confirm its eligibility. Lesion sets are delivered with alternating deflections from the distal renal artery (prior to the bifurcation) to the renal artery ostium proximally. Following ablation, a repeat renal angiogram is performed to confirm uninterrupted flow through the bilateral renal arteries. Some of the heterogeneity in the outcomes of the procedure are likely due to anatomical variability, including greater distal innervation of the artery, where radiofrequency energy is generally avoided due to the possibility of renal artery stenosis, as well as variable presence of surrounding structures, such as small blood vessels that may lead to a cooling effect and lymph nodes that can serve has heat sinks for radiofrequency energy 88,89.
The sympathetic and parasympathetic nervous systems are intricately involved in the modulation of VAs. Cardiac disease leads to overall sympathoexcitation, which combined with structural remodeling, lead to the electrophysiologic substrate necessary for occurrence and maintenance of VAs. A growing body of clinical and preclinical research over the past several decades have led to major advances in our ability to understand and target neural and humoral contributors to these life-threatening arrhythmias. Prospective randomized trials to evaluate neuraxial modulation for treatment of VAs in SHD are needed and ongoing.
-
Vaseghi M, Shivkumar K. The role of the autonomic nervous system in sudden cardiac death. Prog Cardiovasc Dis 2008;50:404-19.
- Wu P, Vaseghi M. The autonomic nervous system and ventricular arrhythmias in myocardial infarction and heart failure. Pacing Clin Electrophysiol 2020;43:172-180.
- Shivkumar K, Ajijola OA, Anand I et al. Clinical neurocardiology defining the value of neuroscience-based cardiovascular therapeutics. J Physiol 2016;594:3911-54.
- Huang WA, Boyle NG, Vaseghi M. Cardiac Innervation and the Autonomic Nervous System in Sudden Cardiac Death. Card Electrophysiol Clin 2017;9:665-679.
- Tan CMJ, Green P, Tapoulal N, Lewandowski AJ, Leeson P, Herring N. The Role of Neuropeptide Y in Cardiovascular Health and Disease. Front Physiol 2018;9:1281.
- Ullman B, Hulting J, Lundberg JM. Prognostic value of plasma neuropeptide-Y in coronary care unit patients with and without acute myocardial infarction. Eur Heart J 1994;15:454-61.
- Ajijola OA, Chatterjee NA, Gonzales MJ et al. Coronary Sinus Neuropeptide Y Levels and Adverse Outcomes in Patients With Stable Chronic Heart Failure. JAMA Cardiol 2019.
- Hoang JD, Salavatian S, Yamaguchi N, Amer Swid M, Vaseghi M. Cardiac sympathetic activation circumvent high dose beta-blocker therapy in part through the release of neuropeptide Y. JCI Insight, 2020. In press.
- Lundberg JM, Franco-Cereceda A, Lacroix JS, Pernow J. Neuropeptide Y and sympathetic neurotransmission. Ann N Y Acad Sci 1990;611:166-74.
- Han J, Garciadejalon P, Moe GK. Adrenergic Effects on Ventricular Vulnerability. Circ Res 1964;14:516-24.
- Kalla M, Hao G, Tapoulal N et al. The cardiac sympathetic co-transmitter neuropeptide Y is pro-arrhythmic following ST-elevation myocardial infarction despite beta-blockade. Eur Heart J 2019.
- Priori SG, Mantica M, Schwartz PJ. Delayed afterdepolarizations elicited in vivo by left stellate ganglion stimulation. Circulation 1988;78:178-85.
- Ben-David J, Zipes DP. Differential response to right and left ansae subclaviae stimulation of early afterdepolarizations and ventricular tachycardia induced by cesium in dogs. Circulation 1988;78:1241-50.
- Coumel P. Cardiac arrhythmias and the autonomic nervous system. J Cardiovasc Electrophysiol 1993;4:338-55.
- Opthof T, Coronel R, Vermeulen JT, Verberne HJ, van Capelle FJ, Janse MJ. Dispersion of refractoriness in normal and ischaemic canine ventricle: effects of sympathetic stimulation. Cardiovasc Res 1993;27:1954-60.
