ICE-Derived Left Atrial and Left Ventricular Endocardial and Myocardial Speckle Tracking Strain Patterns in Atrial Fibrillation at the Time of Radiofrequency Ablation
Jian-Fang Ren1, Shiquan Chen12, David J. Callans1, Qiang Liu2, Gregory Supple1, David S. Frankel1, Pasquale Santangeli1, Ruhong Jiang2, David Lin1, Matthew Hyman1, Lu Yu2, Michael Riley1, Yaxun Sun2, Zuwen Zhang2, Chan Yu2, Robert D. Schaller1, Sanjay Dixit1, Bei Wang2, Chenyang Jiang2, Francis E. Marchlinski1
1Cardiac Electrophysiology Laboratory, Cardiovascular Medicine Division, University of Pennsylvania Health System, Philadelphia, PA, USA.2Department of Cardiology, Sir Run Run Shaw Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang, China.
Intracardiac echocardiography(ICE) has excellent imaging resolution and border recognition which increase strain measurement accuracy. We hypothesized that left atrial(LA) substrate and functional impairment can be detected by measuring LA strain deformation in patients with persistent and paroxysmal atrial fibrillation(AF), as compared to those with no AF. Strain deformation changes in LA and left ventricle(LV) can also be assessed post-ablation to determine its effect.
ICE-derived speckle tracking strain(STS) was prospectively performed in 96 patients, including 62 patients with AF(31 persistent and 31 paroxysmal AF) pre-/post-ablation, and 34 patients with no AF. We measured major strain parameters including longitudinal segmental(endo/myocardial) “average peak overall strain of all segments”(PkAll), peak strain rate(SR),and different time-to-peak strain in LA and LV images.
At baseline, persistent AF patients had significantly lower(p<0.01) LA endocardial(4.3±2.5 vs. 20.3±8.9 and 25.5±12.9 %) and myocardial PkAll(4.4±2.6 vs. 15.7±7.2 and 20.9±9.2 %), endocardial(0.9±0.4 vs. 1.8±0.7 and 2.2±0.6 1/s) and myocardial peak SR(0.7±0.4 vs. 1.5±0.6 and 1.9±0.5 1/s), as compared to paroxysmal AF and no AF patients. After successful ablation, endo-/myocardial LA PkAll and peak SR were significantly improved, most dramatically in patients with persistent AF. LV endocardial/myocardial strain and SR also improved in AF patients post-ablation.
LA longitudinal strain(%)/SR(1/s) parameters in AF patients are more abnormal than those with no AF, suggesting LA substrate/functional damage. AF ablation improved LA strains/SR but with values in paroxysmal > persistent AF suggesting background LA damage in persistent AF.
Key Words : Atrial fibrillation, Catheter ablation, Intracardiac echocardiography, Speckle tracking strain Imaging.
Jian-Fang Ren, MD
Cardiac Electrophysiology Laboratory
Division of Cardiovascular Medicine Hospital of the University of Pennsylvania 9 Founders Pavilion, Cardiology, 3400 Spruce Street
Philadelphia, PA 19104
Speckle tracking strain (STS) is a unitless measurement of dimensional or deformational change. Using image-processing algorithms for routine 2-dimensional digital echocardiographic images, small stable myocardial footprints, or speckles, generated by ultrasound-myocardial tissue interactions are identified within a defined region of interest. Tracked frame-to-frame over the cardiac cycle, distances between speckles or their spatiotemporal displacement (regional strain velocity vectors) provide non-Doppler information about global and segmental myocardial deformation.1-2 Left atrial (LA) and ventricular (LV) STS echocardiography has been used for the assessment of LA and LV function and deformation.3-7 In contrast to LA “strain” assessed with tissue Doppler imaging by transthoracic echocardiography (TTE) 8 which is Doppler angle-dependent, STS is basically angle-independent, and thus less susceptible to the limitations of Doppler echocardiographic assessment of strain.9-10. To challenge STS being angle-independent, one study using TTE with comparison of longitudinal peak systolic strain in standard four-chamber apical, parasternal, subcostal and off-axis apical imaging views reported that TTE-derived longitudinal peak strain values were modestly dependent on angle of insonation and target depth.11. However, based on their methodology the study may emphasize that STS echocardiography needs standard imaging views with higher ultrasonic resolution and measurement reproducibility, rather than angle and depth dependence. LA/LV STS has become a research-based measurement that relates to LA and LV deformation and has been described to be altered in patients with atrial fibrillation (AF)12-13 and many other pathophysiological conditions [1-2, 5-7]. At present, STS software designed and provided for LA and LV STS measurements and imaging views are designed for use by TTE. As compared to TTE, intracardiac echocardiography (ICE) can provide better views for LA and LV imaging with improved resolution and border recognition.14-15 We hypothesized that ICE-derived LA and LV STS measurements may provide accurate quantitative evaluation of myocardial and endocardial deformations which compares deformation to original length, such as longitudinal strain and allows for discrimination between normal active myocardial segmental deformation versus passive displacement of a dysfunctional myocardial segment due to adjacent segment tethering and global cardiac motion. The aim of this study was to determine: A) the effect of AF on LA strain and strain rate; and B) the effect of radiofrequency (RF) AF ablation on LA and LV strain and strain rate measurements.
This was a prospective ICE STS study. The study population of total 96 patients comprised 62 patients with AF and 34 patients with no-AF with relatively normal cardiac structure undergoing first RF AF ablation procedure at the Hospital of the University of Pennsylvania, Philadelphia or the Sir Run Run Shaw Hospital of the Zhejiang University, Hangzhou, China. These patients underwent detailed ICE imaging as part of their procedures. All patients gave written informed consent in accordance with the institutional guidelines of each of the University Health Systems. Patients with previous TTE study or baseline ICE studies with moderate or severe mitral regurgitation and/or LV ejection fraction (EF) lower than 30 % were excluded from this study.
ICE imaging was performed using a Siemens SC-2000 machine and an intracardiac ultrasound catheter with 8 or 10 Fr (Biosense, Siemens-Acuson, Mountain view, CA) advanced through the femoral vein into right atrium (RA) and right ventricle (RV). As previously described 14,16 at baseline tricuspid regurgitation was assessed for estimation of pulmonary artery systolic pressure (PASP) in the RV inflow and outflow view. LA diameter and mitral inflow peak (E and/or A) velocity, LA EF and emptying volume (LAEV) were measured in the LV mitral inflow view. Posterolateral mitral annual early diastolic (e’) peak velocity was measured using Doppler tissue imaging (supplemental figure S1). The peak velocity E or E/A, and E/e’ was calculated for assessing diastolic function and estimating LV filling pressure 17. LV end-diastolic and end-systolic volume, stroke volume, LVEF and LV cardiac output (CO) were assessed in the LV long-axis view with the ICE transducer placed in the RV.
The LA and LV STS were measured using SC2000_eSie VVI (Siemens, Mountain View, CA) for analysis of endo- and myocardial layer strain parameters including segmental longitudinal (endo- and myocardial) “average peak overall strain of all segments” (PkAll), peak end-systolic strain (ESS), overall time-to-peak strain (TPkOvrl), peak strain rate (SR), and time-to-peak systolic SR (TPk), in the LA and LV longitudinal chamber views. The ICE transducer was first positioned at the high RA just adjacent to the junction of the LA roof and superior limbus as the key location to obtain LA longitudinal view for LA STS measurements [Figure 1]. After obtaining mitral inflow view, the transducer is then rotated slightly clockwise to obtain the above-mentioned LA view for STS measurements. This mage consistently includes the posterior mitral annulus and adjacent LA wall instead of the mitral valve inflow structures. The anatomic reference of this LA view for LA STS analysis included three segments: LA roof (with superior limbus)/anterior lateral wall, medial/interatrial septal wall and posterior annulus wall ([Figure 1] and supplemental video clip S2). With the transducer positioned within the RV, LV longitudinal with mitral inflow view was obtained. The anatomic reference of this LV view for LV STS study included six segments: LV posterior wall, inferolateral wall, anterolateral apical (with its papillary muscle), septal apical, and mid and basal septal segments ([Figure 2] and supplemental video clip S3). To avoid the foreshortening of the LV apex the ICE transducer was advanced along the interventricular septum to as near the RV apex as possible. Each LA STS parameter was measured, defined as the average STS measurement of the three segments of the LA imaging view. Each LV STS parameter was measured, defined as the average STS measurement of the six segments of the LV long-axis view. The absolute values of averaged STS measurements were adopted for statistical analysis. Image acquisition was performed with frame rate of 50-90 fps (acoustic clip capture with extended R-R) to ensure high quality. The LA and LV endocardial borders were traced manually and the epicardial borders were created by automated recognition with 5 mm for the LA wall thickness and 10 mm for the LV wall thickness. Based on the actual ICE imaging measurements of the wall thickness, manual correction and visual tracking accuracy of wall thickness and wall contour movement during cardiac cycles were performed in each case. LV papillary muscles were not included in the region of interest.