- Opthof T, Dekker LR, Coronel R, Vermeulen JT, van Capelle FJ, Janse MJ. Interaction of sympathetic and parasympathetic nervous system on ventricular refractoriness assessed by local fibrillation intervals in the canine heart. Cardiovasc Res 1993;27:753-9.
- Yagishita D, Chui RW, Yamakawa K et al. Sympathetic nerve stimulation, not circulating norepinephrine, modulates T-peak to T-end interval by increasing global dispersion of repolarization. Circ Arrhythm Electrophysiol 2015;8:174-85.
- Cao JM, Chen LS, KenKnight BH et al. Nerve sprouting and sudden cardiac death. Circ Res 2000;86:816-21.
- Cao JM, Fishbein MC, Han JB et al. Relationship between regional cardiac hyperinnervation and ventricular arrhythmia. Circulation 2000;101:1960-9.
- Dae MW. Imaging of myocardial sympathetic innervation with metaiodobenzylguanidine. J Nucl Cardiol 1994;1:S23-30.
- Fallavollita JA, Heavey BM, Luisi AJ, Jr. et al. Regional myocardial sympathetic denervation predicts the risk of sudden cardiac arrest in ischemic cardiomyopathy. J Am Coll Cardiol 2014;63:141-9.
- Rabinovitch MA, Rose CP, Rouleau JL et al. Metaiodobenzylguanidine [131I] scintigraphy detects impaired myocardial sympathetic neuronal transport function of canine mechanical-overload heart failure. Circ Res 1987;61:797-804.
- Vaseghi M, Lux RL, Mahajan A, Shivkumar K. Sympathetic stimulation increases dispersion of repolarization in humans with myocardial infarction. Am J Physiol Heart Circ Physiol 2012;302:H1838-46.
- Barber MJ, Mueller TM, Henry DP, Felten SY, Zipes DP. Transmural myocardial infarction in the dog produces sympathectomy in noninfarcted myocardium. Circulation 1983;67:787-96.
- Clemson B, Gaul L, Gubin SS et al. Prejunctional angiotensin II receptors. Facilitation of norepinephrine release in the human forearm. J Clin Invest 1994;93:684-91.
- Donoghue M, Wakimoto H, Maguire CT et al. Heart block, ventricular tachycardia, and sudden death in ACE2 transgenic mice with downregulated connexins. J Mol Cell Cardiol 2003;35:1043-53.
- Gerling IC, Sun Y, Ahokas RA et al. Aldosteronism: an immunostimulatory state precedes proinflammatory/fibrogenic cardiac phenotype. Am J Physiol Heart Circ Physiol 2003;285:H813-21.
- Leenen FH. Brain mechanisms contributing to sympathetic hyperactivity and heart failure. Circ Res. United States, 2007:221-3.
- Reid IA. Interactions between ANG II, sympathetic nervous system, and baroreceptor reflexes in regulation of blood pressure. Am J Physiol 1992;262:E763-78.
- Bourke T, Vaseghi M, Michowitz Y et al. Neuraxial modulation for refractory ventricular arrhythmias: value of thoracic epidural anesthesia and surgical left cardiac sympathetic denervation. Circulation 2010;121:2255-62.
- Hasenbos M, Liem TH, Kerkkamp H, Gielen M. The influence of high thoracic epidural analgesia on the cardiovascular system. Acta Anaesthesiol Belg 1988;39:49-54.
- Blomberg S, Ricksten SE. Thoracic epidural anaesthesia decreases the incidence of ventricular arrhythmias during acute myocardial ischaemia in the anaesthetized rat. Acta Anaesthesiol Scand 1988;32:173-8.
- Meissner A, Eckardt L, Kirchhof P et al. Effects of thoracic epidural anesthesia with and without autonomic nervous system blockade on cardiac monophasic action potentials and effective refractoriness in awake dogs. Anesthesiology 2001;95:132-8; discussion 6A.
- Hoang J, Swid M, Kang K, Chan C, Vaseghi M. Autonomic And Electrophysiological Effects Of Thoracic Epidural Anesthesia On Infarcted Porcine Hearts. Heart Rhythm Scientific Sessions. San Diego, CA, 2020.