Figure 1. 1. ICE-derived LA image and LA strain analysis in a patient (male age 62) with paroxysmal AF. The LA image was obtained with the ICE transducer placed at the junction of the RA and superior vena cava with light clockwise rotation from LV inflow into the LA view; LA strain analysis is performed for the 3-segment (Seg) including LA roof/anterior lateral wall (Seg 1, representated by green line), medial/interatrial septal wall (Seg 2, white line) and posterior annulus wall (Seg 3, with blue line ) (supplemental video clip S2); Longitudinal endocardial and myocardial strains were analyzed with the average overall peak value of all segments “Avg(s)” adopted for further statistical analysis including peak overall strain (PkAll), overall time-to-peak strain (TPkOvrl), peak (Pk) and time-to-peak systolic (TPk) strain rate (SR) parameters. Endo=endocardium; Myo=myocardium; SD=standard deviation.
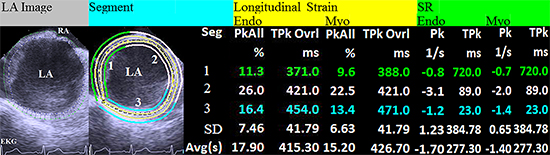
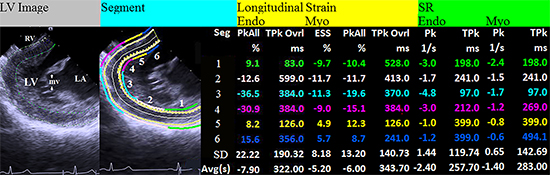
Figure 2. 2. ICE-derived LV image and LV strain analysis in a patient (female, age 70) with paroxysmal AF. The LV longitudinal image was obtained with the ICE transducer placed in the RV; LV strain analysis is performed for the 6-segment (Seg) including LV posterior (Seg 1, green line), inferolateral (Seg 2, white line), anterolateral apical (Seg 3, azure line), septal apical (Seg 4, pink line), mid septal (Seg 5, yellow line) and basal septal wall (Seg 6, blue line) (supplemental video clip S3); Longitudinal endocardial and myocardial strains were analyzed with the average overall peak value of all segments “Avg(s)” adopted for further statistical analysis including peak over strain (PkAll), overall time-to-peak strain (TPkOvrl), end systolic strain (ESS, myocardial), peak (Pk) and time-to-peak systolic (TPk) strain rate (SR) parameters. Endo= endocardium; Myo= myocardium; SD=standard deviation.
RF Ablation for Pulmonary Vein Isolation
Multipolar catheters were placed at the lateral RA and in the coronary sinus. Dual transseptal catheterization with ICE imaging guidance was performed to position a multipolar mapping catheter (Lasso™ or PentaRay) and a mapping/ablation catheter (Biosense Webster, Irvine or Diamond Bar, CA, USA) within the LA using standard techniques. Pulmonary vein (PV) isolation was performed at the LA/antral PV junction using previously described techniques.18-19 All patients received isoproterenol infusion (up to 6 to 20 µg/min) and/or adenosine bolus (up to 12 to 18 mg), in an attempt to provoke PV reconnection and non-PV triggers following PV isolation. Non-PV triggers were routinely targeted by focal ablation unless from superior vena caval or posterior LA origin in which case isolation was also performed. Acute ablation success was defined as the inability to demonstrate reconnected PVs or AF triggers from non-PVs sources in response to isoproterenol (and/or adenosine) and following cardioversion of induced AF during isoproterenol administration at the end of ablation procedure.
The values were expressed as mean±SD. Categorical data were summarized as frequencies and percentages. Differences in clinical and echocardiographic variables between groups were evaluated using unpaired or paired Student t test, and/or Wilcoxon signed-rank test (if the differences between pairs of observations are severely non-normally distributed)20 as appropriate. For categories variables, the chi-square test is used. Intra- and interobserver variations (%) were expressed as Meandifference / Meanboth measurements x 100 ± SDdifference / Meanboth measurements x 100.21-22 All statistical tests are 2-sided, and a p value <0.05 is considered significant.
Patient Characteristics and Treatment of AF
The study population included 96 patients divided into three groups: Group 1 - persistent AF (n=31), Group 2 - paroxysmal AF (n=31) and Group 3 - no AF (n=34, in which 27 with PVCs and 7 with supraventricular tachycardias of non-LA origin). Baseline characteristics of the study groups are presented in [Table 1]. Patients in Group 1 had a slightly higher age and PASP, a higher incidence of hypertension, more dilated LA inner dimension at end-systole (LAIDs) and lower LAEF/LAEV as compared to Group 2 and 3 patients at baseline. There were no significant differences between 3 groups regarding sex, body surface area, systolic blood pressure, LV inner dimension at end-diastole, LVEF and E/e’ parameters. There were significant differences among the Group 1/2 and Group 3 patients regarding resting heart rate, diastolic blood pressure and mitral peak flow velocity E, but all the average values of these parameters were within their normal and mildly variant ranges.
Table 1. Clinical Characteristics
|
p Value |
| Group |
Persistent AF 1(n-=31) |
Paroxysmal AF 2(n-=31) |
no AF 3(n-=34) |
1 vs. 2 |
1 vs. 3 |
2 vs. 3 |
| Age, yrs |
64±8 |
62±8 |
54±13 |
NS |
0.01 |
0.01 |
| Male (%) |
25 (81) |
23 (74) |
23 (68) |
NS |
NS |
NS |
| Body surface area, m² |
1.98±0.23 |
2.08±0.18 |
1.99±0.18 |
NS |
NS |
NS |
| Heart rate, beats/min |
75±15 |
65±13 |
74±10 |
0.04 |
NS |
0.03 |
| SBP, mmHg |
122±11 |
120±9 |
120±11 |
NS |
NS |
NS |
| DBP, mmHg |
73±10 |
67±8 |
66±9 |
0.04 |
0.03 |
NS |
| No heart disease (%) |
6 (19) |
13 (42) |
15 (44) |
NS |
0.04 |
NS |
| Hypertension (%) |
21 (68) |
14 (45) |
14 (41) |
NS |
0.04 |
NS |
| Other (%) |
4 (13) |
4 (13) |
5 (15) |
NS |
NS |
NS |
| Echocardiographic variables |
| LAIDs, cm |
4.9±0.4 |
4.4±0.3 |
4.3±0.3 |
0.01 |
0.01 |
NS |
| LAEF, % |
39±17 |
60±13 |
68±12 |
0.01 |
0.01 |
NS |
| LAEV, ml |
49±20 |
57±16 |
68±21 |
NS |
NS |
NS |
| LVIDd, cm |
5.0±0.5 |
4.8±0.4 |
4.8±0.5 |
NS |
NS |
NS |
| LVEF, % |
54±9 |
58±6 |
56±8 |
NS |
NS |
NS |
| E, cm/s |
70±11 |
61±11 |
72±13 |
0.02 |
NS |
0.04 |
| E/e’ |
9.4±2.5 |
8.2±2.8 |
8.7±3.6 |
NS |
NS |
NS |
| PASP, mmHg |
30±5 |
28±5 |
25±6 |
NS |
0.02 |
NS |
DBP and SBP=diastolic and systolic blood pressure; E=transmitral early diastolic Doppler flow velocity; e’=early diastolic tissue Doppler velocity sampling at posterolateral miitral annulus; LAEF=left atrial ejection fraction; LAEV=LA emptying volume; LAIDs=LA inner dimension at end-systole; LVIDd=left ventricular inner dimension at end-diastole; PASP=pulmonary artery systolic pressure.
At the end of the procedure Group 1 and 2 AF patients had acute ablation success and remained in sinus rhythm, as indicated by PV isolation and no inducible non-PV triggers in response to isoproterenol and/or adenosine and following cardioversion of induced AF at the end of ablation procedure. No patient had acute PV stenosis (based on ICE PV ostial flow velocity measurement <100 cm/s), embolic events or pericardial effusion with cardiac tamponade at the time of procedure.
LA STS Measurements Pre-RF Ablation in AF and No-AF Patients at Baseline
When comparing the LA STS measurements pre-RF ablation between AF patients in Group 1 and Group 2 [Table 2], patients with persistent AF (Group 1) had significantly lower LA longitudinal endocardial PkAll (4.3±2.5 vs. 20.3±8.9 %) and myocardial PkAll (4.4±2.6 vs. 15.7±7.2 %), LA longitudinal endocardial peak SR (0.9±0.4 vs. 1.8±0.7 1/s) and myocardial peak SR (0.7±0.4 vs. 1.5±0.6 1/s) (all p<0.05), whereas patients with paroxysmal AF in Group 2 had significantly longer LA longitudinal endocardial TPkOvrl (632±252 vs. 432±171 ms) and myocardial TPkOvrl (599±221 vs. 433±168 ms), LA longitudinal endocardial TPk SR (846±306 vs. 488±203 ms) and myocardial TPk SR (795±304 vs. 515±197 ms) (all p<0.05) pre-RF ablation.