- Mahajan A, Moore J, Cesario DA, Shivkumar K. Use of thoracic epidural anesthesia for management of electrical storm: a case report. Heart Rhythm 2005;2:1359-62.
- Do DH, Bradfield J, Ajijola OA et al. Thoracic Epidural Anesthesia Can Be Effective for the Short-Term Management of Ventricular Tachycardia Storm. J Am Heart Assoc 2017;6.
- Leriche R, Fontaine R. L'anesthesie isolee du ganglion etoile: sa technique, ses indications, ses resultats. Presse Méd 1934;42:849-50.
- Gu Y, Wang L, Wang X, Tang Y, Cao F, Fang Y. Assessment of ventricular electrophysiological characteristics at periinfarct zone of postmyocardial infarction in rabbits following stellate ganglion block. J Cardiovasc Electrophysiol 2012;23 Suppl 1:S29-35.
- Fudim M, Boortz-Marx R, Patel CB, Sun AY, Piccini JP. Autonomic Modulation for the Treatment of Ventricular Arrhythmias: Therapeutic Use of Percutaneous Stellate Ganglion Blocks. J Cardiovasc Electrophysiol 2017;28:446-449.
- Hayase J, Patel J, Narayan SM, Krummen DE. Percutaneous stellate ganglion block suppressing VT and VF in a patient refractory to VT ablation. J Cardiovasc Electrophysiol 2013;24:926-8.
- Tan AY, Abdi S, Buxton AE, Anter E. Percutaneous stellate ganglia block for acute control of refractory ventricular tachycardia. Heart Rhythm 2012;9:2063-7.
- Cardona-Guarache R, Padala SK, Velazco-Davila L et al. Stellate ganglion blockade and bilateral cardiac sympathetic denervation in patients with life-threatening ventricular arrhythmias. J Cardiovasc Electrophysiol 2017;28:903-908.
- Fudim M, Boortz-Marx R, Ganesh A et al. Stellate ganglion blockade for the treatment of refractory ventricular arrhythmias: A systematic review and meta-analysis. J Cardiovasc Electrophysiol 2017;28:1460-1467.
- Meng L, Tseng CH, Shivkumar K, Ajijola O. Efficacy of Stellate Ganglion Blockade in Managing Electrical Storm: A Systematic Review. JACC Clin Electrophysiol 2017;3:942-949.
- Tian Y, Wittwer ED, Kapa S et al. Effective Use of Percutaneous Stellate Ganglion Blockade in Patients With Electrical Storm. Circ Arrhythm Electrophysiol 2019;12:e007118.
- Fudim M, Qadri YJ, Waldron NH et al. Stellate Ganglion Blockade for the Treatment of Refractory Ventricular Arrhythmias. JACC: Clinical Electrophysiology 2020:1089.
- Nademanee K, Taylor R, Bailey WE, Rieders DE, Kosar EM. Treating electrical storm : sympathetic blockade versus advanced cardiac life support-guided therapy. Circulation 2000;102:742-7.
- Schwartz PJ. Cardiac sympathetic denervation to prevent life-threatening arrhythmias. Nat Rev Cardiol 2014;11:346-53.
- Jonnesco T. Traitement chirurgical de l'angine de poitrine par la rèsection du sympathique cervico-thoracique. Presse Med 1921;20:193-194.
- Estes EH, Jr., Izlar HL, Jr. Recurrent ventricular tachycardia. A case successfully treated by bilateral cardiac sympathectomy. Am J Med 1961;31:493-7.
- Zipes DP, Festoff B, Schaal SF, Cox C, Sealy WC, Wallace AG. Treatment of ventricular arrhythmia by permanent atrial pacemaker and cardiac sympathectomy. Ann Intern Med 1968;68:591-7.
- Schwartz PJ, Priori SG, Cerrone M et al. Left cardiac sympathetic denervation in the management of high-risk patients affected by the long-QT syndrome. Circulation 2004;109:1826-33.
- Collura CA, Johnson JN, Moir C, Ackerman MJ. Left cardiac sympathetic denervation for the treatment of long QT syndrome and catecholaminergic polymorphic ventricular tachycardia using video-assisted thoracic surgery. Heart Rhythm 2009;6:752-9.