Table 2. ICE LA STS Measurements Pre- and Post-RF Ablation in AF Versus Pre-RF Ablation in No AF Groups
|
p Value |
| Group |
Persistent AF 1 |
Paroxysmal AF 2 |
no AF 3 |
1 vs. 2 |
1 vs. 3 |
2 vs. 3 |
|
Pre |
Post |
Pre |
Post |
Pre |
Pre |
Pre |
Pre |
| Longitudinal Strain |
| Endo PkAll, % |
4.3±2.5 |
8.0±4.8* |
20.3±8.9 |
20.5±7.6 |
25.5±12.9 |
0.001 |
0.001 |
NS |
| TPkOvrl, ms |
432±171 |
418±101 |
632±252 |
379±65* |
476±128 |
0.02 |
NS |
NS |
| Myo PkAll, % |
4.4±2.6 |
6.5±3.2* |
15.7±7.2 |
17.3±6.0 |
20.9±9.2 |
0.001 |
0.001 |
NS |
| TPkOvrl, ms |
433±168 |
422±118 |
599±221 |
380±68* |
462±113 |
NS |
NS |
NS |
| Longitudinal SR |
| Endo SR Pk, 1/s |
0.9±0.4 |
1.2±0.5* |
1.8±0.7 |
2.1±0.8 |
2.2±0.6 |
0.001 |
0.001 |
0.04 |
| TPk, ms |
488±203 |
483±151 |
846±306 |
487±159* |
678±209 |
0.001 |
0.01 |
NS |
| Myo SR Pk, 1/s |
0.7±0.4 |
1.0±0.4* |
1.5±0.6 |
1.8±0.6 |
1.9±0.5 |
0.001 |
0.001 |
0.04 |
| TPk, ms |
515±179 |
480±126 |
795±304 |
494±124* |
680±162 |
0.003 |
0.01 |
NS |
*P ≤ 0.02 Post-RF vs. Pre-RF in AF group 1 or 2; NS = statistically no significance.
In comparing LA STS measurements pre-RF ablation between patients of AF Group 1, 2 and Group 3 at baseline, patients with persistent AF (Group 1) had the lowest LA longitudinal endocardial PkAll (4.3±2.5 vs. 25.5±12.9 %) and myocardial PkAll (4.4±2.6 vs. 20.9±9.2 %), LA longitudinal endocardial peak SR (0.9±0.4 vs. 2.2±0.6 1/s) and myocardial peak SR (0.7±0.4 vs. 1.9±0.5 1/s)(all p<0.05). Strain and SR parameters for paroxysmal AF (Group 2) pre-RF ablation, where modestly lower as compared to non-AF Group 3 at baseline, only lower endocardial (1.8±0.7 vs. 2.2±0.6 1/s) and myocardial SR (1.5±0.6 vs. 1.9±0.5 1/s) reached statistical significance (p<0.01).
LA STS measurements pre-RF ablation in AF Group (1 + 2) as compared to no AF Group 3 at baseline, showed similar results as the persistent AF Group 1, including significantly lower LA longitudinal endocardial and myocardial PkAll and peak SR, but not the endo-/myo-cardial TPkOvrl and SR TPk [Figure 3].
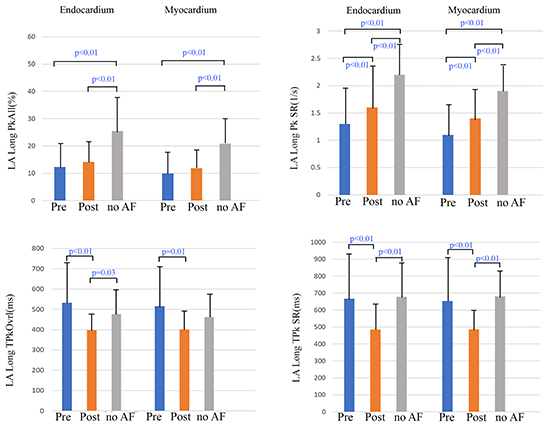
Figure 3. 3. ICE-derived LA STS Measurements (the bars expressed as mean + 1 SD) Pre- and Post-RF ablation in AF Versus Baseline in no AF Patients. LA STS measurements pre-RF ablation in AF group (1+2) as compared to no AF group at baseline, showed similar results as the no-AF versus persistent AF group 1, including significantly lower LA longitudinal (Long) endocardial and myocardial PkAll and peak (Pk) SR, but not the endo-/myocardial TPkOvrl and TPk SR parameters. LA PkAll (%) was only mildly increased, and only Long endo- and myocardial Pk SR (1/s) reached significantly post-RF ablation. However, there were significantly shortening of the LA Long endocardial and myocardial TPkOvrl and TPk SR in AF patients post-RF ablation. AF=atrial fibrillation; ICE=intracardiac echocardiography; LA=left atrium; PkAll=average peak overall strain of all segment; RF=radiofrequency; SR=strain rate; STS=speckle tracking strain; TPk=time-to-peak; TPkOvrl=overall time-to-peak strain.
Changes in LA STS Measurements Post-AF Ablation
Although patients with persistent AF (Group 1) had lowest strain and SR measurements pre-RF, improved LA endo/myocardial PkAll (%) and peak SR (1/s) were observed post successful RF ablation [Table 2]. In general, patients with AF (Group 1 + 2) had an increase of all LA strain and SR parameters with the endo-(1.3±0.7 vs. 1.6±0.8 1/s) and myocardial Pk SR (1.1±0.6 vs. 1.4±0.6 1/s) reaching significant differences (p<0.01) post-RF ablation [Figure 3]. These changes were coincident with a significant increase in LAEF (50±17 vs. 56±12%) and LACO (3344±1400 vs. 4557±1476 ml/min) post-RF. However, the increased values in AF patients post-RF were still significantly lower (p<0.01) than values in no-AF patients at baseline [Figure 3].
There were significant reductions of LA endo-(532±216 vs. 398±83 ms) and myocardial TPkOvrl (516±208 vs. 401±92 ms), and LA endo-(667±286 vs. 485±155 ms) and myocardial SR TPk (653±272 vs. 487±126 ms) (all p ≤ 0.01) post-RF ablation [Figure 3]. The differences in shortening of the TPkOvrl (ms) and SR TPk (ms) post-RF ablation reached significance in paroxysmal AF Group 2 but not the persistent AF Group 1 [Table 2].
LV STS Measurements Pre- and Post-AF Ablation
LV STS measurements pre- and post-RF ablation are listed in [Table 3]. Pre-RF ablation patients with persistent AF (Group 1) had significantly lower LV longitudinal endo- (10.9±4.5 vs. 15.8±5.4 %), myocardial GLS PkAll (7.6±3.7 vs. 10.6±4.0 %), endocardial PkAll (10.9±4.8 vs. 16.2±5.4 %) and myocardial ESS (7.1±3.4 vs. 9.8±3.5 %) as compared to those with paroxysmal AF (Group 2). Patients with AF (Group 1+2) after successful ablation had significant increase in endo- and myocardial strain (%) and the TPkOverl/SR TPk (ms) shortened [Table 3]. These improvements were coincident with a demonstrated increase in LVEF (39±17 vs. 51±12%) and LV CO (3566±1788 vs. 4302±1551 ml/min) (p<0.05).
Table 3. ICE LV STS Measurements Pre- and Post-RF Ablation in AF Groups
| Group |
Persistent AF 1 |
Paroxysmal AF 2 |
AF 1+2 |
|
Pre |
Post |
Pre |
Post |
Pre |
Post |
| Longitudinal Strain |
| Endo |
PkAll, % |
10.9±4.8† |
13.1±5.4‡ |
16.2±5.4 |
18.6±6.6* |
13.6±5.9 |
15.8±6.7* |
|
TPkOvrl, ms |
399±84 |
389±85‡ |
430±95 |
340±42* |
414±92 |
364±64* |
| Myo |
ESS, % |
7.1±3.4† |
8.2±3.3‡ |
9.8±3.5 |
11.0±4.7 |
8.5±3.6 |
9.7±4.1* |
|
PkAll, % |
8.5±3.6 |
9.2±3.9‡ |
10.6±4.2 |
12.4±4.7 |
9.5±3.9 |
10.8±4.6* |
|
TPkOvrl, ms |
384±77 |
359±65 |
431±93 |
408±90 |
342±45* |
351±55* |
| Longitudinal Strain Rate |
| Endo |
SR Pk, 1/s |
1.5±0.6 |
1.7±0.6‡ |
2.0±0.6 |
2.2±0.5 |
1.7±0.6 |
2.0±0.6 |
|
TPk, ms |
228±91 |
212±68 |
253±68 |
192±55* |
240±82 |
202±62* |
| Myo |
SR Pk, 1/s |
1.1±0.4 |
1.2±0.4‡ |
1.4±0.5 |
1.6±0.4 |
1.2±0.5 |
1.4±0.4 |
|
TPk, ms |
224±76 |
198±69 |
251±68 |
198±47* |
238±74 |
198±58* |
*p≤0.04: Post- versus Pre-RF in AF Group or AF Group 1+2; †p<0.04 Pre-RF: AF Group 1 versus AF Group 2; ‡p<0.04 Post-RF: AF Group 1 versus AF Group 2.