- Coleman MA, Bos JM, Johnson JN et al. Videoscopic left cardiac sympathetic denervation for patients with recurrent ventricular fibrillation/malignant ventricular arrhythmia syndromes besides congenital long-QT syndrome. Circ Arrhythm Electrophysiol 2012;5:782-8.
- Schwartz PJ, Locati EH, Moss AJ, Crampton RS, Trazzi R, Ruberti U. Left cardiac sympathetic denervation in the therapy of congenital long QT syndrome. A worldwide report. Circulation 1991;84:503-11.
- Bos JM, Bos KM, Johnson JN, Moir C, Ackerman MJ. Left cardiac sympathetic denervation in long QT syndrome: analysis of therapeutic nonresponders. Circ Arrhythm Electrophysiol 2013;6:705-11.
- Olde Nordkamp LR, Driessen AH, Odero A et al. Left cardiac sympathetic denervation in the Netherlands for the treatment of inherited arrhythmia syndromes. Neth Heart J 2014;22:160-6.
- Jang SY, Cho Y, Kim NK et al. Video-Assisted Thoracoscopic Left Cardiac Sympathetic Denervation in Patients with Hereditary Ventricular Arrhythmias. Pacing Clin Electrophysiol 2017;40:232-241.
- Roston TM, Vinocur JM, Maginot KR et al. Catecholaminergic polymorphic ventricular tachycardia in children: analysis of therapeutic strategies and outcomes from an international multicenter registry. Circ Arrhythm Electrophysiol 2015;8:633-42.
- De Ferrari GM, Dusi V, Spazzolini C et al. Clinical Management of Catecholaminergic Polymorphic Ventricular Tachycardia: The Role of Left Cardiac Sympathetic Denervation. Circulation 2015;131:2185-93.
- Priori SG, Wilde AA, Horie M et al. HRS/EHRA/APHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes: document endorsed by HRS, EHRA, and APHRS in May 2013 and by ACCF, AHA, PACES, and AEPC in June 2013. Heart Rhythm 2013;10:1932-63.
- Vaseghi M, Zhou W, Shi J et al. Sympathetic innervation of the anterior left ventricular wall by the right and left stellate ganglia. Heart Rhythm 2012;9:1303-9.
- Opthof T, Misier AR, Coronel R et al. Dispersion of refractoriness in canine ventricular myocardium. Effects of sympathetic stimulation. Circ Res 1991;68:1204-15.
- Schwartz PJ, Stone HL, Brown AM. Effects of unilateral stellate ganglion blockade on the arrhythmias associated with coronary occlusion. Am Heart J 1976;92:589-99.
- Puddu PE, Jouve R, Langlet F, Guillen JC, Lanti M, Reale A. Prevention of postischemic ventricular fibrillation late after right or left stellate ganglionectomy in dogs. Circulation 1988;77:935-46.
- Ajijola OA, Yagishita D, Reddy NK et al. Remodeling of stellate ganglion neurons after spatially targeted myocardial infarction: Neuropeptide and morphologic changes. Heart Rhythm 2015;12:1027-35.
- Vaseghi M, Gima J, Kanaan C et al. Cardiac sympathetic denervation in patients with refractory ventricular arrhythmias or electrical storm: intermediate and long-term follow-up. Heart Rhythm 2014;11:360-6.
- Vaseghi M, Barwad P, Malavassi Corrales FJ et al. Cardiac Sympathetic Denervation for Refractory Ventricular Arrhythmias. J Am Coll Cardiol 2017;69:3070-3080.
- Dusi V, Gornbein J, Sorg J et al. Quantifying the benefit of cardiac sympathetic denervation in patients with monomorphic ventricular tachycardia referred for catheter ablation: an effect size analysis. American Heart Association Scientific Sessions. Philadelphia, PA: Circulation, 2019:A13872.
- Saenz LC, Corrales FM, Bautista W et al. Cardiac sympathetic denervation for intractable ventricular arrhythmias in Chagas disease. Heart Rhythm 2016;13:1388-94.
- Okada DR, Assis FR, Gilotra NA et al. Cardiac sympathectomy for refractory ventricular arrhythmias in cardiac sarcoidosis. Heart Rhythm 2019;16:1408-1413.