Intra- and Inter-observer Variation
Intra- and inter-observer (between JR and SC) variations for ICE-derived LA (n=20 measurements) and LV (n=23 measurements) strain parameters were assessed in 20 patients. The intra- and interobserver variations of major LA/LV longitudinal endo- and myocardial strain parameters were listed in [Table 4]. The intra-observer variations for LA strains were measured from 11±7 to 21±14%; and for LV strains, from 10±6 to 16±12%. The interobserver variations for LA strains were measured from 16±11 to 59±34%; and for LV strains, from 13±10 to 28±20%.
Table 4. Intra- and Inter-Observer Variation for LA and LV STS Parameters
| Variation |
Intraobserver % |
Interobserver % |
|
LA |
LV |
LA |
LV |
| Longitudinal Strain |
| Endo |
PkAll |
13±7 |
15±7 |
59±34 |
28±20 |
|
TPkOvrl |
19±15 |
10±6 |
23±18 |
15±14 |
| Myo |
PkAll |
11±7 |
16±9 |
43±35 |
28±22 |
|
TPkOvrl |
21±14 |
12±8 |
29±23 |
13±10 |
| Longitudinal SR |
| Endo SR Pk |
|
19±14 |
15±15 |
16±11 |
22±19 |
| Myo SR Pk |
|
19±11 |
10±9 |
19±14 |
18±15 |
*P ≤ 0.02 Post-RF vs. Pre-RF in AF group 1 or 2; NS = statistically no significance.
Novel therapeutic options such as RF catheter ablation techniques and transcatheter closure of the LA appendage or replacement of cardiac valves for severe regurgitation demand advanced imaging to maximize patient safety and procedural outcomes. ICE provides excellent imaging views and resolution for evaluation of cardiac structure and function. ICE has become a valuable and useful imaging tool during interventional electrophysiological procedures.14 In addition to our previously developed ICE-derived RV myocardial strain deformation patterns in arrhythmogenic cardiomyopathy23, this is the first prospectively 2D ICE-derived LA and LV STS evaluation, to our knowledge, in AF patients pre- and post-ablation procedure.
Lower LA STS Values at Baseline Can Identify Background LA Damage in AF Patients
Our findings demonstrate that at baseline LA longitudinal endocardial and myocardial average segmental PkAll strain (%) and peak SR (1/s) in patients with AF were significantly lower (average 52 and 42%, respectively) pre-RF ablation than those without AF using ICE-derived 2D STS. Lower PkAll and peak SR values were more marked in the persistent AF patients (81 and 61%, respectively) than the paroxysmal AF patients (22 and 20%). In this study, patients with AF were older age than those without AF. Patients with persistent AF had slightly larger LAIDs and lower LAEF/LAEV than those of paroxysmal AF and no AF patients. ICE derived E/e’ measurements (average <12 or 15) did not show a higher likelihood of increased LV filling pressure in the AF group.17 Based on evidence of only a mildly enlarged LA and normal PASP measurements, the AF group patients in this study probably represented a less advanced cardiac disease state. The modestly elevated PASP measurements especially in the persistent AF patients, probably still reflects more structural abnormalities as compared to without AF patients with a comparably dilated LA (p=0.02). LA dilation and myocardial fibrosis causing LA dysfunction and electromechanical conduction delay characterize the substrate for AF.24-25 Lower LA strain and SR measurements indicate LA substrate damage during remodeling coincident with LA dilation and increase in fibrosis in patients with AF. Our findings of lower LA strain and SR especially with persistent AF, may reflect LA substrate and functional damage with fibrosis (stiffness) rather than just LA dilation 26. A reduced LA reservoir strain (during LV systole) has been also shown to correlate with LA wall fibrosis determined by delayed-enhancement magnetic resonance imaging in patients with AF.27-28 Therefore, findings of lower strain and SR parameters pre-RF AF ablation in this study suggests their sensitivity in the identification of LA substrate changes due to AF.
Limited Improvement of LA Strain and SR in AF Patients after Ablation
This study showed that patients in both AF Groups demonstrated an increase in strain and SR parameters post-RF. Of note, LA endo-/myocardial strain and SR parameters significantly increased (p ≤ 0.02) after successful RF ablation in the persistent AF patients. This might be attributed to change in heart rhythm from AF to the sinus in the patients with persistent AF. However, these values although significantly increased from baseline are still significantly lower than those of paroxysmal AF and Non-AF patients, indicating more LA substrate and functional damage in persistent AF patients. TTE intra-LA vector flow mapping and tracking study revealed that successful AF catheter ablation slightly improves but does not reverse impaired LA intra-flow and mechanics after 3- and 6-month follow-up.13 Whether the pathophysiological mechanism for the changes observed relates to LA dyssynchrony and heterogeneous deformation remains to be further studied.23
Shortening in LA Time to Peak Strain and SR (ms) after AF ablation
The LA STS measurements of paroxysmal AF pre-RF ablation showed longer LA segmental longitudinal endo- and myocardial TPkOvrl (ms) and TPk SR (ms) than those of persistent AF. Nevertheless, immediately after RF ablation these longitudinal endo- and myocardial TPkOvrl and TPk SR were significantly shortened. A shortened and improved LA longitudinal endo- and myocardial TPkOvrl and SR TPk (ms) in the paroxysmal AF patients post RF ablation may reflect mild and reversible changes due to LA functional abnormalities from the AF. However, maintenance of a shortened time to peak strain and SR (ms) in the persistent AF patients post-RF ablation correlates with the maintenance of sinus rhythm and efficacy of RF catheter ablation. In addition, the changes in segmental atrial velocity and the magnitude and the rate of myocardial deformation during the cardiac cycle should also be considered.[29-30] Whether a prolongation or shortening of the time to peak strain and SR (ms) is related to LA changes in dimension, anatomic/electrophysiological function and/or extent of fibrosis needs to be further studied.
Improvement in LV STS Post-RF Ablation for AF
Our study demonstrated that the patients with AF, especially persistent AF had reduced LV EF and lower LV longitudinal endo- and myocardial strain (%) and SR (1/s) parameters pre-RF AF ablation. The AF may lead to adverse cardiac remodeling and may be primarily responsible for the development of a cardiomyopathy.31-32 However, our results showed that the majority of the LV longitudinal endo- and myocardial strain and SR parameters were improved immediately post acutely successful ablation as compared to results immediately pre-RF ablation. This may be a benign interaction between improvement of reservoir and contractile atrial and LV function immediately after ablation. These results suggest that careful assessment of individual changes in LVEF and STS parameters may provide critical prognostic and diagnostic value immediately after restoration of sinus rhythm.
This was a dual-center, prospective study using ICE imaging with a selected group of patients with AF undergoing first RF ablation procedure. ICE as an invasive catheter-based technique is not indicated for use in normal subjects. We selected a group of patients without AF but with PVCs and other arrhythmias of non-LA origin undergoing first RF ablation procedure, who had STS measurements at baseline as the reference population. The sample size was relatively small and patients with moderate or severe mitral regurgitation were excluded. The STS measurements in this study were obtained from a specific view of the LA and LV using 2D-ICE imaging. STS measurements from multiple views may increase its diagnostic value for more complicated structural heart disease, such as ischemic and nonischemic dilated cardiomyopathy. However, our results of STS measurements at pre-RF ablation have indicated that average segmental endo- and myocardial peak LA strain (%) and peak SR (1/s) parameters can clearly differentiate different severity of LA substrate and functional damage between the persistent and paroxysmal AF groups. Our findings also demonstrated that LA strain (%) and SR (1/s) did improve especially in the persistent AF patients post-RF ablation and furthermore, the majority of LV STS parameters were improved after acutely successful ablation. However, the LA strain values did not return to those observed in no AF patients, suggesting background LA damage,
The results of intraobserver and interobserver variations indicated that intraobserver variations were smaller (than interobserver variations) and acceptable as compared to the marked changes of STS measurements in patients with AF. At present, our ICE-derived STS measurements may not exactly compare with the other techniques especially using different strain software packages since a higher variability and reproducibility of segmental/regional strain measurement has been shown among 7 different vendors.33-34 Finally, STS echocardiography is a novel clinical technique which needs further development and study to confirm reproducibility of strain measurements and other factors that can influence measurements. Nevertheless, our results support these STS parameters as a potential marker of LA substrate and functional damage and an adjunctive measure of the mechanical effects that the LA sustains from AF.
ICE-derived speckle tracking LA longitudinal strain and SR parameters in AF patients, especially with persistent AF were significantly lower than those with no-AF patients, suggesting LA substrate and functional damage. AF ablation did improve those LA strain/SR parameters, especially in persistent AF patients, but still with lower values suggesting background LA damage. The majority of LV strain and SR were improved with successful ablation for AF.