- Irie T, Yamakawa K, Hamon D, Nakamura K, Shivkumar K, Vaseghi M. Cardiac sympathetic innervation via middle cervical and stellate ganglia and antiarrhythmic mechanism of bilateral stellectomy. Am J Physiol Heart Circ Physiol 2017;312:H392-h405.
- Ardell JL, Foreman RD, Armour JA, Shivkumar K. Cardiac sympathectomy and spinal cord stimulation attenuate reflex-mediated norepinephrine release during ischemia preventing ventricular fibrillation. JCI Insight 2019;4.
- Gao L, Zhu Z, Zucker IH, Wang W. Cardiac sympathetic afferent stimulation impairs baroreflex control of renal sympathetic nerve activity in rats. Am J Physiol Heart Circ Physiol 2004;286:H1706-11.
- Gao L, Schultz HD, Patel KP, Zucker IH, Wang W. Augmented input from cardiac sympathetic afferents inhibits baroreflex in rats with heart failure. Hypertension 2005;45:1173-81.
- Wang HJ, Wang W, Cornish KG, Rozanski GJ, Zucker IH. Cardiac sympathetic afferent denervation attenuates cardiac remodeling and improves cardiovascular dysfunction in rats with heart failure. Hypertension 2014;64:745-55.
- Schlaich MP, Sobotka PA, Krum H, Lambert E, Esler MD. Renal sympathetic-nerve ablation for uncontrolled hypertension. N Engl J Med. United States, 2009:932-4.
- Mahfoud F, Schlaich M, Kindermann I et al. Effect of renal sympathetic denervation on glucose metabolism in patients with resistant hypertension: a pilot study. Circulation 2011;123:1940-6.
- Ukena C, Bauer A, Mahfoud F et al. Renal sympathetic denervation for treatment of electrical storm: first-in-man experience. Clin Res Cardiol 2012;101:63-7.
- Armaganijan LV, Staico R, Moreira DA et al. 6-Month Outcomes in Patients With Implantable Cardioverter-Defibrillators Undergoing Renal Sympathetic Denervation for the Treatment of Refractory Ventricular Arrhythmias. JACC Cardiovasc Interv 2015;8:984-90.
- Remo BF, Preminger M, Bradfield J et al. Safety and efficacy of renal denervation as a novel treatment of ventricular tachycardia storm in patients with cardiomyopathy. Heart Rhythm 2014;11:541-6.
- Evranos B, Canpolat U, Kocyigit D, Coteli C, Yorgun H, Aytemir K. Role of Adjuvant Renal Sympathetic Denervation in the Treatment of Ventricular Arrhythmias. Am J Cardiol 2016;118:1207-1210.
- Bradfield JS, Hayase J, Liu K et al. Renal denervation as adjunctive therapy to cardiac sympathetic denervation for ablation refractory ventricular tachycardia. Heart Rhythm 2020;17:220-227.
- Linz D, Wirth K, Ukena C et al. Renal denervation suppresses ventricular arrhythmias during acute ventricular ischemia in pigs. Heart Rhythm 2013;10:1525-30.
- Jackson N, Gizurarson S, Azam MA et al. Effects of Renal Artery Denervation on Ventricular Arrhythmias in a Postinfarct Model. Circ Cardiovasc Interv 2017;10:e004172.
- Jiang W, Chen C, Huo J et al. Comparison between renal denervation and metoprolol on the susceptibility of ventricular arrhythmias in rats with myocardial infarction. Sci Rep 2018;8:10206.
- Zhang WH, Zhou QN, Lu YM et al. Renal Denervation Reduced Ventricular Arrhythmia After Myocardial Infarction by Inhibiting Sympathetic Activity and Remodeling. J Am Heart Assoc 2018;7:e009938.
- Sakakura K, Ladich E, Cheng Q et al. Anatomic assessment of sympathetic peri-arterial renal nerves in man. J Am Coll Cardiol 2014;64:635-43.
- Tzafriri AR, Keating JH, Markham PM et al. Arterial microanatomy determines the success of energy-based renal denervation in controlling hypertension. Sci Transl Med 2015;7:285ra65.